Advanced Photocatalytic N-Alpha Arylation for Scalable Pharmaceutical Intermediate Manufacturing
Introduction to Next-Generation C-H Functionalization Technology
The landscape of organic synthesis is undergoing a paradigm shift towards greener, more efficient methodologies, particularly in the construction of complex carbon-carbon bonds essential for modern drug discovery. Patent CN114805107A introduces a groundbreaking approach to the N-alpha position arylation of nitrogen-containing heterocycles, utilizing a sophisticated dual catalytic system that merges photoredox catalysis with nickel-mediated cross-coupling. This technology addresses the longstanding challenge of direct C(sp3)-H functionalization without the need for harsh pre-activation or stoichiometric oxidants, which have traditionally plagued the synthesis of valuable pharmaceutical intermediates. By leveraging visible light irradiation at room temperature, this method offers a sustainable pathway to access structurally diverse N-alpha aryl compounds with high atom economy. For R&D directors and process chemists, this represents a significant leap forward in synthetic efficiency, enabling the rapid assembly of molecular scaffolds that were previously difficult or expensive to access through conventional thermal methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of C-H bonds at the alpha position of nitrogen heterocycles has been fraught with significant synthetic hurdles that impede efficient manufacturing. Traditional strategies often rely on the use of strong, stoichiometric bases to generate reactive enolates or require the installation of directing groups that necessitate additional synthetic steps for installation and subsequent removal. Furthermore, many classical cross-coupling protocols demand elevated temperatures and the use of expensive palladium catalysts, which can lead to issues with metal contamination in the final active pharmaceutical ingredient (API). The reliance on external Hydrogen Atom Transfer (HAT) reagents in previous radical approaches adds another layer of complexity and cost, as these reagents are often unstable, hazardous, or generate substantial chemical waste. These factors collectively contribute to higher production costs, longer lead times, and increased environmental burdens, making the commercial scale-up of complex pharmaceutical intermediates a challenging endeavor for supply chain managers.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN114805107A presents a streamlined, catalytic solution that operates under remarkably mild conditions. The core innovation lies in the synergistic combination of an iridium-based photoredox catalyst and a nickel catalyst, which together facilitate the coupling of bromo-arenes with nitrogen heterocycles without any外加 (external) HAT reagent. Instead, the system ingeniously utilizes chloride ions, likely derived from the nickel salt or added additives, to generate chlorine radicals via single electron transfer (SET) upon photoexcitation. These radicals selectively abstract the alpha-hydrogen to generate the crucial carbon-centered radical intermediate. This elegant mechanism allows for the direct coupling of C(sp3)-H bonds with aryl halides at room temperature, drastically simplifying the operational procedure. As illustrated by the core scaffold below, this method efficiently produces high-value intermediates with excellent purity.
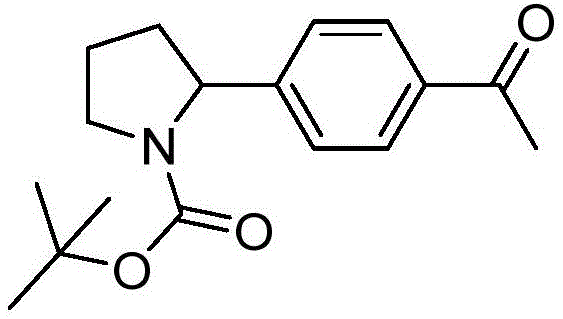
The structural versatility of this approach is evident in the successful synthesis of compounds like the one depicted above, where a pyrrolidine ring is directly coupled to a functionalized aromatic system. This capability to tolerate sensitive functional groups while maintaining high yields positions this technology as a superior alternative for the cost reduction in API manufacturing, particularly for late-stage functionalization where molecule stability is paramount.
Mechanistic Insights into Dual Photoredox and Nickel Catalysis
To fully appreciate the robustness of this synthetic route, one must delve into the intricate catalytic cycle that drives the transformation. The mechanism, detailed in the patent, relies on a carefully orchestrated interplay between two distinct catalytic cycles that converge to form the desired C-C bond. Upon irradiation with blue LEDs (450 nm), the iridium photocatalyst is excited to a high-energy state, denoted as Ir(III)*. This excited species acts as a potent oxidant, engaging in a Single Electron Transfer (SET) process with chloride ions present in the reaction medium. This critical step generates a highly reactive chlorine radical and reduces the photocatalyst to Ir(II). The chlorine radical then performs a selective Hydrogen Atom Transfer (HAT) on the N-alpha position of the protected pyrrolidine substrate, generating the key nucleophilic alpha-amino radical intermediate while releasing HCl, which is neutralized by the base.
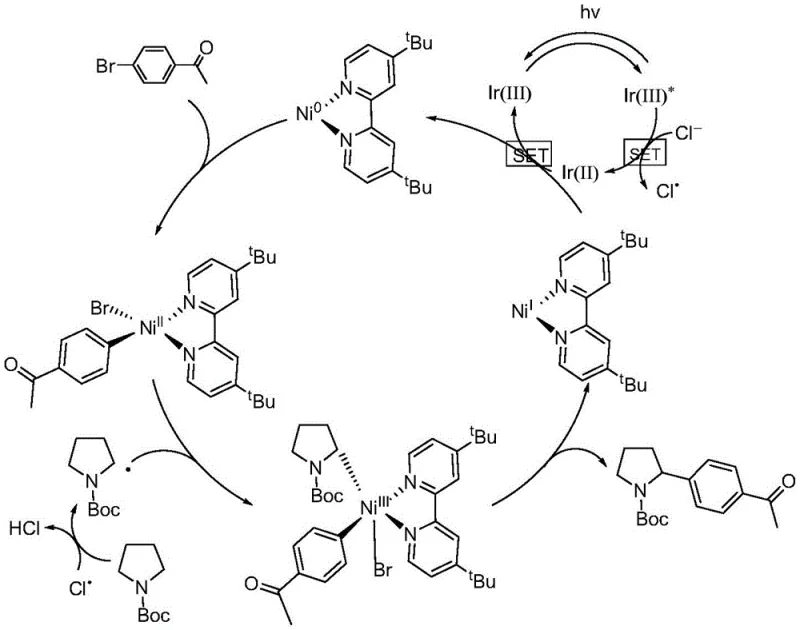
Simultaneously, the nickel catalytic cycle proceeds through oxidative addition of the aryl bromide to a zero-valent nickel species, which is regenerated via reduction by the reduced Ir(II) photocatalyst. The resulting aryl-nickel(II) complex then captures the alpha-amino radical generated in the photoredox cycle, forming a transient nickel(III) species. Finally, reductive elimination from this high-valent nickel center releases the desired N-alpha arylated product and regenerates the nickel(I) or nickel(0) species to close the cycle. This mechanistic understanding is vital for R&D teams aiming to optimize reaction parameters or adapt the protocol to new substrates, as it highlights the importance of maintaining the delicate balance between the two catalytic manifolds to prevent off-cycle decomposition or homocoupling side reactions.
How to Synthesize N-Alpha Aryl Compounds Efficiently
Implementing this photocatalytic methodology in a laboratory or pilot plant setting requires strict adherence to the optimized conditions outlined in the patent to ensure reproducibility and high yield. The process is designed to be operationally simple, avoiding the need for specialized high-pressure equipment or cryogenic conditions, which facilitates easier technology transfer from discovery to production scales. The standard protocol involves dissolving the bromo-arene and nitrogen heterocycle in a mixed solvent system of acetone and ethyl acetate, followed by the addition of the catalytic system comprising the iridium complex, nickel chloride, and dtbbpy ligand. The reaction is then subjected to visible light irradiation under an inert atmosphere for a defined period, typically ranging from 12 to 20 hours, allowing sufficient time for the catalytic turnover to reach completion. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining bromo-arene compounds, nitrogen-containing heterocycles, Ir-based photocatalyst, nickel chloride, dtbbpy ligand, and sodium carbonate base in an acetone and ethyl acetate solvent system.
- Maintain an inert atmosphere and irradiate the mixed solution with 450 nm LED light sources at room temperature for 12 to 20 hours to facilitate the dual catalytic cycle.
- Remove the solvent via rotary evaporation and purify the resulting crude N-alpha aryl compound using thin-layer chromatography with a petroleum ether and ethyl acetate eluent.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this photocatalytic N-alpha arylation technology offers compelling advantages that directly address the pain points of procurement managers and supply chain heads in the fine chemical industry. The elimination of external HAT reagents not only simplifies the bill of materials but also removes a potential source of supply chain volatility, as these specialized reagents can sometimes be costly or subject to long lead times. Furthermore, the use of earth-abundant nickel as the transition metal catalyst, supplemented by a catalytic amount of iridium, presents a significant cost reduction in pharmaceutical intermediate manufacturing compared to processes relying solely on precious metals like palladium. The ability to run reactions at room temperature also translates to substantial energy savings, as there is no need for extensive heating or cooling infrastructure, thereby lowering the overall utility costs associated with production.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, stemming primarily from the simplified reagent profile and the high atom economy of the transformation. By avoiding stoichiometric oxidants and directing groups, the process generates significantly less chemical waste, which reduces the costs associated with waste disposal and environmental compliance. Additionally, the high yields reported across a broad range of substrates mean that less starting material is required to produce a given amount of product, effectively lowering the cost of goods sold (COGS). The use of commercially available and relatively inexpensive solvents like acetone and ethyl acetate further contributes to the overall economic viability of the method, making it an attractive option for large-scale production runs where margin optimization is critical.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on commodity chemicals and robust reaction conditions that are less prone to failure due to minor fluctuations in temperature or pressure. The substrates used, such as bromo-acetophenones and Boc-protected amines, are widely available from multiple global suppliers, reducing the risk of single-source dependency. Moreover, the mild nature of the reaction enhances safety profiles, minimizing the risk of thermal runaways or hazardous exotherms that could disrupt production schedules. This stability ensures a more predictable and continuous supply of high-purity intermediates, which is essential for maintaining the integrity of downstream drug manufacturing timelines and meeting regulatory delivery commitments without unexpected delays.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been a challenge due to light penetration issues, but advancements in LED technology and flow chemistry reactors have made this increasingly feasible for industrial applications. The low energy input required for LED irradiation aligns perfectly with modern sustainability goals and green chemistry principles, helping companies meet stringent environmental regulations regarding carbon footprint and energy consumption. The absence of toxic heavy metal waste streams, thanks to the low loading of catalysts and the use of nickel, simplifies the purification process and reduces the environmental burden of the manufacturing site. This alignment with eco-friendly practices not only mitigates regulatory risks but also enhances the corporate social responsibility profile of the manufacturing partner, a factor of growing importance to end-client pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic arylation technology, based on the specific data and embodiments provided in the patent documentation. These insights are intended to clarify the operational feasibility and strategic value of adopting this method for the synthesis of complex nitrogenous scaffolds. Understanding these nuances is crucial for technical teams evaluating the fit of this chemistry within their existing process portfolios.
Q: What is the primary advantage of this photocatalytic method over traditional C-H functionalization?
A: The primary advantage is the elimination of external Hydrogen Atom Transfer (HAT) reagents. Traditional methods often require stoichiometric additives or harsh pre-activation steps, whereas this patented process utilizes chloride ions generated in situ to mediate hydrogen abstraction, significantly simplifying the workflow and reducing waste.
Q: Can this reaction be scaled for commercial production of pharmaceutical intermediates?
A: Yes, the reaction operates at room temperature using visible light LEDs and commercially available catalysts like nickel chloride and iridium complexes. The mild conditions and high atom economy make it highly suitable for commercial scale-up, offering a safer and more energy-efficient alternative to thermal cross-coupling reactions.
Q: What types of substrates are compatible with this N-alpha arylation protocol?
A: The method demonstrates broad substrate universality. It effectively couples various bromo-arenes, including those with electron-withdrawing or donating groups, with diverse nitrogen-containing heterocycles such as N-Boc-pyrrolidine, piperidines, and amides, yielding high-purity products suitable for drug discovery pipelines.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Alpha Aryl Compound Supplier
As the demand for efficient and sustainable synthetic routes continues to grow, partnering with a technically proficient CDMO becomes a strategic imperative for pharmaceutical innovators. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly transitioned into reliable supply chains. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of N-alpha aryl compounds meets the highest industry standards, minimizing the risk of downstream processing failures. We understand the critical nature of timeline adherence in drug development and are equipped to handle the complexities of photocatalytic processes with precision and care.
We invite you to engage with our technical procurement team to discuss how this advanced arylation technology can be integrated into your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the potential economic benefits tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive both scientific success and commercial efficiency in your pharmaceutical manufacturing endeavors.
