Advanced Two-Step Synthesis of Beta-Apo-8'-Carotenal for Commercial Food Applications
The global demand for high-quality natural and nature-identical colorants in the food industry continues to surge, driving the need for robust synthetic routes for key molecules like beta-apo-8'-carotenal. Patent CN112390737A introduces a significant technological breakthrough in the preparation of this vital oxygen-containing apo-carotenoid compound, which is extensively utilized in ice cream, edible oils, margarine, and various processed food matrices. Unlike previous methodologies that struggled with selectivity and safety, this invention proposes a streamlined two-step sequence starting from beta-apo-8'-carotenoic acid ethyl ester. The process involves a controlled reduction to an intermediate alcohol followed by a selective oxidation to the target aldehyde, offering a pathway that is both industrially viable and environmentally considerate. For procurement specialists and R&D leaders, this represents a shift towards more predictable supply chains and higher quality specifications for food-grade additives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-apo-8'-carotenal has been plagued by significant chemical and safety challenges that hinder efficient large-scale production. One prevalent traditional method involves the direct oxidation of beta-carotene using potassium permanganate, a strong oxidizing agent that lacks the necessary chemoselectivity for such a complex polyene structure. As illustrated in the reaction scheme below, the abundance of conjugated double bonds in the beta-carotene backbone makes it highly susceptible to non-specific attack, leading to extensive over-oxidation and a messy mixture of degradation products.

Furthermore, alternative synthetic routes have relied on condensation reactions involving beta-apo-12'-carotene dimethyl acetal and 1-methoxy-2-methyl-1,3-butadiene catalyzed by boron trifluoride diethyl etherate. While chemically feasible, this approach introduces severe safety hazards due to the generation of toxic and corrosive smog upon contact with moisture or steam. These operational risks necessitate expensive containment systems and specialized waste treatment protocols, creating substantial bottlenecks for manufacturers aiming to scale up production while maintaining strict environmental compliance standards.
The Novel Approach
In stark contrast to these hazardous and inefficient legacy processes, the methodology disclosed in the patent utilizes a gentle reduction-oxidation strategy that fundamentally alters the risk profile of the synthesis. By selecting beta-apo-8'-carotenoic acid ethyl ester as the starting material, the process bypasses the need to manipulate the fragile polyene chain of beta-carotene directly with harsh oxidants. The first stage employs mild reducing agents such as lithium aluminum hydride or sodium borohydride to convert the ester functionality into a primary alcohol without disturbing the sensitive conjugated system. This strategic choice of starting material and reagents ensures that the structural integrity of the carotenoid backbone is preserved throughout the transformation, significantly minimizing the formation of unwanted side products.
Mechanistic Insights into Reduction-Oxidation Cascade
The core of this innovative synthesis lies in the precise control of functional group interconversion under inert atmospheric conditions. In the initial reduction step, the ester group of the raw material undergoes nucleophilic attack by hydride ions provided by the reducing agent, typically dispersed in a solvent like tetrahydrofuran or diethyl ether. The reaction is carefully monitored via thin-layer chromatography to ensure complete conversion to the intermediate apo-alcohol before proceeding to hydrolysis. This step is critical because incomplete reduction would leave unreacted ester impurities that could complicate downstream purification, while the use of inert gases like nitrogen prevents oxidative degradation of the sensitive alcohol intermediate during the process.
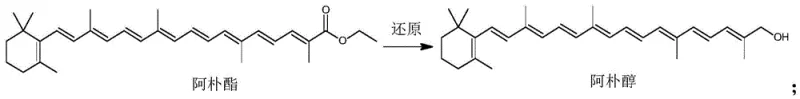
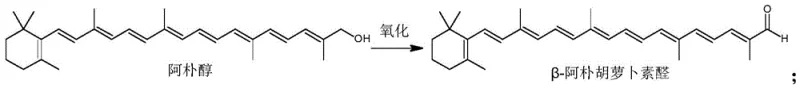
Following the isolation of the alcohol concentrate, the second stage involves a selective oxidation to regenerate the aldehyde functionality at the terminal position. The patent highlights the use of oxidants such as manganese dioxide or pyridinium chlorochromate, which are known for their ability to oxidize allylic or benzylic alcohols to aldehydes without over-oxidizing them to carboxylic acids. As shown in the subsequent reaction diagram, this transformation occurs under mild thermal conditions, typically between 20 and 30 degrees Celsius, which is crucial for preventing thermal isomerization or decomposition of the long conjugated chain. This mechanistic precision ensures a high-purity final product suitable for sensitive food applications.
How to Synthesize Beta-Apo-8'-Carotenal Efficiently
The execution of this synthesis requires strict adherence to anhydrous conditions and temperature controls to maximize yield and purity. The process begins with the preparation of the reducing agent suspension in a dry organic solvent, followed by the controlled addition of the ester substrate to manage exothermic heat release. Once the reduction is complete, a careful hydrolysis step is performed to quench the reaction and precipitate inorganic salts, which are then removed via filtration to yield a clean alcohol concentrate. The detailed standardized operating procedures for scaling this reaction from laboratory glassware to industrial reactors are outlined in the technical guide below.
- Reduce beta-apo-8'-carotenoic acid ethyl ester to apo alcohol using lithium aluminum hydride in tetrahydrofuran under inert atmosphere.
- Hydrolyze the reaction intermediate with water to isolate the apo alcohol concentrate.
- Oxidize the apo alcohol concentrate to beta-apo-8'-carotenal using manganese dioxide in dichloromethane.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain directors and procurement managers, the adoption of this patented methodology offers distinct logistical and economic benefits over traditional manufacturing routes. The elimination of hazardous catalysts like boron trifluoride removes the need for specialized corrosion-resistant equipment and complex scrubbing systems, thereby drastically simplifying the capital expenditure required for plant setup. Furthermore, the use of readily available and inexpensive reagents such as manganese dioxide and common organic solvents ensures a stable supply of raw materials that is less susceptible to market volatility compared to exotic catalysts. This stability translates directly into more predictable production schedules and reduced risk of supply interruptions for downstream food manufacturers.
- Cost Reduction in Manufacturing: The streamlined two-step process significantly lowers operational costs by removing the need for expensive transition metal catalysts and complex purification sequences associated with over-oxidized byproducts. By avoiding the generation of toxic smog and hazardous waste streams, the facility saves substantially on waste disposal fees and environmental compliance monitoring. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to a lower overall cost of goods sold for the final active ingredient.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this process is straightforward, as beta-apo-8'-carotenoic acid ethyl ester and standard reducing agents are commodity chemicals with established global supply networks. This accessibility mitigates the risk of single-source dependency often associated with proprietary catalysts, ensuring that production can continue uninterrupted even during regional supply shocks. The robustness of the chemistry also means that batch-to-batch variability is minimized, allowing for consistent inventory planning and reliable delivery commitments to major food and beverage clients.
- Scalability and Environmental Compliance: The absence of toxic intermediates and the use of green chemistry principles make this process highly scalable without triggering stringent regulatory hurdles. Facilities can expand capacity with confidence, knowing that the effluent profile is manageable and does not require advanced treatment technologies for heavy metals or corrosive gases. This environmental compatibility not only future-proofs the manufacturing site against tightening regulations but also aligns with the sustainability goals of modern multinational corporations seeking eco-friendly ingredient suppliers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on safety, purity, and operational parameters. Understanding these details is essential for technical teams evaluating the feasibility of integrating this technology into existing production lines.
Q: Why is the new synthesis method safer than traditional potassium permanganate oxidation?
A: Traditional methods using potassium permanganate often lead to over-oxidation and poor selectivity due to the multiple double bonds in the carotenoid chain. The new method utilizes controlled reduction followed by selective oxidation, avoiding harsh conditions and toxic byproducts.
Q: What are the primary advantages regarding industrial scalability?
A: The process operates under mild temperatures ranging from 0 to 30 degrees Celsius and uses common solvents like tetrahydrofuran and dichloromethane. This eliminates the need for extreme cryogenic conditions or hazardous catalysts like boron trifluoride, simplifying equipment requirements.
Q: How does this method impact the purity profile of the final aldehyde?
A: By avoiding non-selective oxidants that attack the polyene chain, the method preserves the conjugated system integrity. This results in a cleaner impurity profile with fewer over-oxidized degradation products compared to direct oxidation routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Apo-8'-Carotenal Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of delivering high-purity ingredients that meet the rigorous standards of the global food and nutrition industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of beta-apo-8'-carotenal delivers the consistent color strength and stability required for premium food applications.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific product needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and product quality.
