Advanced Electrochemical Production of 3,4-Dibromomaleimide Derivatives for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking greener, more efficient pathways to synthesize critical intermediates, and the electrochemical synthesis method detailed in patent CN112795943A represents a significant technological leap forward for producing 3,4-dibromomaleimide derivatives. This innovative approach utilizes electricity as a clean reagent to drive the bromination of maleimide substrates, effectively replacing hazardous chemical oxidants and metal catalysts with a sustainable electron transfer process. By leveraging anodic oxidation, this method generates reactive bromine species in situ from inexpensive sodium bromide, ensuring a reaction environment that is not only economically superior but also aligns perfectly with modern green chemistry principles. For R&D directors and procurement managers alike, this technology offers a compelling solution to the longstanding challenges of safety, cost, and environmental compliance associated with traditional halogenation reactions, positioning it as a cornerstone for future reliable pharmaceutical intermediate supplier strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,4-dibromomaleimide has relied on chemically aggressive and operationally complex routes that pose significant risks to both personnel and the environment. One prominent conventional method involves the use of N-bromosuccinimide (NBS) followed by oxidative rearrangement with concentrated nitric acid at low temperatures, a process that generates toxic nitrogen dioxide gas and requires stringent safety controls due to the corrosive and unstable nature of the reagents. Another established route utilizes elemental bromine in a sealed tube with aluminum chloride catalysts at elevated temperatures, necessitating specialized high-pressure equipment and creating severe hazards related to bromine volatility and toxicity. These traditional pathways are characterized by multi-step procedures, poor atom economy, and the generation of substantial hazardous waste, which collectively drive up production costs and complicate regulatory compliance for cost reduction in API manufacturing.
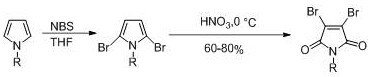
The Novel Approach
In stark contrast to these legacy methods, the electrochemical synthesis described in the patent offers a streamlined, one-pot solution that operates under remarkably mild conditions, typically at room temperature and atmospheric pressure. This novel approach dissolves the maleimide substrate and a cheap bromide salt, such as sodium bromide, in a suitable organic solvent within an anode chamber, where electricity drives the formation of the active brominating agent directly at the electrode surface. This eliminates the need for storing and handling dangerous elemental bromine or strong oxidizing acids, drastically simplifying the operational workflow and reducing the capital investment required for safety infrastructure. The ability to achieve high yields in a single reaction vessel without the need for transition metal catalysts represents a paradigm shift, enabling manufacturers to produce high-purity maleimide derivatives with a significantly reduced environmental footprint and enhanced process safety.
Mechanistic Insights into Electrochemical Anodic Bromination
The core of this technology lies in the precise control of electron transfer at the electrode interface, where bromide anions are oxidized to generate molecular bromine or equivalent electrophilic bromine species in situ. This electrochemically generated bromine immediately reacts with the carbon-carbon double bond of the maleimide ring, forming a positively charged intermediate that subsequently undergoes deprotonation to yield the mono-brominated species, which is further brominated to the final 3,4-dibromo product. The use of inert electrodes, such as platinum or graphite, ensures that the electrode material itself does not participate in the redox chemistry, thereby preventing metal contamination of the product and maintaining the integrity of the catalytic cycle. This mechanism allows for fine-tuning of the reaction rate and selectivity through the adjustment of current density, providing a level of control that is difficult to achieve with bulk chemical reagents.
Furthermore, the electrochemical environment inherently supports superior impurity control by minimizing side reactions that are common in harsh chemical brominations, such as over-oxidation or ring degradation. The mild potential applied across the cell is sufficient to drive the desired bromination but can be regulated to avoid damaging the sensitive maleimide core, resulting in a cleaner crude reaction mixture. This reduction in byproduct formation simplifies downstream purification processes, such as extraction and crystallization, leading to higher overall recovery rates and consistent product quality. For quality assurance teams, this mechanistic advantage translates to a more robust and reproducible manufacturing process that consistently meets the stringent purity specifications required for pharmaceutical and electronic material applications.
How to Synthesize 3,4-Dibromomaleimide Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this electrochemical technology, emphasizing the importance of cell configuration and electrolyte composition for optimal results. The process begins with the preparation of an H-type electrolytic cell separated by a cation exchange membrane, which prevents the migration of cathodic reduction products into the anode chamber where the target reaction occurs. Operators dissolve the specific N-substituted maleimide and an excess of sodium bromide in a polar aprotic solvent like DMF, optionally buffered to maintain a stable pH, before applying a constant current to initiate the reaction. Detailed standard operating procedures for scaling this reaction from laboratory to pilot plant are critical for success, and the specific parameters for electrode spacing, stirring rates, and workup procedures are essential for maximizing efficiency.
- Prepare an H-type electrolytic cell with inert electrodes (platinum or graphite) and add the maleimide substrate and sodium bromide dissolved in an organic solvent like DMF to the anode chamber.
- Conduct constant current electrolysis at room temperature (20-50°C) with a current density of 5-30 mA/cm² until TLC monitoring indicates complete conversion.
- Extract the reaction mixture from the anode chamber with ethyl acetate, dry the organic layer, and concentrate to isolate the high-purity 3,4-dibromomaleimide product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this electrochemical methodology offers profound benefits for procurement and supply chain management, primarily driven by the substitution of expensive and hazardous reagents with commodity chemicals. The elimination of NBS, nitric acid, and elemental bromine removes significant cost drivers associated with the purchase, storage, and disposal of dangerous materials, while the absence of metal catalysts negates the need for costly removal steps and heavy metal testing. This shift not only lowers the direct material costs but also reduces the regulatory burden and insurance premiums associated with handling hazardous substances, contributing to a leaner and more resilient supply chain. For organizations focused on commercial scale-up of complex pharmaceutical intermediates, this technology provides a pathway to more predictable pricing and reduced vulnerability to supply disruptions of specialty reagents.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally improved by the use of sodium bromide, a widely available and inexpensive salt, as the sole source of bromine, replacing costly oxidants and catalysts. By removing the requirement for specialized high-pressure reactors or cryogenic cooling systems needed for traditional methods, the capital expenditure for production facilities is significantly lowered, allowing for better allocation of resources. Additionally, the simplified one-pot nature of the reaction reduces labor hours and utility consumption, as there are fewer unit operations and less energy required for heating or cooling, leading to substantial operational savings over the lifecycle of the product.
- Enhanced Supply Chain Reliability: Relying on stable, non-volatile solid salts like sodium bromide instead of volatile liquids like bromine or unstable acids enhances the safety and reliability of the raw material supply chain. The reduced hazard profile simplifies logistics and warehousing requirements, allowing for larger inventory buffers without triggering stringent hazardous material storage regulations. This stability ensures continuous production capability even during market fluctuations for specialty chemicals, securing the supply of critical intermediates for downstream drug synthesis and minimizing the risk of production stoppages due to reagent shortages or transport restrictions.
- Scalability and Environmental Compliance: The mild reaction conditions and aqueous-compatible electrolyte systems make this process highly scalable, as heat management and pressure containment are far less challenging than in exothermic chemical brominations. The green nature of the process, characterized by the absence of heavy metal waste and reduced acidic effluent, aligns with increasingly strict environmental regulations, facilitating easier permitting and community acceptance for manufacturing sites. This environmental compatibility not only future-proofs the production asset against tightening regulations but also enhances the brand value of the final product by appealing to sustainability-conscious partners and customers in the global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of 3,4-dibromomaleimide, drawing directly from the experimental data and beneficial effects reported in the patent literature. These insights are designed to clarify the operational feasibility and strategic advantages of adopting this technology for industrial applications, helping stakeholders make informed decisions about process integration. Understanding these nuances is key to leveraging the full potential of electrochemical manufacturing for high-value organic intermediates.
Q: What are the primary advantages of the electrochemical method over traditional bromination?
A: The electrochemical method eliminates the need for hazardous external oxidants like nitric acid or elemental bromine, operates under mild room temperature and pressure conditions, and utilizes cheap, safe sodium bromide, significantly reducing environmental impact and operational risks.
Q: How does this process ensure high purity for pharmaceutical applications?
A: By avoiding metal catalysts and harsh acidic conditions that often lead to complex byproduct profiles, the electrochemical route offers a cleaner reaction profile. The use of inert electrodes and controlled current density minimizes over-oxidation, facilitating easier purification and higher final purity.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process is highly scalable due to its one-pot nature and mild operating conditions. The simplicity of the equipment (standard electrolytic cells) and the use of inexpensive, non-hazardous raw materials like sodium bromide make it ideal for large-scale commercial manufacturing without significant safety infrastructure investments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4-Dibromomaleimide Supplier
As the demand for high-quality maleimide derivatives continues to grow in the pharmaceutical and organic electronics sectors, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthesis technologies and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, utilizing advanced electrochemical reactors and rigorous QC labs to guarantee batch-to-batch consistency. We are committed to delivering stringent purity specifications that meet the exacting standards of global drug developers, leveraging our deep technical expertise to optimize yield and minimize impurities through precise process control.
We invite you to engage with our technical procurement team to discuss how this innovative electrochemical route can be tailored to your specific project needs, offering a Customized Cost-Saving Analysis that highlights the economic benefits of switching from traditional chemical methods. By collaborating with us, you gain access to specific COA data and route feasibility assessments that validate the commercial viability of this green synthesis approach for your supply chain. Let us help you secure a sustainable and cost-effective source of 3,4-dibromomaleimide, driving your projects forward with confidence and efficiency.
