Advanced Electrochemical Synthesis of 2-Mercaptobenzothiazole Metal Compounds for Industrial Scale-Up
The chemical industry is currently witnessing a paradigm shift towards greener, more atom-economical manufacturing processes, a transition vividly exemplified by the technology disclosed in patent CN103510104A. This pivotal intellectual property introduces a novel electrochemical methodology for the synthesis of 2-mercaptobenzothiazole metal compounds, a class of substances critical to both the agrochemical sector as fungicides and the polymer industry as rubber vulcanization accelerators. Unlike traditional stoichiometric approaches that rely heavily on metal salts and generate substantial inorganic waste, this invention leverages the power of direct current electrolysis to generate metal cations in situ from a sacrificial anode. This fundamental change in reaction engineering not only streamlines the synthetic pathway but also addresses the growing regulatory pressure regarding industrial effluent management. For R&D directors and process chemists seeking to modernize their production lines, this electrochemical route represents a significant technological leap, offering a robust platform for generating high-purity intermediates with minimal environmental footprint.
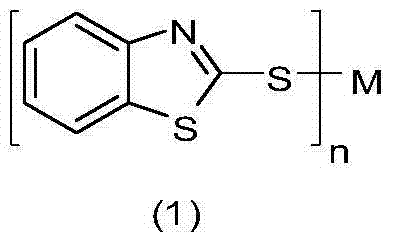
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of 2-mercaptobenzothiazole metal complexes has been dominated by salt metathesis reactions, where a soluble metal salt reacts with 2-mercaptobenzothiazole under homogeneous conditions. While chemically straightforward, this conventional approach suffers from severe inefficiencies that hinder large-scale commercial viability. A primary bottleneck is the poor solubility of 2-mercaptobenzothiazole in many common organic solvents, necessitating the use of excessive volumes of solvent to maintain a homogeneous reaction mixture, which subsequently increases downstream solvent recovery costs. Furthermore, when aqueous conditions are employed to mitigate solvent usage, the process invariably requires the addition of stoichiometric amounts of strong alkali to form the reactive thiolate salt prior to metal coordination. This requirement generates copious amounts of saline wastewater containing dissolved inorganic salts, creating a significant burden on waste treatment facilities and violating the principles of green chemistry. The accumulation of these inorganic byproducts often complicates the isolation of the final product, leading to lower overall yields and purity profiles that may require additional recrystallization steps.
The Novel Approach
In stark contrast to these legacy methods, the electrochemical synthesis described in the patent data offers a transformative solution by fundamentally altering the source of the metal cation. By utilizing the metal itself as a sacrificial anode, the process bypasses the need for expensive and waste-generating metal salt precursors entirely. The electrolytic cell facilitates the direct oxidation of the metal anode into cations, which immediately react with the thiolate anions generated at the cathode surface. This "on-demand" generation of reactive species ensures that the reaction proceeds with high atom economy, as the only byproduct is hydrogen gas, which can be safely vented or captured. Moreover, the patent highlights the remarkable versatility of this system, demonstrating successful synthesis with a wide array of metals including Zinc, Copper, Nickel, and Iron, simply by swapping the anode material. This flexibility allows manufacturers to produce a diverse portfolio of metal complexes using a single, standardized reactor setup, thereby maximizing capital efficiency and reducing the complexity of the supply chain for raw materials.
Mechanistic Insights into Sacrificial Anode Electrolysis
The core of this innovative process lies in the elegant simplicity of its electrochemical mechanism, which operates through a dual-electrode system driven by direct current. At the anode, the selected metal M (such as Zn or Cu) undergoes oxidative dissolution, releasing metal cations (M^n+) directly into the electrolyte solution without the introduction of counter-ions like chlorides or sulfates that would otherwise contaminate the product stream. Simultaneously, at the inert cathode, the 2-mercaptobenzothiazole molecules undergo reductive deprotonation to form the corresponding mercapto anions, accompanied by the evolution of hydrogen gas. This spatial separation of oxidation and reduction half-reactions within the same vessel creates a highly controlled environment where the metal cations and organic anions meet and precipitate as the insoluble metal complex. The use of specific conductive agents, such as quaternary ammonium salts or simple inorganic chlorides, ensures sufficient conductivity in the organic or aqueous medium to sustain the current flow without participating in the primary redox chemistry, thus maintaining the high purity of the final precipitate.
From an impurity control perspective, this mechanism offers distinct advantages over thermal or chemical precipitation methods. Because the metal ions are generated electrochemically rather than introduced as bulk salts, the risk of incorporating anionic impurities from the metal source is virtually eliminated. The precipitation of the product occurs directly from the solution as it forms, which often leads to the formation of well-defined crystalline structures, as evidenced by the sharp X-ray powder diffraction peaks observed in the patent examples. This inherent crystallinity simplifies the filtration and washing steps, as the solid product is less likely to trap mother liquor or soluble impurities compared to amorphous precipitates formed in rapid chemical mixing. Furthermore, the ability to monitor the reaction progress via Thin Layer Chromatography (TLC) allows for precise endpoint determination, ensuring that the reaction is stopped exactly when the starting material is consumed, thereby preventing over-electrolysis or decomposition of the sensitive thiol functionality.
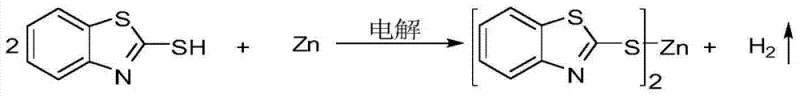
How to Synthesize 2-Mercaptobenzothiazole Metal Compounds Efficiently
Implementing this electrochemical protocol requires careful attention to the selection of solvents and conductive agents to optimize conductivity and solubility. The patent data suggests that polar aprotic solvents like DMF and DMSO, as well as lower alcohols like methanol and isopropanol, provide excellent media for dissolving the organic substrate while supporting ion transport. The concentration of the conductive agent is a critical parameter, typically maintained between 0.01 and 0.1 mol/L to balance conductivity with ease of product isolation. Once the electrolyte is prepared and the sacrificial anode installed, the application of a moderate current density (typically 10-100 mA) initiates the reaction, which proceeds smoothly at temperatures ranging from room temperature to 65°C. The detailed标准化 synthesis steps see the guide below.
- Prepare the electrolytic cell by dissolving 2-mercaptobenzothiazole and a conductive agent (such as tetrabutylammonium bromide) in a suitable solvent like methanol or acetonitrile.
- Insert the target metal (e.g., Zinc, Copper) as the sacrificial anode and an inert material like graphite as the cathode, then apply a direct current of 10-100mA.
- Monitor the reaction via TLC until the starting material disappears, then filter the resulting suspension, wash the precipitate, and dry to obtain the pure metal complex.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electrochemical technology translates into tangible strategic benefits that extend far beyond simple yield improvements. The elimination of metal salt precursors removes a significant cost center from the Bill of Materials, as high-purity metal foils or rods are often more cost-effective and stable than their corresponding salt counterparts. Additionally, the capability to recycle the filtrate back into the electrolytic cell dramatically reduces the consumption of organic solvents, which are often subject to volatile pricing and strict environmental regulations regarding VOC emissions. This closed-loop solvent system not only lowers raw material costs but also minimizes the volume of hazardous waste requiring disposal, leading to substantial savings in waste management fees and reducing the facility's overall environmental liability.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the removal of stoichiometric base requirements and the associated neutralization steps. In traditional synthesis, the cost of acids and bases for pH adjustment and the subsequent treatment of saline wastewater can be prohibitive at scale. By generating the reactive species electrochemically, the process avoids these auxiliary chemical costs entirely. Furthermore, the high purity of the crude product reduces the need for energy-intensive recrystallization or chromatographic purification, allowing for a more streamlined downstream processing workflow that lowers utility consumption and labor hours per kilogram of product.
- Enhanced Supply Chain Reliability: Relying on elemental metals as the primary source of the metal component simplifies the supply chain significantly. Elemental metals like zinc and copper are commodity chemicals with stable, global supply networks, unlike specialized metal salts which may have fewer suppliers and longer lead times. This commoditization of raw materials reduces the risk of supply disruptions and provides procurement teams with greater leverage in negotiations. The robustness of the electrochemical cell design also means that the process is less sensitive to variations in raw material quality, ensuring consistent production output even when sourcing from different vendors.
- Scalability and Environmental Compliance: The scalability of electrochemical processes is well-documented in the fine chemical industry, and this specific method is no exception. The modular nature of electrolytic cells allows for capacity expansion by simply adding more cells in parallel, avoiding the engineering challenges associated with scaling up large batch reactors. From a compliance standpoint, the near-zero discharge of saline wastewater aligns perfectly with increasingly stringent environmental regulations in major manufacturing hubs. This "green" credential not only future-proofs the manufacturing site against regulatory changes but also enhances the brand value of the final product for customers who prioritize sustainable sourcing in their own supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this electrochemical synthesis route. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the technology's fit within your existing manufacturing infrastructure. Understanding these nuances is essential for making informed decisions about process adoption and capital investment.
Q: What are the primary advantages of the electrochemical method over traditional salt metathesis?
A: The electrochemical method eliminates the need for pre-formed metal salts and stoichiometric bases, thereby avoiding the generation of large quantities of saline wastewater and significantly simplifying the post-reaction purification process.
Q: Which metals can be utilized as sacrificial anodes in this synthesis protocol?
A: The patent specifies a versatile range of metals including Zinc, Copper, Nickel, Manganese, Iron, Tin, and Aluminum, allowing for the production of various metal complexes suitable for different industrial applications.
Q: Is the electrolyte solution reusable in this continuous process?
A: Yes, the filtrate obtained after separating the solid product can be returned directly to the electrolytic cell for recycling as the electrolyte, which drastically reduces solvent consumption and operational costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Mercaptobenzothiazole Metal Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in producing high-value fine chemical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN103510104A are fully realized in practical, industrial settings. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and stringent purity specifications are maintained through our rigorous QC labs, guaranteeing that every batch of 2-mercaptobenzothiazole metal compound meets the exacting standards required for agrochemical and polymer applications.
We invite you to collaborate with us to explore how this technology can optimize your specific supply chain. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how switching to this electrochemical route can improve your margins. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of these critical intermediates.
