Scalable Synthesis of 4β,5β-Epoxy-Δ6-Steren-3β-Alcohol for Advanced Steroidal Drug Manufacturing
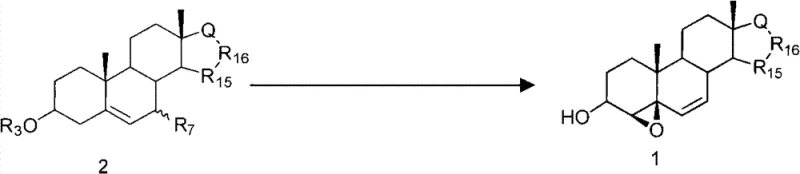
The pharmaceutical industry continuously seeks robust and scalable pathways for synthesizing complex steroidal intermediates, particularly those serving as precursors for high-value hormones like Drospirenone. Patent CN102002091B discloses a highly efficient preparation method for 4β,5β-epoxy-Δ6-steren-3β-alcohol, a critical building block in modern endocrinology drug development. This technology addresses significant bottlenecks in traditional synthesis by employing a streamlined three-step sequence: catalytic elimination, protective group removal, and stereoselective epoxidation. Unlike legacy processes that struggle with scarce starting materials, this innovation leverages cheap and readily available 3β-R3O-7-R7-Δ5-sterene compounds. The result is a process characterized by high立体 selectivity, operational simplicity, and yields that are conducive to large-scale industrial production, making it a cornerstone technology for reliable pharmaceutical intermediate suppliers aiming to optimize their API manufacturing pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4β,5β-epoxy-Δ6-steren-3-ol compounds has been fraught with supply chain vulnerabilities and chemical inefficiencies. Prior art, such as the method reported in CN101735300A, typically relies on steroid-4,6-dien-3-one as the primary raw material. While chemically feasible, this starting material is notoriously difficult to obtain on a large commercial scale, creating a bottleneck for procurement managers who require consistent volume availability. Furthermore, conventional routes often involve multi-step sequences with harsh reaction conditions that can compromise the stereochemical integrity of the molecule. The reliance on expensive or unstable precursors not only inflates the cost of goods sold but also introduces variability in the impurity profile, necessitating rigorous and costly downstream purification processes that delay time-to-market for final drug products.
The Novel Approach
The methodology outlined in CN102002091B represents a paradigm shift by utilizing 3β-R3O-7-R7-Δ5-sterene compounds as the foundational feedstock. These starting materials are significantly more accessible and cost-effective than the diene-ketones used in older protocols. The novel approach simplifies the synthetic trajectory into three distinct, high-yielding transformations. By focusing on a catalytic elimination strategy followed by mild deprotection and controlled epoxidation, the process minimizes the formation of unwanted byproducts. This reduction in chemical complexity translates directly to enhanced process safety and reduced waste generation. For supply chain heads, this means a more resilient production schedule with fewer dependencies on exotic reagents, ensuring a steady flow of high-purity intermediates essential for the continuous manufacturing of life-saving hormonal therapies.
Mechanistic Insights into Catalytic Elimination and Epoxidation
The core of this technological advancement lies in the precise control of stereochemistry during the construction of the Δ4,6-diene system and the subsequent epoxide ring. The first step involves a catalytic elimination reaction where the 7-position leaving group (R7) is removed to establish the conjugated diene system. This transformation is facilitated by Lewis acidic catalysts such as magnesium bromide or tribromopyridinium, often in the presence of absorbents like anhydrous magnesium sulfate to drive the equilibrium forward. The reaction proceeds under relatively mild thermal conditions, typically between 10°C and 125°C, preserving the sensitive functional groups elsewhere on the steroid skeleton. This careful modulation of reaction parameters ensures that the resulting 3β-R3O-Δ4,6-steradiene is formed with high regioselectivity, setting the stage for the subsequent functionalization.

Following the establishment of the diene framework, the process moves to deprotection and epoxidation. The removal of the 3β-protecting group (R3) is achieved using standard basic or acidic hydrolysis conditions, depending on the nature of the protecting group, yielding the free 3β-hydroxyl species. The final and most critical step is the epoxidation of the Δ4,6-diene system. Using oxidants like m-chloroperoxybenzoic acid (m-CPBA) or peracetic acid, the reaction selectively targets the C4-C5 double bond. The presence of the 3β-hydroxyl group plays a pivotal directing role, guiding the oxidant to approach from the alpha face to generate the desired 4β,5β-epoxy configuration. This intrinsic stereocontrol eliminates the need for chiral auxiliaries or complex resolution steps, significantly streamlining the production of this high-value pharmaceutical intermediate.

How to Synthesize 4β,5β-Epoxy-Δ6-Steren-3β-Alcohol Efficiently
Implementing this synthesis route requires strict adherence to the optimized reaction conditions detailed in the patent to ensure maximum yield and purity. The process begins with the preparation of the reaction vessel under anhydrous conditions, followed by the sequential addition of the steroidal substrate and the specific catalyst system. Temperature control is paramount during the elimination phase to prevent side reactions, while the epoxidation step requires careful monitoring of oxidant stoichiometry to avoid over-oxidation. The workup procedures, involving aqueous quenching and recrystallization, are designed to remove metal salts and organic impurities effectively. For a comprehensive understanding of the specific reagent quantities, solvent choices, and isolation techniques required for GMP-compliant production, please refer to the standardized protocol below.
- Perform catalytic elimination on 3β-R3O-7-R7-Δ5-sterene using magnesium bromide or tribromopyridinium to form the Δ4,6-diene system.
- Execute a deprotection reaction under mild alkaline conditions to remove the 3β-protecting group, yielding the free 3β-hydroxyl diene.
- Conduct stereoselective epoxidation using m-CPBA or peracetic acid to install the critical 4β,5β-epoxy functionality.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis method offers profound advantages for organizations focused on cost reduction in pharmaceutical intermediate manufacturing. The primary driver of value is the substitution of scarce, expensive starting materials with abundant, commodity-grade steroidal precursors. This shift fundamentally alters the cost structure of the supply chain, reducing the volatility associated with raw material pricing. Additionally, the reduction in the total number of synthetic steps decreases the consumption of solvents, reagents, and energy, leading to substantial operational expenditure savings. For procurement managers, this translates into a more competitive pricing model for the final API, allowing for better margin management in an increasingly price-sensitive global market.
- Cost Reduction in Manufacturing: The elimination of complex, multi-step sequences found in legacy methods significantly lowers the overall manufacturing cost. By avoiding the use of hard-to-source starting materials like steroid-4,6-dien-3-one, companies can bypass premium pricing tiers associated with niche chemicals. Furthermore, the high stereoselectivity of the epoxidation step reduces the need for expensive chromatographic separations or recrystallizations to remove diastereomers, thereby cutting down on both material loss and processing time. This efficiency gain allows for a leaner production model that maximizes output per batch while minimizing waste disposal costs.
- Enhanced Supply Chain Reliability: Reliance on readily available raw materials ensures a stable and continuous supply chain, mitigating the risk of production stoppages due to vendor shortages. The robustness of the catalytic elimination and epoxidation steps means that the process is less susceptible to minor fluctuations in reaction conditions, resulting in consistent batch-to-batch quality. This reliability is crucial for supply chain heads who must guarantee uninterrupted delivery to downstream API manufacturers. The simplified logistics of sourcing common reagents further enhance the agility of the supply network, enabling faster response times to market demand surges.
- Scalability and Environmental Compliance: The operational simplicity of this method makes it highly amenable to scale-up from pilot plant to commercial tonnage production. The use of standard solvents like dichloromethane, pyridine, and methanol facilitates easy integration into existing manufacturing infrastructure without the need for specialized equipment. Moreover, the high atom economy and reduced step count contribute to a smaller environmental footprint, aligning with modern green chemistry principles. This compliance with environmental standards reduces regulatory hurdles and potential liabilities, ensuring long-term sustainability for the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the feasibility and advantages of the process. Understanding these details is essential for technical teams evaluating the integration of this route into their existing production portfolios.
Q: What are the key advantages of this synthesis route over conventional methods?
A: This method utilizes cheap and readily available raw materials compared to prior art which relies on scarce steroid-4,6-dien-3-one. It features fewer reaction steps, simpler operation, and high stereoselectivity suitable for industrial scale-up.
Q: Which catalysts are preferred for the elimination step?
A: The patent specifies magnesium bromide, tribromopyridinium, lithium bromide, or montmorillonite K-10 as effective catalysts, often used in conjunction with absorbents like anhydrous magnesium sulfate.
Q: Is this intermediate suitable for Drospirenone synthesis?
A: Yes, the 4β,5β-epoxy-Δ6-sterene structural unit is a versatile precursor for progestins like Drospirenone, allowing for positioning-directed Simmons-Smith addition to achieve the 6β,7β-methylene structure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4β,5β-Epoxy-Δ6-Steren-3β-Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality steroidal intermediates in the development of next-generation hormonal therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 4β,5β-epoxy-Δ6-steren-3β-alcohol supports the efficacy and safety of your final drug product. Our facility is equipped to handle the specific catalytic and oxidative conditions required by this patent, maintaining full compliance with international regulatory standards.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can drive efficiency and reduce costs in your API production workflow. Let us be your partner in transforming complex chemical challenges into commercial successes.
