Advanced Enzymatic Synthesis of Lopinavir Intermediates for Commercial Scale-Up
Introduction to Patent CN113512569B: A Paradigm Shift in Antiretroviral Synthesis
The global demand for effective antiretroviral therapies continues to drive innovation in the synthesis of key pharmaceutical building blocks. Patent CN113512569B introduces a groundbreaking enzymatic synthesis method for Lopinavir intermediates, addressing critical challenges in stereocontrol and environmental sustainability. Lopinavir, a potent HIV protease inhibitor, requires the precise construction of multiple chiral centers, traditionally a bottleneck in manufacturing. This patent outlines a sophisticated route that replaces hazardous chemical reagents with highly selective biocatalysts, specifically targeting the reduction of a ketone precursor to a chiral alcohol with exceptional enantiomeric excess. By integrating TEMPO-mediated oxidation with a robust enzyme-coupled cofactor regeneration system, this technology offers a viable pathway for producing high-purity pharmaceutical intermediates. For R&D directors and supply chain leaders, understanding this shift from classical organic synthesis to chemo-enzymatic cascades is essential for maintaining competitiveness in the generic and branded API markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Lopinavir intermediates has relied heavily on the chiral pool strategy, utilizing expensive natural amino acids like L-phenylalanine and L-valine as starting materials. While effective, this approach often necessitates complex protection and deprotection sequences, leading to significant atom economy losses. Furthermore, traditional chemical methods for establishing the critical hydroxyl stereocenter frequently involve non-stereoselective reductions followed by tedious chiral resolution steps. These resolution processes inherently discard at least 50% of the material as the unwanted enantiomer, drastically inflating production costs and generating substantial chemical waste. The accumulation of impurities from multiple synthetic steps also complicates quality control, requiring rigorous chromatographic separations that are difficult to scale industrially. Consequently, manufacturers face persistent pressure to find more direct and waste-minimizing routes to meet the stringent purity specifications required for HIV medications.
The Novel Approach
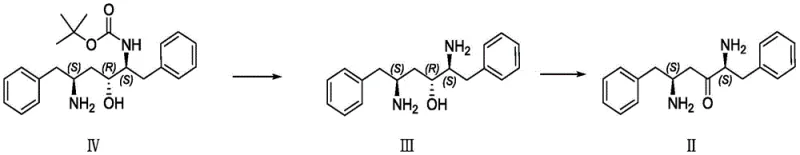
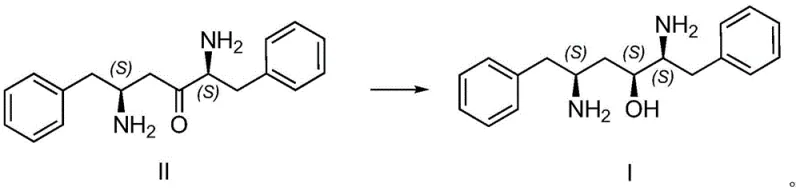
The methodology described in patent CN113512569B presents a streamlined alternative that circumvents these inefficiencies through a clever combination of chemical oxidation and biocatalysis. The process begins with the deprotection of a Boc-protected amine precursor (Compound IV) followed by a highly selective Anelli oxidation to generate the ketone substrate (Compound II). Unlike traditional oxidants that may over-oxidize or damage sensitive functional groups, this TEMPO-mediated system operates under mild conditions to deliver the ketone in high yield. The true innovation lies in the subsequent step, where a specialized ketone carbonyl reductase is employed to asymmetrically reduce the ketone to the desired alcohol (Compound I). This enzymatic step eliminates the need for chiral resolution entirely, as the enzyme inherently selects for the correct stereochemistry. By merging these distinct chemical and biological transformations, the novel approach achieves a level of precision and efficiency that conventional purely chemical routes struggle to match.
Mechanistic Insights into Ketone Carbonyl Reductase Catalysis
The core of this synthesis lies in the sophisticated mechanism of the biocatalytic reduction step, which utilizes a coupled enzyme system to drive the reaction to completion. The primary catalyst, a ketone carbonyl reductase (KRED), facilitates the transfer of a hydride ion from the reduced cofactor NADPH to the prochiral ketone carbonyl of Compound II. This transfer occurs with strict stereochemical control, ensuring the formation of the (S)-configured alcohol center essential for Lopinavir's biological activity. To make this process economically viable on an industrial scale, the expensive NADPH cofactor must be continuously regenerated. This is achieved through the addition of a second enzyme, isopropanol dehydrogenase (IPDH), which oxidizes isopropanol to acetone while reducing NADP+ back to NADPH. This cofactor recycling loop allows for the use of catalytic amounts of the expensive nicotinamide cofactor, significantly lowering the cost of goods. The reaction is conducted in a buffered aqueous system with isopropanol serving as both the hydrogen donor and a co-solvent to enhance substrate solubility, creating an optimal environment for enzyme stability and activity.
From an impurity control perspective, the specificity of the enzyme acts as a powerful filter against structural analogs and side products. In chemical reductions using borohydrides or aluminum hydrides, there is often a risk of reducing other functional groups or forming racemic mixtures that require extensive downstream purification. In contrast, the active site of the ketone carbonyl reductase is sterically constrained, physically preventing the binding and reduction of incorrect isomers or unrelated impurities. This "lock-and-key" mechanism ensures that the resulting product stream is exceptionally clean, with the patent reporting enantiomeric excess (ee) values as high as 99.8%. Such high optical purity is critical for regulatory compliance, as even trace amounts of the wrong enantiomer can pose toxicity risks or reduce the efficacy of the final antiretroviral drug. This mechanistic elegance translates directly into simplified isolation procedures and higher overall process yields.
How to Synthesize Lopinavir Intermediate Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for transitioning from laboratory scale to pilot production. The process is divided into distinct stages: the initial acidic deprotection to reveal the free amine, the controlled oxidation to install the ketone functionality, and finally, the biocatalytic reduction to set the chiral alcohol. Each step is optimized for maximum recovery and purity, utilizing common solvents like dichloromethane and ethyl acetate which are familiar to standard chemical manufacturing facilities. The biocatalytic step, in particular, is designed for ease of operation, requiring only moderate temperatures and standard stirring equipment. For process chemists looking to implement this route, the key lies in maintaining the precise pH and temperature conditions during the enzymatic phase to ensure maximal enzyme turnover. Detailed standardized operating procedures for scaling this specific chemo-enzymatic sequence are provided in the technical guide below.
- Deprotect Compound IV using trifluoroacetic acid in dichloromethane at 20-25°C to yield Compound III.
- Oxidize Compound III using sodium chlorate, potassium bromide, and TEMPO catalyst at -5 to 5°C to generate the ketone substrate, Compound II.
- Perform biocatalytic asymmetric reduction of Compound II using ketone carbonyl reductase and isopropanol dehydrogenase to obtain the chiral alcohol, Compound I.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this enzymatic synthesis route offers compelling strategic advantages beyond mere technical feasibility. The shift towards biocatalysis aligns perfectly with modern green chemistry initiatives, potentially reducing the regulatory burden associated with hazardous waste disposal. By eliminating the need for stoichiometric amounts of heavy metal reducing agents or toxic chiral resolving agents, the process significantly lowers the environmental footprint of the manufacturing site. This reduction in hazardous material usage not only simplifies compliance with environmental regulations but also mitigates the risks associated with the storage and handling of dangerous chemicals. Furthermore, the high selectivity of the enzymatic step reduces the complexity of downstream purification, leading to shorter batch cycles and increased throughput capacity. These operational efficiencies translate into a more resilient supply chain capable of meeting fluctuating market demands for antiretroviral medications without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The implementation of this enzymatic route drives cost efficiency primarily through the elimination of chiral resolution steps, which traditionally result in the loss of half the produced material. By achieving high enantioselectivity directly, the process maximizes the yield of the desired isomer from the starting materials, effectively doubling the utility of the raw material input compared to resolution-based methods. Additionally, the cofactor regeneration system minimizes the consumption of expensive NADPH, a significant cost driver in biocatalysis, making the process economically competitive with established chemical methods. The mild reaction conditions also reduce energy consumption associated with heating or cryogenic cooling, further contributing to lower operational expenditures.
- Enhanced Supply Chain Reliability: Reliance on complex chiral pools or specialized resolving agents can introduce vulnerabilities in the supply chain, especially when sourcing high-purity natural amino acids. This synthetic route utilizes robust, commercially available enzymes and simple chemical precursors that are less susceptible to supply disruptions. The ability to produce the intermediate with consistent high purity reduces the likelihood of batch failures or rejections during quality control testing, ensuring a steady flow of material to downstream API manufacturers. This reliability is crucial for maintaining continuous production schedules for life-saving HIV medications, where interruptions can have severe public health consequences.
- Scalability and Environmental Compliance: The transition from bench to plant is facilitated by the use of standard unit operations such as extraction, crystallization, and filtration, which are well-understood in the fine chemical industry. The aqueous nature of the biocatalytic step reduces the volume of organic solvents required, simplifying solvent recovery and waste treatment processes. This alignment with green chemistry principles not only enhances the corporate sustainability profile but also future-proofs the manufacturing process against increasingly stringent environmental regulations. The scalability of the enzyme system allows for flexible production volumes, enabling manufacturers to respond agilely to market needs without significant capital investment in new specialized equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the enzymatic synthesis of Lopinavir intermediates. These insights are derived directly from the experimental data and claims presented in patent CN113512569B, providing a factual basis for decision-making. Understanding the nuances of enzyme stability, substrate loading, and workup procedures is vital for successful technology transfer. We encourage potential partners to review these details closely to assess the fit for their specific manufacturing capabilities.
Q: What are the advantages of using enzymatic reduction over chemical resolution for Lopinavir intermediates?
A: Enzymatic reduction offers superior stereoselectivity, achieving ee values exceeding 99% without the need for cumbersome chiral resolution steps. This significantly reduces raw material loss and simplifies the purification process compared to traditional chemical methods.
Q: What are the critical reaction conditions for the biocatalytic step?
A: The biocatalytic step operates under mild conditions, typically around 33°C in a PB buffer system at pH 7.5-8.0. Isopropanol serves a dual role as both a co-solvent and a hydrogen donor for cofactor regeneration, ensuring efficient conversion.
Q: How does this process impact the impurity profile of the final intermediate?
A: The high specificity of the ketone carbonyl reductase minimizes the formation of diastereomers and side products. Combined with the high-purity oxidation step (99.4% purity), the overall process yields an intermediate with exceptional chemical and optical purity, facilitating downstream API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lopinavir Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving antiretroviral therapies. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN113512569B can be successfully translated to industrial reality. We are committed to delivering Lopinavir intermediates with stringent purity specifications, leveraging our rigorous QC labs to verify every batch against the highest international standards. Our state-of-the-art facilities are equipped to handle both chemical and biocatalytic processes, providing us with the flexibility to optimize synthesis routes for maximum efficiency and cost-effectiveness.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to secure a stable supply of this critical intermediate. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
