Advanced One-Pot Synthesis of Propionyl Triketone Ester for High-Efficiency Herbicide Manufacturing
Advanced One-Pot Synthesis of Propionyl Triketone Ester for High-Efficiency Herbicide Manufacturing
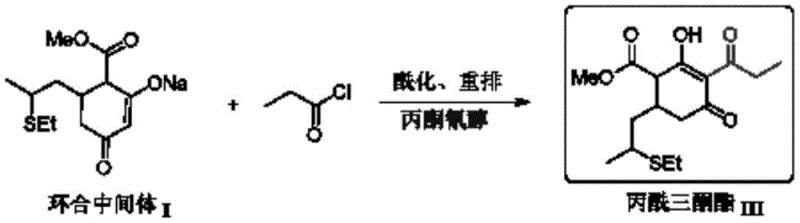
The global demand for high-efficiency herbicides like Clethodim necessitates robust and cost-effective supply chains for key intermediates. A groundbreaking technical development disclosed in patent CN115215775A introduces a revolutionary synthesis method for propionyl triketone ester, a critical precursor in Clethodim production. This innovation shifts the paradigm from traditional multi-step batch processes to a streamlined one-pot acylation rearrangement, utilizing acetone cyanohydrin as a highly efficient liquid organic catalyst. For R&D directors and procurement managers in the agrochemical sector, this represents a pivotal opportunity to optimize manufacturing protocols, reduce reliance on expensive catalysts like DMAP, and enhance overall process throughput. The following analysis dissects the technical merits and commercial implications of this novel pathway for reliable agrochemical intermediate suppliers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of propionyl triketone ester has relied on a disjointed two-step sequence involving distinct acylation and catalytic rearrangement stages. This conventional methodology typically employs expensive catalysts such as 4-Dimethylaminopyridine (DMAP) or solid molecular sieves, which introduce significant operational bottlenecks. The use of solid heterogeneous catalysts often results in poor mass transfer efficiency, complicating solid-liquid separation and leading to inconsistent reaction kinetics. Furthermore, the high cost of DMAP coupled with the difficulty in recovering and recycling these catalysts inflates the overall production expenditure. From a supply chain perspective, the necessity of isolating the acylation intermediate II adds unnecessary unit operations, extending cycle times and increasing the risk of material loss during transfer. These inefficiencies collectively hinder the ability to achieve cost reduction in herbicide manufacturing at a commercial scale.
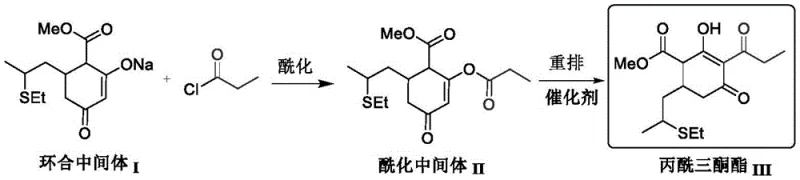
The Novel Approach
In stark contrast, the methodology outlined in patent CN115215775A achieves a direct transformation from cyclization intermediate I to the final propionyl triketone ester III in a single reactor vessel. By leveraging acetone cyanohydrin as a liquid-phase catalyst, the process enables a homogeneous liquid-liquid reaction system that eliminates the need for isolating the acylation intermediate II. This one-pot strategy not only drastically simplifies the operational workflow but also ensures superior dispersion of the catalyst within the reaction medium. The elimination of intermediate isolation steps translates to reduced solvent consumption, lower energy requirements for heating and cooling cycles, and minimized labor intervention. For a reliable agrochemical intermediate supplier, adopting this continuous or semi-continuous one-pot protocol offers a tangible pathway to enhancing production efficiency while maintaining high product purity standards required by downstream formulators.
Mechanistic Insights into Acetone Cyanohydrin-Catalyzed Acylation Rearrangement
The core innovation lies in the unique catalytic behavior of acetone cyanohydrin, which functions effectively in a homogeneous liquid phase alongside organic amines such as diisopropylethylamine or triethylamine. Unlike solid catalysts that rely on surface area availability, this liquid organic catalyst integrates seamlessly into the solvent matrix, typically toluene, ensuring uniform contact with the reactants. The mechanism involves the activation of the acylating agent, propionyl chloride, facilitating a rapid nucleophilic attack by the enolate form of the cyclization intermediate. This is immediately followed by an in-situ rearrangement that constructs the critical triketone backbone without the accumulation of stable intermediate species. The patent specifies that the molar amount of catalyst fed is preferably between 4% and 15% relative to propionyl chloride, a ratio that balances catalytic activity with economic feasibility. This precise control over stoichiometry prevents side reactions and ensures that the conversion rate of raw materials consistently exceeds 97%, with product selectivity maintained at or above 95%.
Furthermore, the reaction conditions are optimized to mitigate thermal degradation and impurity formation, which are common pitfalls in exothermic acylation reactions. The process operates within a temperature window of 50 to 95°C, with a preferred range of 70 to 85°C, allowing for controlled heat management during the dropwise addition of propionyl chloride over 1.5 to 4 hours. This gradual addition profile prevents localized hot spots that could lead to polymerization or decomposition of the sensitive triketone structure. Post-reaction workup is equally streamlined; the addition of water induces phase separation where the upper organic layer contains the crude product ready for subsequent hydrolysis and decarboxylation. The lower aqueous phase, containing residual catalyst, is easily treated by pH adjustment and oxidation, ensuring environmental compliance without complex purification trains. This robust mechanistic design underscores the feasibility of commercial scale-up of complex agrochemical intermediates with minimal technical risk.
How to Synthesize Propionyl Triketone Ester Efficiently
Implementing this advanced synthesis route requires careful attention to reagent quality and process parameters to replicate the high yields reported in the patent literature. The procedure begins with the preparation of a reaction mixture containing the cyclization intermediate, solvent, amine base, and the acetone cyanohydrin catalyst, followed by the controlled introduction of the acylating agent. Maintaining strict temperature control during the exothermic addition phase is critical to preventing runaway reactions and ensuring the formation of the desired regioisomer. Once the addition is complete, the mixture is held at elevated temperatures to drive the rearrangement to completion, monitored via liquid chromatography to confirm the disappearance of starting materials. The detailed standardized synthetic steps见下方的指南 ensure reproducibility and safety for pilot and plant-scale operations.
- Mix cyclization intermediate I with toluene solvent, organic amine (e.g., diisopropylethylamine), and acetone cyanohydrin catalyst in a reaction flask.
- Dropwise add propionyl chloride over 1.5 to 4 hours while maintaining temperature between 50 to 95°C.
- After addition, maintain heat for rearrangement reaction, then wash with water and separate the organic phase containing the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this one-pot synthesis methodology offers profound strategic benefits beyond mere technical elegance. The primary advantage stems from the substantial cost savings achieved by replacing high-value catalysts like DMAP with the significantly more affordable acetone cyanohydrin. This substitution directly impacts the bill of materials, lowering the variable cost per kilogram of the final intermediate without compromising quality. Additionally, the consolidation of two distinct process steps into a single operation reduces the total processing time, thereby increasing the asset utilization rate of existing reactor infrastructure. This efficiency gain allows manufacturers to respond more agilely to market fluctuations and urgent customer demands, effectively reducing lead time for high-purity agrochemical intermediates. The simplified workflow also minimizes the potential for human error and material loss during transfers, further enhancing the overall yield and reliability of the supply chain.
- Cost Reduction in Manufacturing: The economic model of this new process is driven by the elimination of expensive catalytic systems and the reduction of unit operations. By avoiding the use of solid molecular sieves or precious metal-based catalysts, the process removes the need for costly filtration and regeneration steps. The liquid nature of the catalyst allows for easier handling and potentially simpler recovery or disposal protocols, contributing to lower waste management expenses. Furthermore, the high selectivity of the reaction minimizes the formation of by-products, reducing the load on downstream purification units and saving on solvent and energy consumption. These cumulative effects result in a leaner manufacturing cost structure that enhances competitiveness in the global agrochemical market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available and stable raw materials such as propionyl chloride and acetone cyanohydrin. The robustness of the reaction conditions, which tolerate a reasonable range of temperatures and mixing rates, ensures consistent output even in large-scale production environments. The ability to produce the intermediate in fewer steps reduces the dependency on multiple processing lines, decreasing the likelihood of bottlenecks caused by equipment maintenance or scheduling conflicts. This streamlining ensures a steady flow of material to downstream customers, securing the continuity of herbicide production schedules and strengthening long-term supplier relationships.
- Scalability and Environmental Compliance: From an environmental standpoint, the process addresses key regulatory concerns regarding wastewater and hazardous waste. The patent details an effective treatment method for the aqueous waste stream, where residual cyanide is oxidized to safe levels using sodium hypochlorite under alkaline conditions. This proactive approach to effluent management ensures compliance with stringent environmental regulations, mitigating the risk of fines or operational shutdowns. The scalability of the liquid-liquid homogeneous system is inherently superior to heterogeneous slurry systems, as it avoids issues related to agitation power and heat transfer limitations in large vessels. This makes the technology ideally suited for expansion from pilot batches to multi-ton commercial production campaigns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on operational specifics. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their own manufacturing portfolios. The answers reflect the practical realities of scaling this chemistry while maintaining adherence to safety and quality standards.
Q: What are the advantages of using acetone cyanohydrin over DMAP in this synthesis?
A: Acetone cyanohydrin acts as a liquid organic catalyst that facilitates a homogeneous liquid-liquid reaction, unlike solid catalysts which suffer from separation issues. It significantly lowers catalyst costs compared to expensive 4-Dimethylaminopyridine (DMAP) and allows for a one-step process instead of two.
Q: How does the new process impact wastewater treatment?
A: The process generates an aqueous phase containing residual catalyst which can be effectively treated by adjusting pH to 9-12 and adding sodium hypochlorite, ensuring cyanide residues are reduced to below 0.5mg/L for compliant discharge.
Q: What is the expected conversion rate for this one-pot method?
A: According to patent data, the conversion rate of raw materials exceeds 97%, with product selectivity greater than or equal to 95%, demonstrating high efficiency suitable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Propionyl Triketone Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the one-pot acylation rearrangement technology described in patent CN115215775A for the agrochemical industry. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that validate every batch against the highest international standards. We are uniquely positioned to leverage this cost-effective synthesis route to deliver high-purity Propionyl Triketone Ester with unmatched consistency and reliability.
We invite forward-thinking partners to collaborate with us to capitalize on these technological advancements. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing capabilities can drive value for your herbicide supply chain. Let us be your strategic partner in achieving operational excellence and market leadership.
