Advanced Synthesis of 2-(2,4,6-Trichlorophenoxy)ethyl Propylamine for Agrochemical Production
The global demand for high-performance fungicides continues to drive innovation in the synthesis of critical agrochemical intermediates. Patent CN111574385B introduces a groundbreaking methodology for the production of 2-(2,4,6-trichlorophenoxy)ethyl propylamine, a pivotal precursor in the manufacturing of prochloraz. This technical disclosure addresses long-standing inefficiencies in traditional synthetic routes by leveraging a dual-catalyst system involving boron trifluoride diethyl etherate and cesium hydroxide monohydrate. For R&D directors and procurement specialists, this represents a significant shift towards safer, more cost-effective manufacturing protocols that do not compromise on chemical integrity. The patent details a robust two-step sequence that operates under remarkably mild conditions, avoiding the extreme pressures and temperatures that have historically plagued this sector. By optimizing reaction kinetics through specific Lewis acid and phase-transfer-like catalysis, the process achieves a total molar yield of 94.18 percent and a product purity exceeding 98.0 percent. This level of efficiency is not merely a laboratory curiosity but a viable blueprint for industrial scale-up, offering a reliable agrochemical intermediate supplier pathway that aligns with modern green chemistry principles and stringent regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-(2,4,6-trichlorophenoxy)ethyl propylamine has been fraught with operational hazards and economic inefficiencies that hinder large-scale adoption. One prevalent prior art method, documented in Chinese patent CN 101402608, relies on the reaction of 2,4,6-trichlorophenol with dichloroethane in the presence of liquid alkali to form an etherate intermediate. This approach is fundamentally flawed due to the propensity for elimination side reactions under strongly basic conditions, which drastically reduces yield and complicates downstream purification. Furthermore, the subsequent amination step with n-propylamine necessitates high-temperature and high-pressure environments, imposing severe demands on reactor metallurgy and safety systems. 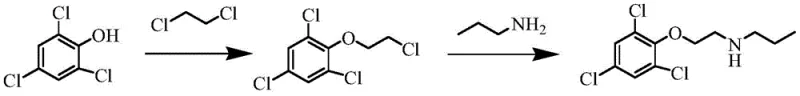 Another traditional route, reported by Wearangxia et al., initiates from 2-phenoxyethanol and involves chlorination followed by conversion to an ethyl chloride using thionyl chloride. This multi-step sequence not only generates hazardous waste streams but also requires high-pressure conditions for the final amination with N-propylamine.
Another traditional route, reported by Wearangxia et al., initiates from 2-phenoxyethanol and involves chlorination followed by conversion to an ethyl chloride using thionyl chloride. This multi-step sequence not only generates hazardous waste streams but also requires high-pressure conditions for the final amination with N-propylamine. 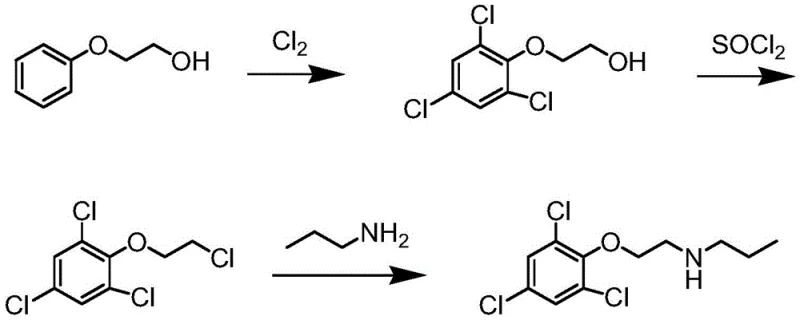 These legacy methods suffer from difficult reaction control, high equipment investment costs, and significant safety risks, making them increasingly obsolete in a competitive market focused on cost reduction in pesticide intermediate manufacturing.
These legacy methods suffer from difficult reaction control, high equipment investment costs, and significant safety risks, making them increasingly obsolete in a competitive market focused on cost reduction in pesticide intermediate manufacturing.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the methodology disclosed in CN111574385B offers a streamlined, two-step synthesis that operates under atmospheric pressure and moderate temperatures. The innovation lies in the strategic selection of catalysts that activate specific functional groups without inducing degradation. The first step utilizes boron trifluoride diethyl etherate to facilitate the nucleophilic attack of ethylamine on the phenolic substrate, a transformation that traditionally requires much harsher conditions. The second step employs cesium hydroxide monohydrate to promote the alkylation of the intermediate amine salt with 1-bromopropane. This novel approach effectively bypasses the need for high-pressure autoclaves and corrosive liquid alkalis, thereby simplifying the engineering requirements for commercial scale-up of complex agrochemical intermediates. The result is a process that is not only chemically superior in terms of selectivity but also economically advantageous due to reduced energy consumption and lower capital expenditure on specialized pressure vessels.
Mechanistic Insights into BF3 and CsOH Catalyzed Substitution
The core of this technological advancement rests on the precise mechanistic role of the catalysts in lowering activation energy barriers while maintaining high regioselectivity. In the initial formation of Compound A, boron trifluoride acts as a potent Lewis acid, coordinating with the oxygen atom of the 2,4,6-trichlorophenol to enhance the electrophilicity of the aromatic ring or facilitate the displacement mechanism depending on the specific pathway, ultimately allowing ethylamine to attach under mild thermal conditions of 50-55 ℃. 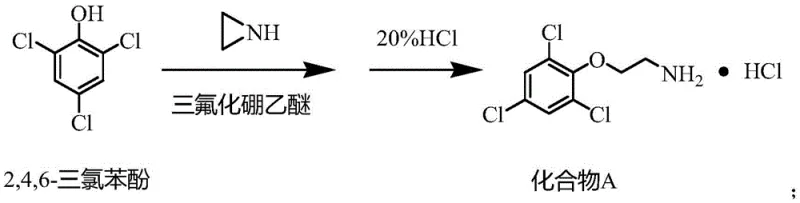 This mild thermal profile is crucial for preventing the thermal decomposition of the sensitive trichlorophenol backbone. Following the isolation of Compound A as a hydrochloride salt, the second stage involves a nucleophilic substitution where cesium hydroxide monohydrate plays a dual role. It acts to neutralize the amine salt, generating the free amine in situ, while the large cesium cation likely assists in solubilizing the nucleophile in the polar aprotic solvent DMF, enhancing its reactivity towards 1-bromopropane.
This mild thermal profile is crucial for preventing the thermal decomposition of the sensitive trichlorophenol backbone. Following the isolation of Compound A as a hydrochloride salt, the second stage involves a nucleophilic substitution where cesium hydroxide monohydrate plays a dual role. It acts to neutralize the amine salt, generating the free amine in situ, while the large cesium cation likely assists in solubilizing the nucleophile in the polar aprotic solvent DMF, enhancing its reactivity towards 1-bromopropane. 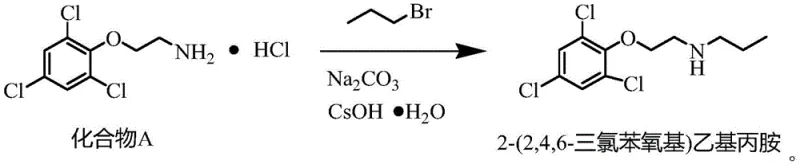 This synergy between the base and the solvent environment ensures that the alkylation proceeds rapidly at just 30-35 ℃, a temperature range that virtually eliminates the formation of elimination byproducts which are common in high-temperature aminations.
This synergy between the base and the solvent environment ensures that the alkylation proceeds rapidly at just 30-35 ℃, a temperature range that virtually eliminates the formation of elimination byproducts which are common in high-temperature aminations.
Controlling the impurity profile in such a densely functionalized molecule is paramount for its downstream utility in fungicide synthesis. The patent specifies rigorous monitoring via HPLC to ensure residual starting materials, such as 2,4,6-trichlorophenol, are reduced to less than 1 percent before proceeding to the next stage. The use of sodium carbonate as an acid-binding agent in the second step is particularly insightful; it buffers the reaction medium, preventing the accumulation of hydrobromic acid which could otherwise protonate the amine product and halt the reaction or lead to salt formation that complicates isolation. Furthermore, the workup procedure involves precise pH adjustments—first to acidic conditions to remove basic impurities, and then to neutral or slightly basic conditions to liberate the free amine. This meticulous control over the acid-base equilibrium ensures that the final product achieves a purity of over 98.0 percent, meeting the stringent specifications required for high-purity agrochemical intermediates used in sensitive biological applications.
How to Synthesize 2-(2,4,6-Trichlorophenoxy)ethyl Propylamine Efficiently
Implementing this synthesis route requires adherence to specific stoichiometric ratios and thermal profiles to maximize the reported 94.18 percent total molar yield. The process begins with the dissolution of 2,4,6-trichlorophenol in toluene, followed by the catalytic addition of ethylamine and boron trifluoride etherate. Operators must maintain the reaction temperature strictly between 50-55 ℃ during the dropwise addition and subsequent heat preservation period to ensure complete conversion without side reactions. Once Compound A is isolated and dried, it is transferred to a DMF solution containing cesium hydroxide monohydrate and sodium carbonate. The addition of 1-bromopropane must be controlled carefully at 30-35 ℃ to manage the exotherm and maintain selectivity. The detailed standardized synthetic steps, including exact weights, molar ratios, and workup procedures described in the patent, are outlined below for technical reference.
- React 2,4,6-trichlorophenol with ethylamine using boron trifluoride diethyl etherate catalyst in toluene at 50-55°C, followed by salification with hydrochloric acid to obtain Compound A.
- React Compound A with 1-bromopropane using cesium hydroxide monohydrate catalyst and sodium carbonate as an acid-binding agent in DMF at 30-35°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible benefits that extend far beyond simple yield improvements. The elimination of high-pressure reaction steps fundamentally alters the cost structure of manufacturing by removing the need for expensive autoclaves and specialized pressure-rated piping, leading to substantial cost savings in capital equipment investment. Additionally, the use of readily available and inexpensive raw materials such as ethylamine and 1-bromopropane, coupled with common solvents like toluene and DMF, ensures a stable and resilient supply chain that is less susceptible to the volatility associated with exotic reagents. The mild operating temperatures also translate to significantly reduced energy consumption for heating and cooling, further driving down the operational expenditure per kilogram of produced intermediate. This process optimization allows for a more predictable production schedule, reducing lead time for high-purity agrochemical intermediates and ensuring consistent availability for downstream formulation plants.
- Cost Reduction in Manufacturing: The replacement of harsh liquid alkali and high-pressure conditions with mild catalytic systems eliminates the need for costly corrosion-resistant alloys and pressure vessels, directly lowering CAPEX. Furthermore, the high selectivity of the BF3 and CsOH catalysts minimizes the formation of byproducts, reducing the burden on purification units and lowering waste disposal costs associated with hazardous chemical treatment.
- Enhanced Supply Chain Reliability: By utilizing commodity chemicals like toluene, DMF, and sodium carbonate, the process avoids reliance on specialized or single-source reagents that often bottleneck production. The robustness of the reaction conditions means that production can be maintained even with slight variations in raw material quality, ensuring continuous supply continuity for critical fungicide production lines without unexpected shutdowns.
- Scalability and Environmental Compliance: The atmospheric pressure operation and low-temperature profile make this process inherently safer and easier to scale from pilot plant to multi-ton commercial production. The simplified workup and reduced generation of hazardous elimination byproducts align with stricter environmental regulations, facilitating easier permitting and reducing the environmental footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on yield expectations, safety parameters, and scalability potential. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their own manufacturing portfolios.
Q: What are the key advantages of the BF3/CsOH catalytic route over traditional methods?
A: The novel route eliminates the need for high-pressure equipment and harsh liquid alkali conditions, significantly reducing safety risks and capital expenditure while achieving a total molar yield of 94.18%.
Q: How is product purity controlled in this synthesis method?
A: Purity is maintained through precise temperature control (30-55°C) and specific pH adjustments during workup, resulting in a final product purity exceeding 98.0% as verified by HPLC.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the use of common solvents like toluene and DMF, combined with mild reaction temperatures and atmospheric pressure operations, makes the process highly scalable and safe for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(2,4,6-Trichlorophenoxy)ethyl Propylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced agrochemicals depends on the reliability and quality of the underlying intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory patent to industrial reality is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-(2,4,6-trichlorophenoxy)ethyl propylamine meets the exacting standards required for prochloraz synthesis. We combine deep technical expertise with a commitment to operational excellence, making us the ideal partner for companies seeking to secure their supply chain against market fluctuations.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply network. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of technical confidence and commercial security.
