Revolutionizing Nitrogen Heterocycle Production with Metal-Free Base-Promoted Catalysis
Revolutionizing Nitrogen Heterocycle Production with Metal-Free Base-Promoted Catalysis
The landscape of organic synthesis for nitrogen-containing heterocycles is undergoing a significant transformation, driven by the urgent need for greener and more cost-effective manufacturing processes. Patent CN113087673B introduces a groundbreaking preparation method for alkyl and alkenyl substituted nitrogen-containing heterocyclic compounds that fundamentally shifts away from traditional transition metal catalysis. This innovation leverages a synergistic promotion system involving a strong base and a specific additive to achieve efficient C-alkylation and alkenylation of 2-methyl nitrogen heterocycles using primary alcohols. For R&D directors and procurement managers in the pharmaceutical and fine chemical sectors, this represents a pivotal opportunity to streamline supply chains and reduce the environmental footprint of intermediate production. The technology eliminates the reliance on precious metals, addressing critical pain points regarding residual metal contamination and raw material volatility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alkyl or alkenyl substituted nitrogen heterocycles has heavily depended on transition metal catalysts such as Ruthenium, Iridium, Platinum, Manganese, Iron, Nickel, or Cobalt. While these methods can be effective, they introduce substantial complexities into the manufacturing workflow. The use of precious metals inevitably drives up the cost of goods sold (COGS), making the final intermediates expensive and susceptible to market fluctuations in metal prices. Furthermore, the presence of metal residues in the final product is a major regulatory hurdle for pharmaceutical applications, necessitating additional purification steps like metal scavenging or extensive chromatography. These post-processing requirements not only extend the production lead time but also generate significant hazardous waste, complicating environmental compliance and increasing the overall cost of waste disposal.
The Novel Approach
In stark contrast, the method disclosed in CN113087673B achieves the same transformational chemistry through a metal-free and ligand-free pathway. By utilizing economically accessible 2-methyl nitrogen heterocycles and primary alcohols as starting materials, the process employs potassium tert-butoxide as a base and 18-crown-6 as an additive to drive the reaction forward. This approach bypasses the need for expensive metal catalysts entirely, resulting in a much cleaner reaction profile. The absence of metals means that the downstream purification is significantly simplified, often requiring only concentration and standard chromatographic separation. This novel strategy not only enhances the economic viability of producing these valuable intermediates but also aligns perfectly with the principles of green chemistry by reducing toxicity and waste generation.
Mechanistic Insights into Base-Promoted C-Alkylation and Alkenylation
The core of this technological breakthrough lies in the unique activation mechanism facilitated by the base-additive system. Potassium tert-butoxide acts as a strong base to deprotonate the methyl group at the 2-position of the nitrogen heterocycle, generating a reactive nucleophilic species. Simultaneously, the additive, specifically 18-crown-6, complexes with the potassium cation, enhancing the solubility and reactivity of the alkoxide species in the organic solvent, typically toluene. This synergistic effect lowers the activation energy for the nucleophilic attack on the primary alcohol, which serves as the alkylating or alkenylating agent. Unlike metal-catalyzed borrowing hydrogen methodologies, this base-promoted pathway avoids the formation of metal-hydride intermediates, thereby eliminating a major source of potential side reactions and impurities associated with metal coordination spheres.
From an impurity control perspective, this mechanism offers distinct advantages for producing high-purity pharmaceutical intermediates. Since no transition metals are involved, the risk of metal-catalyzed decomposition or isomerization of sensitive functional groups is minimized. The reaction conditions are tunable, with alkylation typically occurring at higher temperatures around 150°C and alkenylation at slightly lower temperatures around 100°C, allowing for selective formation of the desired product. The robustness of this base-mediated system ensures consistent batch-to-batch reproducibility, a critical factor for maintaining stringent quality standards in API synthesis. The resulting products, whether alkyl or alkenyl substituted, exhibit high structural integrity with minimal byproduct formation, facilitating easier isolation and characterization.
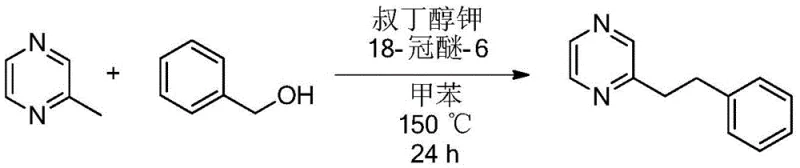
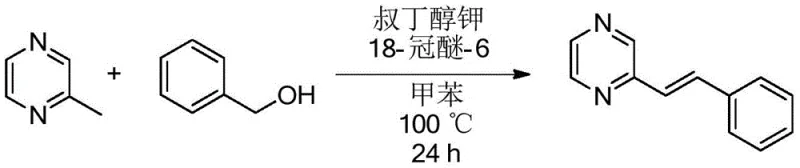
How to Synthesize Alkyl/Alkenyl Substituted Nitrogen Heterocycles Efficiently
The practical implementation of this synthesis route is straightforward and designed for operational ease in both laboratory and pilot plant settings. The process begins by combining the 2-methyl nitrogen heterocycle, the chosen primary alcohol, the base, and the additive in an organic solvent such as toluene. The mixture is then heated under stirring for a defined period, typically 24 hours, to ensure complete conversion. Following the reaction, the workup involves simple concentration of the reaction liquid followed by purification, often using thin-layer or column chromatography with a hexane and ethyl acetate system. This streamlined workflow minimizes unit operations and reduces the demand for specialized equipment, making it highly attractive for cost-sensitive manufacturing environments.
- Mix 2-methyl nitrogen heterocycle, primary alcohol, potassium tert-butoxide, and 18-crown-6 in toluene.
- Heat the mixture to 150°C for alkylation or 100°C for alkenylation under stirring for 24 hours.
- Concentrate the reaction liquid and purify via column chromatography using n-hexane and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis technology translates into tangible strategic benefits that extend beyond mere technical feasibility. The primary driver for value creation is the drastic reduction in raw material costs associated with eliminating precious metal catalysts. By replacing expensive metals like Ruthenium or Iridium with commodity chemicals like potassium tert-butoxide, the direct material cost of the synthesis is significantly lowered. Furthermore, the simplification of the purification process reduces the consumption of solvents and adsorbents, contributing to additional operational savings. This cost structure provides a competitive edge in pricing negotiations and improves margin stability against volatile metal markets.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes a significant cost center from the bill of materials. Precious metals not only carry a high price tag but also require recovery processes to be economically viable, adding capital expenditure for recovery units. By adopting this base-promoted method, manufacturers can avoid these capital and operational expenses entirely. Additionally, the use of common solvents like toluene and readily available bases ensures a stable and low-cost supply chain for reagents. The overall process efficiency is enhanced by the high yields reported, reaching up to 95% in specific examples, which maximizes the output per unit of input material and further drives down the unit cost of production.
- Enhanced Supply Chain Reliability: Relying on precious metals introduces supply chain risks related to geopolitical instability and mining constraints. This new method utilizes commodity chemicals that are produced in vast quantities globally, ensuring a secure and continuous supply of critical reagents. The robustness of the reaction conditions, which do not require inert gas protection, simplifies the logistical requirements for storage and handling. This resilience allows for more flexible production scheduling and reduces the risk of delays caused by reagent shortages. Consequently, lead times for delivering high-purity intermediates can be optimized, providing greater reliability to downstream pharmaceutical customers who depend on just-in-time delivery models.
- Scalability and Environmental Compliance: The simplicity of the reaction setup facilitates seamless scale-up from gram to ton scales without the need for complex engineering controls associated with air-sensitive metal catalysts. The absence of heavy metals simplifies wastewater treatment and waste disposal, reducing the environmental compliance burden and associated fees. This green chemistry profile aligns with increasingly strict global environmental regulations, future-proofing the manufacturing process against tighter emission standards. The ability to scale efficiently while maintaining high purity makes this technology ideal for the commercial production of complex pharmaceutical intermediates and fine chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this metal-free synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into existing manufacturing lines.
Q: What are the key advantages of this metal-free synthesis method?
A: The primary advantage is the elimination of expensive and toxic transition metal catalysts like Ruthenium or Iridium. This significantly reduces raw material costs and simplifies the purification process by removing the need for heavy metal scavenging steps, ensuring higher purity suitable for pharmaceutical applications.
Q: Can this method be scaled for industrial production?
A: Yes, the process utilizes inexpensive and readily available reagents such as potassium tert-butoxide and toluene. The reaction conditions are robust, operating at standard heating temperatures without the need for inert gas protection, which facilitates easy scale-up from laboratory to commercial manufacturing.
Q: What types of substrates are compatible with this reaction?
A: The method demonstrates broad substrate scope, accommodating various 2-methyl nitrogen heterocycles including pyrazines, quinolines, and benzoxazoles. It effectively couples with a wide range of primary alcohols, including benzyl alcohols with different substituents and linear alkyl alcohols, to produce both alkyl and alkenyl substituted derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkyl/Alkenyl Substituted Nitrogen-Containing Heterocyclic Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free synthesis technologies in modernizing the production of critical pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which guarantee that every batch of alkyl or alkenyl substituted nitrogen heterocycles meets the highest industry standards. We are well-equipped to handle the specific nuances of base-promoted reactions, optimizing parameters to maximize yield and minimize impurities for your specific application needs.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can enhance your supply chain efficiency and product quality. Together, we can drive innovation and value in the development of high-performance nitrogen-containing heterocyclic compounds.
