Advanced Synthesis of Renieramycin Intermediates: A Scalable Pictet-Spengler Strategy for Commercial API Production
Advanced Synthesis of Renieramycin Intermediates: A Scalable Pictet-Spengler Strategy for Commercial API Production
The development of efficient synthetic routes for complex marine alkaloids remains a critical challenge in modern pharmaceutical chemistry, particularly for compounds exhibiting potent anticancer activity. Patent CN103304478A introduces a groundbreaking methodology for the preparation of a novel class of bis-tetrahydroisoquinoline compounds, serving as advanced intermediates for the synthesis of renieramycin-type alkaloids. These natural products, including renieramycins, saframycins, and ecteinascidins, possess a distinctive five-ring rigid backbone that confers significant biological activity against various human cancer cell lines. The structural complexity of these molecules, as illustrated in the general framework below, has historically hindered their widespread clinical application due to low natural abundance and difficult total synthesis.
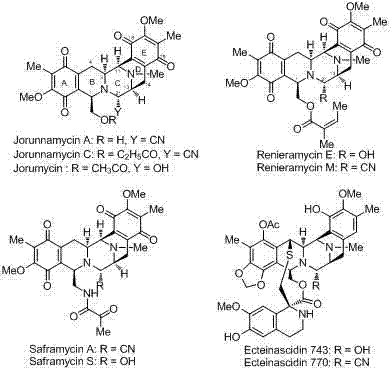
This patent discloses a highly efficient pathway that overcomes traditional bottlenecks by utilizing a convergent Pictet-Spengler cyclization strategy. By constructing the core bis-tetrahydroisoquinoline skeleton through the coupling of an aldehyde fragment containing a tetrahydroisoquinoline unit with a trisubstituted phenylalaninol fragment, the inventors have achieved a process characterized by high yield and exceptional stereocontrol. For procurement managers and supply chain directors in the fine chemical sector, this innovation represents a pivotal shift towards more economically viable manufacturing of high-value pharmaceutical intermediates. The ability to access these complex scaffolds from cheap, renewable chiral pools like L-Tyrosine drastically alters the cost structure of producing next-generation antitumor agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the asymmetric synthesis of renieramycin alkaloids was plagued by inefficient multi-step sequences and reliance on expensive reagents. As depicted in the reaction schemes of earlier literature, methods developed by researchers such as Williams involved coupling two chirality-defined, tetrasubstituted benzene alanine derivatives via an amide linkage, followed by ring closure. While chemically elegant, these routes often suffered from lengthy synthetic sequences and the necessity of preparing multiple distinct fragments, which inflated production costs. Furthermore, alternative strategies reported by Liu and others utilized similar coupling tactics but relied on tetrasubstituted benzene L-alanine fragments that were difficult and costly to prepare from methyl esters of tyrosine.
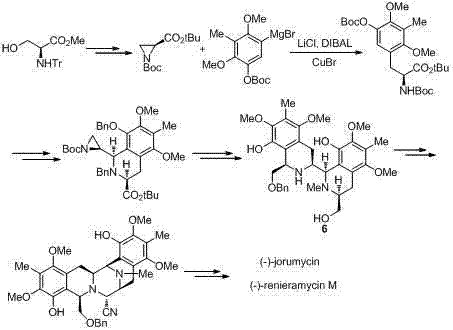
Another notable approach by Zhu involved the reaction of lower ring nitrogen compounds with aryl Grignard reagents, followed by dual cyclizations to obtain tetrahydroisoquinoline intermediates. Although this method successfully yielded targets like (-)-jorumycin, it required harsh reaction conditions and expensive organometallic reagents that pose significant safety and handling challenges on an industrial scale. The cumulative effect of these limitations—low overall yields, harsh operational parameters, and high raw material costs—rendered existing methods unsuitable for the large-scale preparation required to support clinical development and commercial supply of these potent therapeutics.
The Novel Approach
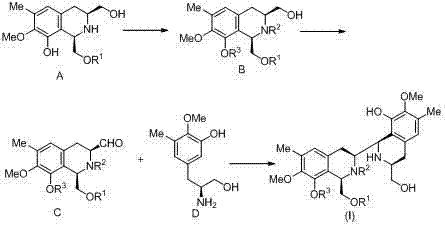
In stark contrast to the cumbersome methodologies of the past, the present invention offers a succinct and robust solution centered on the Pictet-Spengler cyclization. This novel approach constructs the critical C-N bond and the associated ring system in a single, high-yielding transformation between aldehyde compound C and amino alcohol compound D. The elegance of this strategy lies in its atom economy and the use of readily accessible starting materials derived efficiently from L-Tyrosine. By avoiding the need for expensive Grignard reagents or complex amide coupling steps, the new route significantly simplifies the process flow. The reaction conditions are remarkably mild, typically employing acetic acid as a catalyst and molecular sieves as a water-retaining agent in a mixed solvent system, allowing for precise control over the reaction environment.
The versatility of this method is further enhanced by the flexibility in protecting group selection, with benzyl, Boc, and allyl groups preferred for optimal results. The cyclization can be performed across a broad temperature range from -20°C to 120°C, providing process engineers with the flexibility to optimize kinetics and selectivity based on specific reactor capabilities. This adaptability, combined with the high isolated yields reported (up to 89% for key steps), establishes this Pictet-Spengler protocol as a superior platform for the commercial scale-up of complex pharmaceutical intermediates, effectively addressing the supply chain vulnerabilities associated with previous synthetic routes.
Mechanistic Insights into Acid-Catalyzed Pictet-Spengler Cyclization
The core of this technological breakthrough is the acid-catalyzed condensation between the aldehyde functionality of fragment C and the amine group of fragment D. Mechanistically, the reaction initiates with the formation of an iminium ion intermediate, facilitated by the protonation of the carbonyl oxygen by acetic acid. This electrophilic species is then attacked by the electron-rich aromatic ring of the tetrahydroisoquinoline moiety in an intramolecular electrophilic aromatic substitution. The presence of molecular sieves is critical in this mechanism, as they sequester the water byproduct, driving the equilibrium towards the formation of the cyclic product and preventing hydrolysis of the sensitive iminium intermediate. This dehydration step is essential for achieving the high conversion rates observed in the patent examples.
From an impurity control perspective, the stereoselectivity of this cyclization is governed by the pre-existing chirality of the L-Tyrosine-derived side chains. The rigid transition state imposed by the bulky protecting groups (such as the tert-butoxycarbonyl or benzyl groups) directs the approach of the nucleophile, ensuring the formation of the desired diastereomer with high fidelity. This intrinsic stereocontrol minimizes the generation of unwanted isomers that would otherwise require resource-intensive chromatographic separation. Furthermore, the use of mild Lewis or Brønsted acids prevents the degradation of sensitive functional groups, such as the phenolic ethers, which might be compromised under the harsher conditions required by Grignard-based alternatives. This mechanistic robustness ensures a clean reaction profile, directly translating to higher purity specifications for the final high-purity pharmaceutical intermediates.
How to Synthesize Bis-tetrahydroisoquinoline Intermediate Efficiently
The practical implementation of this synthesis involves a sequential preparation of the two coupling fragments followed by the key cyclization event. First, the amino alcohol precursor (Compound B) is subjected to Swern oxidation conditions using oxalyl chloride and dimethyl sulfoxide at low temperatures (-80°C to 40°C) to generate the reactive aldehyde Compound C. This aldehyde is then immediately engaged in the Pictet-Spengler reaction with the trisubstituted phenylalaninol fragment (Compound D) in a mixture of dichloromethane and trifluoroethanol. The detailed standardized operating procedures, including specific molar ratios, addition rates, and workup protocols for maximizing yield and purity, are outlined in the comprehensive guide below.
- Preparation of Aldehyde Fragment C: Oxidize the protected amino alcohol precursor (Compound B) using Swern oxidation conditions (oxalyl chloride, DMSO) at low temperature to generate the reactive aldehyde species.
- Pictet-Spengler Cyclization: React Aldehyde C with the trisubstituted phenylalaninol fragment (Compound D) in a mixed solvent system (dichloromethane/trifluoroethanol) with acetic acid catalysis and molecular sieves to form the bis-tetrahydroisoquinoline core.
- Downstream Functionalization: Convert the resulting intermediate (Compound E) through deprotection, oxidation, and cyanation steps to access final bioactive alkaloids like (-)-Jorunnamycin A.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers transformative benefits in terms of cost structure and supply reliability. The shift from expensive, multi-step sequences to a convergent, high-yield process directly addresses the economic barriers that have historically limited the availability of renieramycin-class drugs. By leveraging cheap and abundant L-Tyrosine as the foundational chiral building block, manufacturers can decouple production costs from the volatility of specialized reagent markets. This strategic sourcing advantage ensures a more stable pricing model for long-term supply agreements, making the development of these potent anticancer agents more financially sustainable for pharmaceutical partners.
- Cost Reduction in Manufacturing: The elimination of costly organometallic reagents, such as aryl Grignard reagents, and the reduction in the total number of synthetic steps result in substantial cost savings. The high yield of the key cyclization step (reported up to 89%) minimizes material loss and reduces the burden on downstream purification processes. Furthermore, the use of common solvents and simple catalysts like acetic acid lowers the operational expenditure associated with solvent recovery and waste disposal, contributing to a leaner and more cost-effective manufacturing footprint.
- Enhanced Supply Chain Reliability: The reliance on L-Tyrosine, a commodity amino acid with a robust global supply chain, mitigates the risk of raw material shortages that often plague specialty chemical synthesis. The operational simplicity of the process, which avoids cryogenic conditions and pyrophoric reagents, allows for production in a wider range of facilities, thereby diversifying the potential supplier base. This flexibility enhances supply continuity and reduces the lead time for delivering critical intermediates to drug development pipelines.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of heavy metal catalysts simplify the scale-up process from laboratory to commercial production. The process generates less hazardous waste compared to traditional methods involving stoichiometric metal reagents, aligning with increasingly stringent environmental regulations. This green chemistry profile not only reduces compliance costs but also appeals to environmentally conscious stakeholders, facilitating smoother regulatory approvals for the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and advantageous effects described in the patent documentation, providing clarity on the feasibility and benefits of adopting this route for industrial applications.
Q: What represents the primary cost advantage of this new synthetic route compared to previous methods?
A: The primary economic advantage lies in the utilization of inexpensive L-Tyrosine as the sole chiral pool starting material for both coupling fragments. Unlike prior art requiring expensive Grignard reagents or complex amide couplings, this Pictet-Spengler approach utilizes readily available reagents and achieves high overall yields, significantly reducing raw material expenditure.
Q: How does the Pictet-Spengler cyclization ensure stereochemical purity in the final intermediate?
A: The reaction leverages the inherent chirality of the L-Tyrosine-derived fragments. The cyclization proceeds with excellent stereo- and regioselectivity under mild acidic conditions, minimizing the formation of diastereomeric impurities and reducing the need for difficult downstream purification steps.
Q: Is this synthesis method suitable for large-scale industrial manufacturing?
A: Yes, the process is designed for scalability. It operates under mild temperature ranges (-20°C to 120°C) and avoids hazardous or highly sensitive reagents. The use of standard solvents like dichloromethane and simple workup procedures (filtration of molecular sieves, extraction) facilitates easy translation from laboratory to commercial production scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Renieramycin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of this novel Pictet-Spengler strategy to revolutionize the production of bis-tetrahydroisoquinoline alkaloids. As a premier CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent to plant is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific solvent systems and protection group chemistries required by this route, while our rigorous QC labs guarantee that every batch meets stringent purity specifications essential for oncology drug development.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cost-effective technology for their pipeline programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain for these critical anticancer intermediates is both robust and economically optimized for the future.
