Advanced Manufacturing of Bis-Benzenesulfonimide: A High-Purity Electroplating Intermediate Solution
The global electroplating industry is currently undergoing a significant transformation driven by the demand for higher quality nickel plating that is both durable and aesthetically superior. Traditional additives like saccharin, while effective as levelers and softeners, often suffer from critical drawbacks such as causing the plating layer to turn yellow over time, which compromises the final product's visual appeal and longevity. Addressing these challenges requires advanced chemical solutions, specifically high-purity intermediates that can replace traditional additives without introducing new defects. Patent CN101671285A introduces a groundbreaking preparation method for Bis-benzenesulfonimide (BBI), a specialized electroplating intermediate capable of replacing up to 90% of saccharin usage while eliminating the risk of yellowing. This technology represents a pivotal shift in specialty chemical manufacturing, offering a robust alternative to imported German products that have historically dominated the market due to their superior solubility profiles. By leveraging a novel aqueous alkaline condensation process, this method ensures that the final BBI product exhibits exceptional dissolution characteristics, preventing the accumulation of insoluble matter in plating tanks that often leads to efficiency losses. For R&D directors and procurement specialists alike, understanding the nuances of this synthesis is crucial for securing a reliable electroplating intermediate supplier who can deliver consistent quality at a competitive price point.
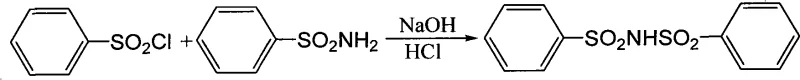
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of BBI and similar sulfonimide derivatives has been plagued by significant technical hurdles that directly impact the downstream application in electroplating baths. Conventional synthesis routes often fail to adequately separate the target imide from unreacted starting materials, particularly benzenesulfonamide, which has limited solubility in neutral or acidic conditions typically used in workup procedures. This incomplete purification results in a final product containing insoluble impurities that precipitate out in the plating tank, creating roughness on the plated surface and reducing the overall throwing power of the bath. Furthermore, many existing domestic production methods struggle to match the solubility standards set by international giants like BASF, forcing manufacturers to rely on expensive imports that strain operational budgets. The reliance on these inferior or costly alternatives creates a bottleneck in the commercial scale-up of complex specialty chemicals, as plating facilities cannot afford the downtime associated with filtering out insoluble residues or the variability in plating quality. Additionally, the lack of efficient recycling mechanisms for unreacted raw materials in older processes leads to higher waste generation and increased raw material consumption, further exacerbating the cost structure and environmental footprint of the manufacturing operation.
The Novel Approach
The innovative methodology described in the patent fundamentally reengineers the purification landscape by exploiting the differential solubility of BBI sodium salts versus benzenesulfonamide in alkaline environments. Instead of relying on traditional acidification steps that might co-precipitate impurities, this process maintains a strictly controlled alkaline environment (pH 8-9) during the reaction and initial separation phases. This strategic pH control ensures that the sodium salt of BBI precipitates selectively from the filtrate, while any unreacted benzenesulfonamide remains dissolved in the alkaline mother liquor due to its favorable solubility properties under these specific conditions. This elegant separation mechanism not only guarantees a product purity of ≥99.5% but also significantly enhances the water solubility of the final BBI, allowing up to 6g to dissolve in 100g of water at room temperature without solid residue. Moreover, the process incorporates a built-in recycling loop where the unreacted benzenesulfonamide, separated during the initial centrifugation, is directly reused in subsequent batches, thereby optimizing raw material utilization. This approach effectively breaks the technological blockade previously held by foreign entities, enabling local manufacturers to produce high-performance high-purity electroplating intermediates that rival the best global standards while drastically simplifying the purification workflow.
Mechanistic Insights into Aqueous Alkaline Condensation
The core chemical transformation involves the nucleophilic attack of the sulfonamide nitrogen on the sulfonyl chloride sulfur, facilitated by the presence of a base which acts as an acid scavenger to drive the equilibrium forward. In this specific protocol, sodium hydroxide serves a dual purpose: it initially dissolves the benzenesulfonamide to form a reactive species and subsequently neutralizes the hydrochloric acid byproduct generated during the formation of the sulfonimide bond. The reaction is conducted at a precise temperature range of 53-57°C, which is critical for balancing reaction kinetics with selectivity; temperatures below this range slow down the conversion, while excessive heat can promote side reactions or hydrolysis of the sensitive sulfonyl chloride moiety. Following the initial addition of benzenesulfonyl chloride, the mixture is held at this temperature for 2-4 hours to ensure complete conversion, after which a reflux step at 100-105°C is employed. This high-temperature reflux is not merely for acceleration but serves a vital purification function by hydrolyzing any residual unreacted benzenesulfonyl chloride into water-soluble benzenesulfonic acid, which is easily removed in the aqueous phase, thus preventing chlorine-containing impurities from contaminating the final crystal lattice.
Impurity control is further refined through the unique precipitation strategy employed in the workup phase. By adjusting the pH to between 4.0 and 7.5 using hydrochloric acid only after the selective precipitation of the BBI sodium salt, the process ensures that the final free acid form of BBI crystallizes with minimal inclusion of organic byproducts. The mechanistic advantage lies in the fact that benzenesulfonamide, being more soluble in the alkaline filtrate than the BBI sodium salt, is effectively washed away before the final acidification step. This contrasts sharply with methods that acidify the entire reaction mass immediately, which would cause both the product and the starting material to crash out together, necessitating difficult and yield-reducing recrystallization steps. The result is a crystalline product with a sharp melting range of 150-158°C and a white to pale yellow appearance, indicative of high chemical integrity. For technical teams, this mechanism underscores the importance of precise pH and temperature control in achieving the cost reduction in specialty chemical manufacturing by minimizing the need for energy-intensive recrystallization or chromatographic purification.
How to Synthesize Bis-Benzenesulfonimide Efficiently
The synthesis of BBI via this patented route offers a streamlined pathway that balances high yield with operational simplicity, making it highly attractive for industrial adoption. The process begins with the preparation of an alkaline solution where sodium hydroxide is dissolved in water and heated, creating the necessary medium for the subsequent dissolution of benzenesulfonamide. Once the amide is fully solubilized at 53-57°C, benzenesulfonyl chloride is introduced dropwise to manage the exotherm and maintain reaction control, followed by a critical holding period to maximize conversion. The detailed standardized synthetic steps, including specific reagent quantities, stirring rates, and precise filtration protocols required to replicate this high-purity outcome, are outlined in the technical guide below.
- Dissolve sodium hydroxide in water and heat to 50-57°C, then add benzenesulfonamide maintaining temperature at 53-57°C until fully dissolved.
- Dropwise add benzenesulfonyl chloride over 30 minutes while maintaining pH 8-9 with supplemental NaOH, followed by保温 (holding) for 2-4 hours.
- Reflux at 100-105°C for 1 hour to hydrolyze residual chloride, cool, acidify to pH 4.0-7.5, and separate the solid product via centrifugation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthesis route translates into tangible strategic benefits that extend beyond mere technical specifications. The ability to produce BBI domestically with quality equivalent to premium European imports eliminates the reliance on volatile international supply chains and reduces exposure to currency fluctuations and long lead times associated with overseas shipping. Furthermore, the process design inherently supports reducing lead time for high-purity electroplating intermediates by utilizing readily available commodity chemicals such as benzenesulfonyl chloride, benzenesulfonamide, and caustic soda, which are stable and easy to source in bulk quantities. The integration of a raw material recycling loop means that the effective consumption of the more expensive sulfonamide reagent is minimized, as unreacted material is recovered and fed back into the process, driving down the variable cost per kilogram of finished product without compromising on quality standards.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the elimination of expensive purification steps and the recovery of unreacted starting materials. By avoiding the need for complex solvent extraction or multiple recrystallizations typically required to remove sulfonamide impurities, the operational expenditure on solvents, energy, and labor is substantially reduced. Additionally, the direct recycling of the solid residue (unreacted benzenesulfonamide) recovered during centrifugation means that the net raw material cost is lowered, as less fresh feedstock is required per unit of output. This efficiency allows manufacturers to offer a highly competitive pricing structure compared to imported alternatives, providing a clear margin advantage for downstream electroplating facilities looking to optimize their bill of materials.
- Enhanced Supply Chain Reliability: Dependence on single-source foreign suppliers for critical plating additives poses a significant risk to production continuity, especially given the historical technology blockades mentioned in the patent background. Implementing this localized synthesis method diversifies the supply base and ensures a steady flow of materials, as the raw ingredients are common industrial chemicals with robust global availability. The simplified process flow, which relies on standard reactor equipment (stainless steel kettles with reflux condensers) and common unit operations like centrifugation and vacuum drying, facilitates rapid scale-up and reduces the risk of equipment-related bottlenecks. This reliability ensures that plating shops can maintain continuous operation without the fear of sudden shortages or quality deviations that often accompany long-distance logistics.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the aqueous nature of the reaction medium offers distinct advantages over solvent-heavy organic syntheses. The use of water as the primary solvent reduces the emission of volatile organic compounds (VOCs) and simplifies wastewater treatment protocols, aligning with increasingly stringent environmental regulations. The process generates minimal hazardous waste, as the primary byproduct is sodium chloride (from the neutralization of HCl), which is manageable within standard industrial effluent systems. Furthermore, the scalability is proven by the patent's successful transition from laboratory to pilot scales, demonstrating that the heat transfer and mixing requirements can be met in large-scale reactors without losing the critical control over temperature and pH that defines the product quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Bis-benzenesulfonimide, derived directly from the patent's experimental data and comparative analysis. These insights are intended to clarify the operational parameters and quality benchmarks associated with this specific manufacturing route, helping stakeholders make informed decisions about integrating this intermediate into their supply chains. Understanding these details is essential for validating the process capability against internal quality assurance standards.
Q: How does this process improve the solubility of BBI compared to conventional methods?
A: The patented method utilizes a specific alkaline precipitation technique where the sodium salt of BBI precipitates directly from the filtrate, while unreacted benzenesulfonamide remains dissolved due to its high solubility in the alkaline solution, resulting in a product with significantly improved water solubility (6g/100g water).
Q: What is the expected purity and yield of the BBI produced via this route?
A: According to the patent data, the process consistently achieves a chemical purity of ≥99.5% with a yield reaching approximately 86%, matching the quality standards of leading international competitors like BASF.
Q: Can unreacted raw materials be recovered in this synthesis?
A: Yes, the process includes a separation step where unreacted benzenesulfonamide is isolated as a solid during the initial centrifugation and can be directly recycled into the next production batch, enhancing overall atom economy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bis-Benzenesulfonimide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to high-performance electroplating additives requires a partner with deep technical expertise and a commitment to quality consistency. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate balance of pH and temperature required for BBI synthesis is maintained even at full industrial scale. We operate stringent purity specifications and utilize rigorous QC labs to verify that every batch meets the ≥99.5% purity threshold and the critical solubility metrics necessary for defect-free nickel plating. Our infrastructure is designed to support the complex needs of the fine chemical sector, delivering materials that not only meet but exceed the performance of legacy imported products.
We invite you to collaborate with us to optimize your electroplating formulations and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific consumption volumes. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our advanced BBI synthesis can enhance your plating quality while securing your supply chain against global disruptions.
