Advanced Synthetic Route for Vildagliptin Intermediate Ensuring High Purity and Scalability for Global Pharma Supply Chains
Advanced Synthetic Route for Vildagliptin Intermediate Ensuring High Purity and Scalability for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust synthetic pathways for critical antidiabetic agents, particularly for the next-generation DPP-IV inhibitor Vildagliptin. A pivotal breakthrough in this domain is documented in Chinese Patent CN112028806B, which discloses a highly efficient synthetic method for the key intermediate, (S)-1-(2-chloroacetyl)pyrrolidine-2-carbonitrile. This specific molecular architecture serves as the foundational building block for Vildagliptin, a drug with immense market potential due to its efficacy in regulating blood glucose levels when combined with other hypoglycemic agents. The structural integrity of this intermediate is paramount, as any stereochemical deviation or impurity carryover can compromise the safety profile of the final Active Pharmaceutical Ingredient (API). The patent outlines a sophisticated three-step sequence starting from L-prolinamide, leveraging phase transfer catalysis and a novel stabilization strategy during the dehydration cyanation step to achieve unprecedented purity levels.
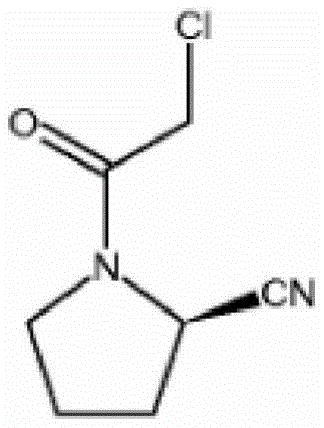
For R&D directors and process chemists, the significance of this patent lies in its ability to resolve long-standing issues regarding impurity profiles and yield loss. Traditional routes often struggle with the reactivity of the imino group, leading to complex byproduct mixtures that are notoriously difficult to separate. By implementing a strategic Boc-protection followed by a stabilized cyanuric chloride dehydration and a salt-formation purification step, this methodology effectively isolates the desired chiral nitrile species. This approach not only simplifies the downstream processing but also establishes a reliable benchmark for quality that aligns with the stringent regulatory requirements of global health authorities. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic nuances is essential for delivering consistent, high-quality materials to our partners.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (S)-1-(2-chloroacetyl)pyrrolidine-2-carbonitrile has been plagued by significant technical hurdles that impact both cost and quality. Early methodologies, such as those described in WO2000034241A1, attempted a direct chloroacetylation and cyanidation on L-prolinamide without adequate protection of the reactive imino group. This lack of protection inevitably resulted in the generation of numerous amino-impurity byproducts during the amide dehydration and cyanidation phases. Consequently, the crude product required extensive and yield-depleting purification processes, leading to poor overall recovery rates that worsened as production scales increased. Furthermore, subsequent improvements like WO2011101861 introduced Boc-protection but relied on trifluoroacetic anhydride (TFAA) as a dehydrating agent. While TFAA is effective, it is prohibitively expensive and generates acidic byproducts that complicate waste treatment and increase the environmental footprint of the manufacturing process.
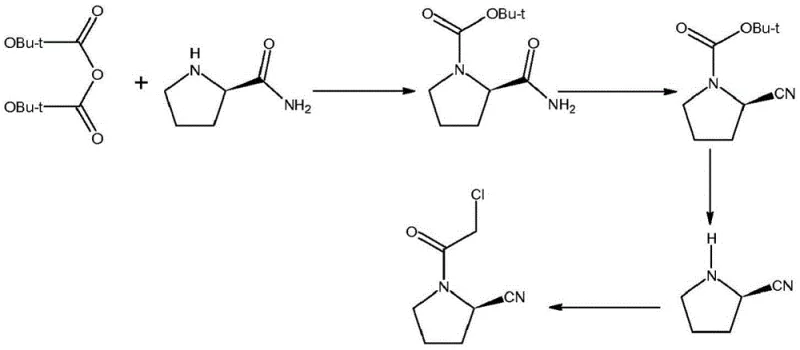
Even more recent attempts, such as the route depicted in WO2014020462A1 which utilizes the cheaper cyanuric chloride, have faced stability issues. In these conventional cyanuric chloride protocols, the dehydration process is often incomplete, and the material system remains unstable throughout the reaction. This instability leads to the accumulation of impurities, particularly (S)-1-(2-chloroacetyl)pyrrolidine-2-formamide, which possesses physicochemical properties strikingly similar to the target product. Separating these structurally analogous impurities is exceptionally challenging, often necessitating aggressive crystallization conditions that sacrifice substantial amounts of product yield. The absence of an intermediate crystallization step in these prior art routes means that impurities generated during the Boc protection and dehydration stages are carried through to the final step, directly compromising the quality of the downstream Vildagliptin API.
The Novel Approach
The methodology presented in CN112028806B represents a paradigm shift by introducing critical stabilization agents directly into the reaction matrix. Unlike previous iterations, this novel approach performs the dehydration cyanation in a pure DMF system but uniquely incorporates acid carbonate (such as sodium bicarbonate) and anhydrous sulfate at a precise juncture. This addition serves a dual purpose: it neutralizes acidic byproducts that could degrade the sensitive nitrile group and acts as a drying agent to maintain anhydrous conditions essential for the cyanuric chloride mechanism. Furthermore, the process integrates a strategic deprotection and salt-formation step using p-toluenesulfonic acid. By converting the free base into a stable p-toluenesulfonate salt prior to the final acylation, the process effectively precipitates out soluble impurities that would otherwise persist. This intermediate isolation step is the key differentiator, ensuring that only high-purity material enters the final chloroacetylation stage, thereby drastically reducing the burden on the final crystallization and preserving yield.
Mechanistic Insights into Stabilized Cyanuric Chloride Dehydration
To fully appreciate the technical superiority of this route, one must dissect the catalytic and stabilization mechanisms employed. The process initiates with the Boc-protection of L-prolinamide, facilitated by a quaternary ammonium salt phase transfer catalyst (e.g., benzyltriethylammonium chloride). This catalyst accelerates the interfacial reaction between the organic substrate and the aqueous base (potassium carbonate), ensuring rapid and complete conversion while minimizing the residence time of the sensitive amide in the reaction mixture. Following this, the core innovation occurs during the dehydration step. Cyanuric chloride activates the amide oxygen, facilitating the elimination of water to form the nitrile. However, this reaction generates HCl, which can catalyze side reactions or decompose the product. The introduction of acid carbonate acts as a buffered base, scavenging protons without creating a strongly alkaline environment that might hydrolyze the nitrile. Simultaneously, the anhydrous sulfate sequesters trace moisture, driving the equilibrium towards the nitrile product and preventing the hydrolysis of cyanuric chloride itself.
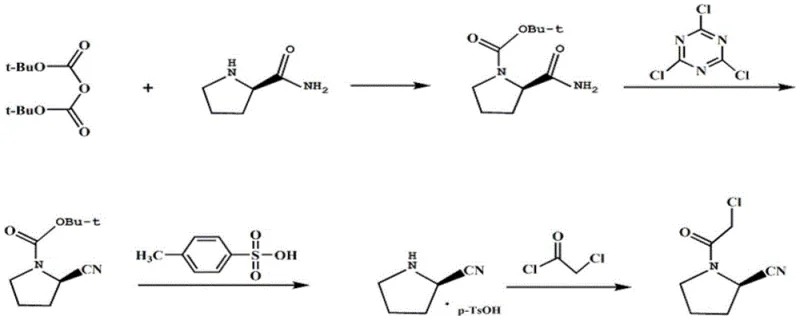
The subsequent purification mechanism relies on the differential solubility of the p-toluenesulfonate salt. After the dehydration is complete, the solvent is swapped, and p-toluenesulfonic acid is introduced under reflux. This converts the crude (S)-pyrrolidine-2-carbonitrile into its corresponding salt, which crystallizes out of the acetonitrile solution upon cooling. Crucially, neutral and basic impurities, including unreacted starting materials and the troublesome formamide byproducts, remain in the mother liquor. This solid-liquid separation acts as a powerful purification gate, removing the majority of the impurity load before the final step. Finally, the regenerated free base undergoes chloroacetylation with chloroacetyl chloride in the presence of triethylamine. The use of a weak polar solvent like n-hexane or methyl tert-butyl ether for the final crystallization exploits the low solubility of the target chloroacetyl derivative, allowing it to precipitate in high purity while leaving polar impurities in solution. This multi-layered approach to impurity control is what enables the achievement of 99.7% purity.
How to Synthesize (S)-1-(2-chloroacetyl)pyrrolidine-2-carbonitrile Efficiently
Implementing this synthetic route requires precise control over reaction parameters, particularly temperature and stoichiometry, to replicate the high yields reported in the patent literature. The process is designed to be operationally simple, avoiding cryogenic conditions or high-pressure equipment, which facilitates easy technology transfer from the laboratory to the pilot plant. Operators must pay close attention to the addition rate of cyanuric chloride and the timing of the stabilizer addition to prevent exotherms that could degrade the product. The following guide outlines the standardized operational framework derived from the patent examples, serving as a baseline for process optimization and scale-up activities.
- Perform Boc protection on L-prolinamide using di-tert-butyl dicarbonate and a quaternary ammonium salt catalyst in dichloromethane.
- Execute dehydration cyanation in pure DMF using cyanuric chloride, stabilized by acid carbonate and anhydrous sulfate, followed by p-toluenesulfonic acid salt formation.
- Conduct final chloroacetylation with chloroacetyl chloride in dichloromethane/triethylamine, followed by crystallization in a weak polar solvent.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic methodology offers tangible strategic benefits beyond mere technical elegance. The primary value driver is the significant reduction in raw material costs achieved by substituting expensive dehydrating agents like trifluoroacetic anhydride with commodity chemicals like cyanuric chloride and sodium bicarbonate. This substitution not only lowers the direct bill of materials but also simplifies the procurement landscape by relying on widely available bulk chemicals rather than specialized reagents subject to supply volatility. Furthermore, the enhanced stability of the reaction system reduces the risk of batch failures, a critical factor in maintaining supply continuity for high-demand diabetes medications. By minimizing the generation of hard-to-remove impurities at the source, the process eliminates the need for repetitive recrystallizations or chromatographic purifications, which are time-consuming and resource-intensive operations.
- Cost Reduction in Manufacturing: The economic efficiency of this process is driven by the elimination of costly reagents and the optimization of yield. By avoiding the use of trifluoroacetic anhydride, manufacturers can realize substantial savings on raw material expenditures. Additionally, the high yield of over 80% implies that less starting material is required to produce the same amount of finished goods, effectively lowering the cost per kilogram. The simplified workup procedures, which rely on standard filtration and distillation rather than complex separations, further reduce utility consumption and labor costs, contributing to a leaner manufacturing model.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the chemical process. The mild reaction conditions (10-25°C) reduce the dependency on specialized heating or cooling infrastructure, allowing for production in a wider range of facilities. The use of common solvents such as dichloromethane, DMF, and acetonitrile ensures that solvent supply chains remain stable and predictable. Moreover, the intermediate salt formation step provides a stable storage form of the chiral nitrile, allowing manufacturers to decouple the upstream synthesis from the final acylation step, thereby offering greater flexibility in production scheduling and inventory management.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this route is inherently greener and safer. The replacement of harsh fluorinated reagents reduces the generation of hazardous fluoride waste, simplifying effluent treatment and lowering disposal costs. The process is demonstrated to be scalable, with examples showing successful translation from gram to multi-kilogram scales without loss of efficiency. The high purity of the final product reduces the solvent load associated with purification, aligning with modern green chemistry principles and helping pharmaceutical partners meet their sustainability goals while ensuring regulatory compliance for impurity limits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy processes in terms of purity, yield, and operational safety.
Q: How does the new process improve yield compared to conventional cyanuric chloride methods?
A: The novel method introduces acid carbonate and anhydrous sulfate during the dehydration step in pure DMF. This addition stabilizes the reaction system, prevents material degradation, and ensures complete conversion, thereby significantly boosting overall yield to over 80%.
Q: What is the primary advantage of forming the p-toluenesulfonate salt intermediate?
A: Forming the (S)-pyrrolidine-2-carbonitrile p-toluenesulfonate salt acts as a critical purification checkpoint. It allows for the effective removal of difficult-to-separate impurities generated during the dehydration phase before the final acylation, ensuring the final API intermediate reaches 99.7% purity.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process utilizes mild reaction conditions (10-25°C) and avoids expensive or hazardous reagents like trifluoroacetic anhydride. The use of common solvents like DCM and DMF, combined with robust crystallization steps, makes it highly scalable and safe for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vildagliptin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceuticals like Vildagliptin depends on the reliability and quality of the supply chain. Our technical team has thoroughly analyzed the synthetic pathway described in CN112028806B and possesses the expertise to execute this optimized route with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our partners receive a consistent supply of high-purity intermediates regardless of market demand fluctuations. Our facility is equipped with rigorous QC labs capable of detecting trace impurities at ppm levels, guaranteeing that every batch meets stringent purity specifications required for GMP manufacturing of the final API.
We invite global pharmaceutical companies and contract manufacturers to collaborate with us to leverage this advanced technology for their supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this optimized route can improve your margins. We encourage you to contact our technical procurement team today to request specific COA data from our pilot batches and to discuss detailed route feasibility assessments for your upcoming projects. Let us be your strategic partner in delivering high-quality antidiabetic solutions to the global market.
