Advanced Manufacturing of Chiral 5-Substituted Proline Compounds for Global Pharmaceutical Supply Chains
Introduction to Patent CN112679407B: A Breakthrough in Chiral Proline Synthesis
The pharmaceutical industry continuously demands efficient, scalable, and cost-effective routes for synthesizing chiral building blocks, particularly for treating chronic conditions such as diabetes, epilepsy, and hypertension. Patent CN112679407B introduces a highly optimized preparation method for chiral 5-substituted proline compounds, addressing critical bottlenecks in traditional synthetic pathways. This technology leverages a concise four-step sequence comprising a Grignard reaction, a stereoselective reduction, an acylation, and a final hydrolysis. Unlike conventional methods that often rely on expensive chiral auxiliaries or difficult separation processes, this novel approach utilizes the inherent stereoelectronic properties of the intermediates to drive high diastereoselectivity. The process is characterized by mild reaction conditions, ranging from cryogenic temperatures of -40°C for the initial addition to moderate heating at 60-80°C for hydrolysis, ensuring operational safety and ease of control. For global procurement teams and R&D directors, this patent represents a significant opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials with reduced lead times and lower environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-substituted proline derivatives has been plagued by inefficiencies that hinder commercial viability. Traditional routes often involve multi-step sequences with poor atom economy, requiring harsh reagents that generate significant hazardous waste. A major痛点 (pain point) in legacy processes is the reliance on transition metal catalysts for asymmetric hydrogenation or coupling, which introduces the risk of heavy metal contamination—a critical failure mode for API intermediates intended for human consumption. Furthermore, achieving the desired (2R,5R) or (2R,5S) configuration frequently necessitates chiral resolution via crystallization or preparative HPLC, which drastically cuts overall yields and inflates production costs. These methods are often difficult to scale beyond the laboratory bench due to exothermic risks and the complexity of removing trace metal impurities to meet stringent regulatory standards. Consequently, supply chains for these key intermediates have remained fragile and expensive, limiting the speed at which new drug candidates can reach clinical trials.
The Novel Approach
The methodology disclosed in CN112679407B offers a transformative solution by streamlining the synthesis into a robust, four-step linear progression that avoids these pitfalls. The process initiates with a Grignard reaction on an N-Boc protected pyroglutamic acid ester, followed immediately by an in-situ deprotection to generate a reactive imine species. This strategy bypasses the isolation of unstable intermediates, enhancing throughput. The core innovation lies in the subsequent reduction step, where a simple hydride source induces the formation of the chiral center with high fidelity, driven by the dominant conformation of the cyclic system. 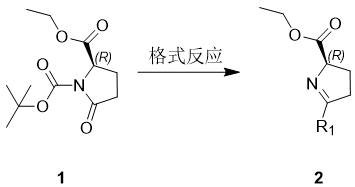 By eliminating the need for exotic chiral catalysts and utilizing commodity chemicals like sodium borohydride, this route achieves cost reduction in pharmaceutical intermediate manufacturing that is both immediate and substantial. The simplicity of the workup procedures—primarily involving extraction, drying, and concentration—further underscores the industrial applicability of this method, making it an ideal candidate for rapid commercial scale-up of complex pharmaceutical intermediates.
By eliminating the need for exotic chiral catalysts and utilizing commodity chemicals like sodium borohydride, this route achieves cost reduction in pharmaceutical intermediate manufacturing that is both immediate and substantial. The simplicity of the workup procedures—primarily involving extraction, drying, and concentration—further underscores the industrial applicability of this method, making it an ideal candidate for rapid commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Stereoselective Reduction and Cyclization
From a mechanistic perspective, the success of this synthesis hinges on the precise control of stereochemistry during the reduction phase. In Step S2, the imine intermediate generated from the Grignard adduct undergoes reduction using sodium borohydride in an alcoholic solvent, typically ethanol. The cyclic constraint of the pyrrolidine ring imposes significant steric bias, forcing the hydride attack to occur from the less hindered face. This results in the preferential formation of the cis-isomer, where the 5-substituent and the 2-carboxyl group adopt a specific spatial relationship. 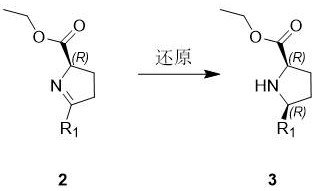 The patent data indicates that this reduction is not merely a functional group transformation but a stereo-defining event that locks the molecule into the desired (R,R) or (R,S) configuration without the need for external chiral induction. Following reduction, the free amine is immediately protected via acylation (Step S3) using Boc anhydride and triethylamine. This acylation serves a dual purpose: it stabilizes the sensitive amine against oxidation and prepares the molecule for the final hydrolysis. The final step involves saponification of the ethyl ester using lithium hydroxide, a reagent chosen for its solubility profile and mild basicity, which cleaves the ester bond without racemizing the sensitive alpha-chiral center. This mechanistic elegance ensures that the final product, such as (2R,5R)-N-Boc-5-methylproline, is obtained with purities exceeding 97%, as demonstrated in the patent examples.
The patent data indicates that this reduction is not merely a functional group transformation but a stereo-defining event that locks the molecule into the desired (R,R) or (R,S) configuration without the need for external chiral induction. Following reduction, the free amine is immediately protected via acylation (Step S3) using Boc anhydride and triethylamine. This acylation serves a dual purpose: it stabilizes the sensitive amine against oxidation and prepares the molecule for the final hydrolysis. The final step involves saponification of the ethyl ester using lithium hydroxide, a reagent chosen for its solubility profile and mild basicity, which cleaves the ester bond without racemizing the sensitive alpha-chiral center. This mechanistic elegance ensures that the final product, such as (2R,5R)-N-Boc-5-methylproline, is obtained with purities exceeding 97%, as demonstrated in the patent examples.
How to Synthesize Chiral 5-Substituted Proline Efficiently
Implementing this synthesis requires strict adherence to temperature controls and reagent stoichiometry to maximize the diastereomeric ratio. The process begins with the careful addition of the Grignard reagent at -40°C to prevent side reactions, followed by a controlled warm-up to -20°C to drive the reaction to completion over 12 hours. The subsequent reduction must be performed with vigorous stirring to ensure homogeneous mixing of the hydride source, which is critical for consistent stereochemical outcomes. While the patent provides specific molar ratios and solvent volumes, scaling this process requires attention to heat dissipation during the exothermic Grignard and reduction steps. The detailed standardized synthetic steps, including specific quenching protocols and purification parameters, are outlined below to guide process chemists in replicating this high-yielding route.
- Perform a Grignard reaction on N-Boc-D-pyroglutamic acid ethyl ester at -40°C followed by deprotection to form an imine intermediate.
- Execute a stereoselective reduction using sodium borohydride in ethanol to establish the cis-configuration with high diastereomeric excess.
- Conduct an acylation reaction using Boc anhydride and triethylamine to protect the amine functionality.
- Finalize the synthesis via alkaline hydrolysis using lithium hydroxide to yield the target carboxylic acid compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling strategic advantages that extend beyond mere technical feasibility. The shift away from precious metal catalysts to base-metal-free hydride reductions fundamentally alters the cost structure of the intermediate, removing the volatility associated with rhodium or palladium pricing. Moreover, the use of widely available starting materials like N-Boc-D-pyroglutamic acid ethyl ester ensures a stable supply base, reducing the risk of raw material shortages that often plague specialized chemical markets. The operational simplicity of the process, characterized by standard unit operations like filtration and liquid-liquid extraction, means that existing multipurpose reactors can be utilized without significant capital expenditure on new equipment. This flexibility allows for agile response to market demand fluctuations, ensuring continuity of supply for downstream API manufacturers.
- Cost Reduction in Manufacturing: The elimination of expensive chiral catalysts and the avoidance of low-yield resolution steps result in a drastically simplified cost model. By utilizing sodium borohydride, a low-cost reducing agent, instead of complex asymmetric hydrogenation systems, the direct material costs are significantly lowered. Furthermore, the high total yield reported (over 70% in examples) minimizes waste disposal costs and maximizes the output per batch, driving down the cost per kilogram of the final active pharmaceutical ingredient precursor.
- Enhanced Supply Chain Reliability: The reliance on commodity reagents such as ethanol, tetrahydrofuran, and lithium hydroxide mitigates supply chain risks associated with specialty chemicals. These materials are produced globally in massive quantities, ensuring that production schedules are not disrupted by vendor-specific bottlenecks. Additionally, the robustness of the reaction conditions allows for longer campaign runs and easier technology transfer between manufacturing sites, providing multinational clients with a diversified and resilient sourcing strategy for their critical proline intermediates.
- Scalability and Environmental Compliance: The process is inherently green by design, avoiding the generation of heavy metal waste streams that require costly remediation. The mild reaction temperatures and the use of recyclable solvents align with modern sustainability goals, facilitating easier regulatory approval in environmentally stringent jurisdictions. The straightforward workup procedures facilitate easy scale-up from pilot plant to commercial tonnage, ensuring that the supply can grow in lockstep with the clinical and commercial success of the downstream drug products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral proline synthesis technology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on purity profiles, scalability limits, and regulatory considerations. Understanding these nuances is essential for technical teams evaluating this route for integration into their existing manufacturing portfolios.
Q: How is stereocontrol achieved in this proline synthesis route?
A: Stereocontrol is primarily achieved during the reduction step (Step S2). The reduction of the imine intermediate using sodium borohydride generates a dominant cis-conformation due to steric hindrance and thermodynamic stability, ensuring high optical purity without the need for complex chiral chromatography.
Q: What are the cost advantages of this method compared to traditional routes?
A: This method eliminates the need for expensive transition metal catalysts and complex resolution steps. By utilizing commodity reagents like sodium borohydride and standard Grignard reagents, the process significantly reduces raw material costs and simplifies downstream purification, leading to substantial overall cost reduction in API manufacturing.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for scalability. The reaction conditions are mild (ranging from -40°C to 80°C), and the workup procedures involve standard extractions and filtrations rather than sensitive column chromatography. The patent reports a total yield of over 70% in pilot examples, indicating robustness for kilogram-to-ton scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 5-Substituted Proline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Whether you require custom synthesis of specific 5-substituted proline derivatives or optimization of existing routes for cost efficiency, our technical expertise ensures that your supply chain remains robust and compliant with global regulatory standards.
We invite you to engage with our technical procurement team to discuss how this patented technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this streamlined synthesis. We encourage potential partners to contact us directly to obtain specific COA data for our reference standards and to receive comprehensive route feasibility assessments that will accelerate your drug development timeline.
