Scalable Synthesis of Telaprevir Intermediate via Mild Cyanation and Chiral Resolution
Introduction to Advanced Telaprevir Intermediate Manufacturing
The global demand for direct-acting antiviral agents, particularly for the treatment of chronic hepatitis C, has necessitated the development of robust and scalable synthetic routes for key pharmaceutical intermediates. Patent CN103450066A discloses a highly efficient preparation method for a cyclopenta[c]pyrrole compound, specifically Formula I, which serves as a critical chiral building block for the synthesis of Telaprevir (VX-960). This technology represents a significant leap forward in process chemistry by replacing complex, low-yielding sequences with a streamlined approach that begins with the readily available 3-azabicyclo[3.3.0]octane. By integrating oxidation, cyanation, and a highly selective chiral resolution step, this method addresses the longstanding challenges of racemization and harsh reaction conditions that have historically plagued the manufacturing of this scaffold. For pharmaceutical manufacturers, adopting this pathway offers a strategic advantage in securing a stable supply of high-purity intermediates essential for the production of next-generation protease inhibitors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
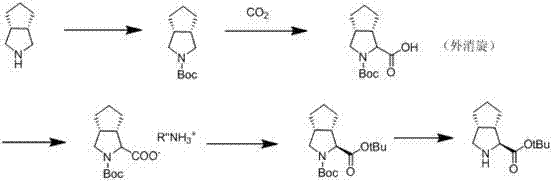
Historically, the synthesis of the pentamethylene[c]pyrrole core found in Telaprevir has been fraught with significant technical and economic hurdles. Prior art, such as the methods disclosed in CN101291909, often relies on extremely harsh reaction conditions, including the requirement for cryogenic temperatures as low as -78°C and strict nitrogen protection atmospheres. These conditions not only demand specialized and expensive reactor infrastructure but also introduce severe safety risks and operational complexities that are difficult to manage on a multi-ton scale. Furthermore, alternative biological routes described in documents like WO2010008828 depend on the fermentation of specific enzymes, which are commercially available only at exorbitant prices, thereby inflating the cost of goods sold (COGS) and creating supply chain vulnerabilities. Additionally, many traditional chemical routes suffer from chiral instability, leading to racemization of the critical stereocenters and necessitating multiple, yield-depleting purification steps via silica gel column chromatography.
The Novel Approach
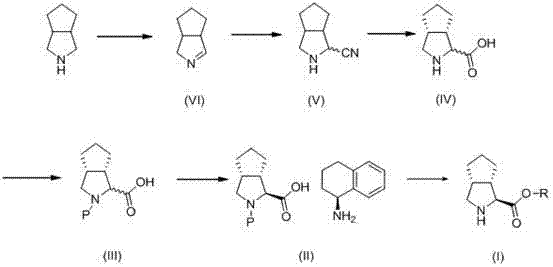
In stark contrast to these legacy methods, the novel approach detailed in CN103450066A utilizes a chemically elegant sequence that operates under remarkably mild conditions, typically ranging from 0°C to 50°C. The process initiates with the oxidation of 3-azabicyclo[3.3.0]octane to an enamine intermediate, followed by a safe cyanation step using trimethylsilyl cyanide (TMSCN) rather than hazardous hydrogen cyanide gas. This strategic substitution eliminates the need for specialized gas handling equipment and significantly enhances operator safety. The subsequent hydrolysis and protection steps are designed to maintain the integrity of the chiral centers, avoiding the racemization issues common in earlier syntheses. By focusing on solution-phase chemistry with high-yielding transformations, this method drastically simplifies the workup procedures, removing the need for labor-intensive column chromatography and enabling a direct path to industrial viability.
Mechanistic Insights into Chiral Resolution and Cyanation
The core of this synthetic strategy lies in its precise control over stereochemistry during the formation of the cyclopenta[c]pyrrole ring system. The cyanation step, utilizing TMSCN in a protic solvent mixture such as methanol and dichloromethane, proceeds through a mechanism that favors the formation of the desired nitrile without compromising the adjacent stereocenters. Unlike radical-based cyanation methods that might lead to scrambling of chirality, this ionic pathway ensures high fidelity in the molecular architecture. Following hydrolysis to the carboxylic acid (Formula IV), the process employs a classical yet highly effective diastereomeric salt formation technique. By reacting the racemic protected acid with an optically active amine, such as (S)-1,2,3,4-tetrahydronaphthylamine or S(-)-α-phenylethylamine, the desired enantiomer is selectively crystallized out of the solution. This resolution step is critical, as it effectively purifies the material to an ee value greater than 99%, ensuring that the downstream coupling reactions for Telaprevir assembly proceed with minimal impurity burden.
Furthermore, the protection group strategy employed in this route is specifically tailored to facilitate easy removal in the final stages without affecting the sensitive ester functionality. Whether using tert-butyloxycarbonyl (Boc) or benzyloxycarbonyl (Cbz) groups, the chemistry is optimized to withstand the acidic or basic conditions of the preceding hydrolysis and resolution steps. The final esterification is conducted under mild conditions using coupling agents like DCC and DMAP, which prevents epimerization at the alpha-carbon. This meticulous attention to mechanistic detail ensures that the final product, Formula I, meets the stringent purity specifications required for GMP manufacturing of antiviral APIs. The ability to achieve HPLC purity of 99.9% directly from the crystallization steps underscores the robustness of this chemical design.
How to Synthesize Telaprevir Intermediate Efficiently
The synthesis of this high-value intermediate is structured around a logical sequence of transformations that maximize atom economy and minimize waste generation. The process begins with the conversion of the bicyclic amine starting material into an enamine, followed by the introduction of the cyano group which serves as the precursor for the carboxylic acid moiety. The subsequent resolution step is the pivotal point where optical purity is established, leveraging the differential solubility of diastereomeric salts. For process chemists looking to implement this technology, the protocol emphasizes the importance of temperature control during the cyanation and esterification phases to prevent side reactions. The detailed standardized synthetic steps for this efficient route are outlined in the guide below.
- Oxidize 3-azabicyclo[3.3.0]octane using hypohalite followed by dehydrohalogenation under alkaline conditions to form the enamine intermediate (Formula VI).
- Perform cyanation on Formula VI using trimethylsilyl cyanide (TMSCN) in a protic solvent to yield the nitrile compound (Formula V), avoiding hazardous HCN gas.
- Hydrolyze the nitrile to the carboxylic acid (Formula IV), protect the amine (e.g., Boc), and resolve the racemate using a chiral amine like (S)-1,2,3,4-tetrahydronaphthylamine to obtain high-purity Formula II.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers profound advantages in terms of cost stability and operational reliability. By eliminating the dependency on cryogenic cooling systems and expensive biocatalysts, the manufacturing process becomes significantly less capital intensive and more energy efficient. The use of commodity chemicals like TMSCN and sodium hypochlorite, rather than specialized reagents, insulates the production cost from volatile market fluctuations associated with niche biochemicals. Moreover, the high yields reported in the patent examples, such as 97.5% in the initial oxidation step and over 90% in the final esterification, translate directly into reduced raw material consumption per kilogram of finished product. This efficiency gain is a critical driver for lowering the overall cost of goods, allowing for more competitive pricing in the global pharmaceutical market.
- Cost Reduction in Manufacturing: The elimination of extreme low-temperature requirements (-78°C) removes the need for specialized cryogenic reactors and the substantial energy costs associated with maintaining such conditions. Additionally, replacing expensive enzymatic processes with robust chemical catalysis using affordable reagents like TMSCN drastically reduces the variable cost per batch. The high overall yield of the process minimizes the loss of valuable chiral materials, ensuring that the maximum amount of starting material is converted into saleable product, thereby optimizing the return on investment for large-scale production campaigns.
- Enhanced Supply Chain Reliability: The reliance on 3-azabicyclo[3.3.0]octane as a starting material provides a strong foundation for supply continuity, as this feedstock is widely available from multiple chemical suppliers globally. Unlike fermentation-based routes that are susceptible to biological variability and long lead times for enzyme production, this purely chemical synthesis offers predictable cycle times and consistent output quality. The robustness of the reaction conditions means that production is less likely to be interrupted by minor deviations in process parameters, ensuring a steady flow of intermediates to downstream API manufacturing sites.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard unit operations such as extraction, crystallization, and filtration that are easily transferable from pilot plant to commercial manufacturing suites. The avoidance of hazardous gases like HCN and the reduction of solvent-intensive chromatography steps significantly lower the environmental footprint of the manufacturing process. This alignment with green chemistry principles not only simplifies regulatory compliance regarding waste disposal but also enhances the sustainability profile of the supply chain, a key metric for modern pharmaceutical procurement strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical benefits of this novel route. Understanding these specifics is vital for technical teams evaluating the feasibility of integrating this intermediate into their existing production portfolios.
Q: How does this novel synthesis route improve upon previous methods for Telaprevir intermediates?
A: Unlike prior art methods that require harsh cryogenic conditions (-78°C) or expensive biocatalysts, this patent utilizes mild reaction temperatures (0-50°C) and readily available chemical reagents like TMSCN, significantly simplifying the operational complexity and reducing equipment costs for industrial scale-up.
Q: What is the expected optical purity of the intermediate produced via this method?
A: The process employs a robust chiral resolution step using optically active amines such as (S)-1,2,3,4-tetrahydronaphthylamine, consistently achieving an ee value greater than 99% and HPLC purity of 99.9%, which is critical for the downstream synthesis of the antiviral API.
Q: Is the starting material 3-azabicyclo[3.3.0]octane commercially viable for large-scale production?
A: Yes, 3-azabicyclo[3.3.0]octane is a stable and accessible feedstock. The synthetic route converts this raw material efficiently through oxidation and cyanation steps with high yields (e.g., 97.5% in the first step), ensuring a reliable supply chain without the bottlenecks associated with fermentation-based enzymatic processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Telaprevir Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the timely delivery of life-saving antiviral medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of Telaprevir intermediate meets the exacting standards required by global regulatory bodies. Our facility is equipped to handle the specific chemical transformations outlined in this patent, including the safe handling of cyanation reagents and the precise execution of chiral resolution protocols.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this more efficient manufacturing process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will strengthen your supply chain and reduce your overall production costs.
