Revolutionizing 2-Amino-6-Nitrobenzothiazole Production with Green Ionic Liquid Technology for Global Markets
Introduction to Advanced Green Synthesis Technologies
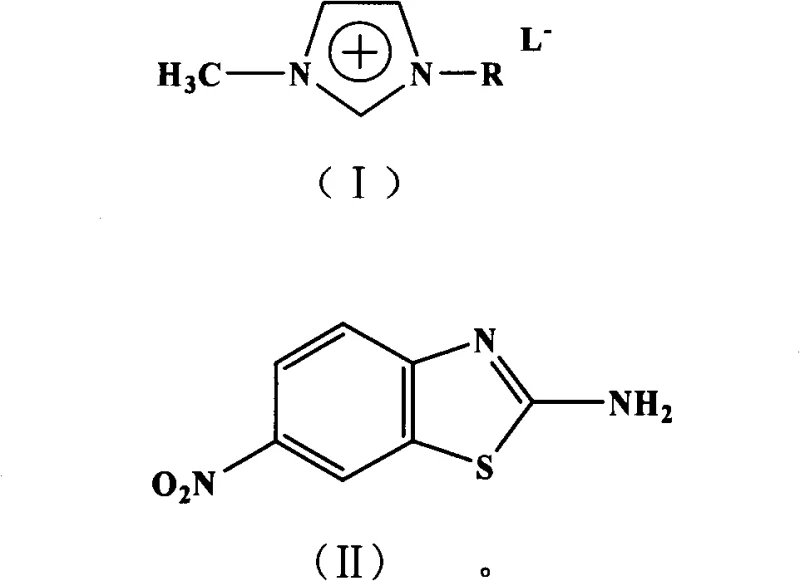
The global demand for high-performance azo dyes and pharmaceutical intermediates necessitates a shift towards more sustainable and efficient manufacturing processes. Patent CN101906083B introduces a groundbreaking methodology for the preparation of 2-amino-6-nitrobenzothiazole, a critical building block in the synthesis of heterocyclic disperse dyes such as Disperse Red 145 and 177. This innovation leverages the unique properties of acidic ionic liquids, which function simultaneously as the reaction medium and the catalyst, thereby streamlining the synthetic route. By replacing traditional harsh acidic conditions with tunable ionic systems, this technology addresses significant environmental and operational challenges faced by modern chemical manufacturers. The process utilizes p-nitroaniline and ammonium thiocyanate as starting materials, undergoing a one-pot reaction that minimizes unit operations and maximizes atom economy. For R&D directors and procurement specialists seeking a reliable dye intermediate supplier, understanding the mechanistic advantages of this patent is crucial for evaluating long-term supply chain stability and cost efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 2-amino-6-nitrobenzothiazole has relied on methodologies that are increasingly untenable in the context of modern environmental regulations and cost pressures. Traditional routes often involve multi-step sequences, such as the thiocyanation of aniline followed by oxidative cyclization, acetylation, nitration, and hydrolysis, or direct nitration in concentrated sulfuric acid. These conventional processes generate substantial quantities of waste acid, posing severe disposal challenges and increasing the environmental footprint of the facility. Furthermore, the use of concentrated sulfuric acid leads to significant equipment corrosion, necessitating expensive maintenance schedules and specialized reactor materials. The separation of products from strong acid media is also energy-intensive and complex, often requiring extensive neutralization and washing steps that dilute wastewater streams. For a procurement manager focused on cost reduction in fine chemical manufacturing, these hidden costs associated with waste treatment and equipment degradation represent a significant burden on the overall cost of goods sold (COGS).
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes acidic ionic liquids to create a benign and highly efficient reaction environment. This method allows for the direct conversion of p-nitroaniline and ammonium thiocyanate into the target benzothiazole derivative through a streamlined one-pot procedure. The ionic liquid not only dissolves the reactants effectively due to its wide liquid temperature range and good solubility characteristics but also provides the necessary acidic protons or Lewis acid sites to catalyze the cyclization. This eliminates the need for external strong mineral acids, drastically reducing the generation of hazardous waste. The mild reaction conditions, typically ranging from 80°C to 110°C, further enhance safety profiles and reduce energy consumption compared to high-temperature nitration processes. By integrating the solvent and catalyst functions, this technology simplifies the workflow, making it an ideal candidate for the commercial scale-up of complex dye intermediates while adhering to green chemistry principles.
Mechanistic Insights into Acidic Ionic Liquid Catalysis
The efficacy of this synthesis relies heavily on the dual functionality of the acidic ionic liquid, which acts as both a solvent and a Brønsted or Lewis acid catalyst. The cationic component, typically an imidazolium derivative substituted with sulfonic acid groups, provides a structured environment that stabilizes transition states during the formation of the p-nitrophenylthiourea intermediate. Subsequently, the acidic nature of the ionic liquid promotes the intramolecular cyclization and dehydration steps required to form the benzothiazole ring. Unlike homogeneous mineral acids that are difficult to separate, the ionic liquid forms a distinct phase or can be easily separated from the organic product due to its negligible vapor pressure and immiscibility with certain organic extraction solvents like toluene. This phase behavior is critical for maintaining high selectivity and preventing side reactions that often plague traditional acid-catalyzed nitrations. The tunability of the ionic liquid's acidity allows chemists to optimize the reaction rate without compromising product quality, ensuring a consistent impurity profile that meets stringent pharmaceutical and dye industry standards.
Controlling the impurity profile is paramount for applications in high-value sectors such as pharmaceuticals and advanced materials. The use of ionic liquids minimizes the formation of oxidation by-products and polymeric tars that are common in sulfuric acid-mediated reactions. The mild acidity prevents the over-nitration or degradation of the sensitive amino group, which is a frequent issue in conventional nitration protocols. Furthermore, the ability to recycle the ionic liquid means that the catalytic environment remains consistent across batches, leading to reproducible quality. For R&D teams, this consistency reduces the need for extensive purification steps downstream, such as column chromatography or repeated recrystallizations. The structural integrity of the final product, confirmed by melting points matching literature values (252-253°C), demonstrates that the ionic liquid method preserves the chemical fidelity of the molecule better than harsher traditional methods.
How to Synthesize 2-Amino-6-Nitrobenzothiazole Efficiently
The practical implementation of this synthesis involves a straightforward protocol that balances reaction kinetics with ease of isolation. The process begins by charging p-nitroaniline and ammonium thiocyanate into the acidic ionic liquid medium, ensuring a molar ratio that favors complete conversion while minimizing excess reagent waste. The reaction is then heated to a moderate temperature range, typically between 80°C and 110°C, and maintained for a duration of 1 to 5 hours depending on the specific ionic liquid anion used. Monitoring via TLC ensures that the reaction proceeds to completion before moving to the workup phase. The detailed standardized synthesis steps, including specific workup procedures and purification parameters, are outlined below to assist technical teams in replicating this high-efficiency route.
- Charge p-nitroaniline and ammonium thiocyanate into an acidic ionic liquid medium (such as imidazolium-based salts) with a molar ratio of 1: 1 to 1:3.
- Maintain the reaction temperature between 80°C and 110°C for 1 to 5 hours to facilitate the formation of p-nitrophenylthiourea and subsequent cyclization.
- Extract the reaction mixture with toluene, wash with distilled water, dry over anhydrous magnesium sulfate, and remove solvent under reduced pressure to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ionic liquid technology translates into tangible strategic advantages beyond mere technical novelty. The elimination of concentrated sulfuric acid from the process flow removes a major hazard from the facility, reducing insurance premiums and safety compliance costs. Moreover, the recyclability of the ionic liquid catalyst represents a significant shift in material economics; instead of consuming stoichiometric or excess amounts of acid that become waste, the catalyst is retained and reused, lowering the recurring cost of raw materials. This closed-loop system aligns perfectly with corporate sustainability goals and reduces the regulatory burden associated with hazardous waste discharge. The simplified post-processing, involving simple extraction and distillation, shortens the batch cycle time, allowing for increased throughput without expanding physical plant capacity. These factors collectively contribute to a more resilient and cost-effective supply chain for high-purity dye intermediates.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of expensive waste treatment protocols associated with spent sulfuric acid. By utilizing a reusable ionic liquid, the facility avoids the continuous purchase of fresh acid and the costly neutralization of acidic effluent. Additionally, the reduction in equipment corrosion extends the lifespan of reactors and piping, deferring capital expenditure on replacements. The high yield reported in the patent examples (around 88-89%) ensures that raw material utilization is maximized, further driving down the unit cost of the final API intermediate. These qualitative efficiencies compound over time to deliver substantial cost savings without compromising on product quality.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the volatility of raw material markets and regulatory restrictions on hazardous chemicals. This technology mitigates those risks by relying on stable, commercially available starting materials like p-nitroaniline and ammonium thiocyanate. The robustness of the ionic liquid system means that production is less susceptible to fluctuations in utility costs or minor variations in operating conditions. Furthermore, the simplified logistics of handling non-corrosive ionic liquids compared to bulk sulfuric acid reduces transportation and storage complexities. This reliability ensures that downstream customers, such as dye manufacturers, receive their orders on schedule, fostering stronger long-term partnerships and reducing the risk of production stoppages due to supply shortages.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to industrial production often encounters bottlenecks related to heat transfer and mixing, especially in viscous or heterogeneous systems. The liquid nature of the ionic medium at reaction temperatures facilitates excellent heat and mass transfer, making the scale-up of complex dye intermediates more predictable and safer. From an environmental perspective, the drastic reduction in "three wastes" (waste gas, waste water, and waste residue) positions this method favorably against increasingly strict global environmental regulations. Facilities adopting this technology can operate with a smaller environmental footprint, securing their social license to operate and avoiding potential fines or shutdowns associated with non-compliance. This future-proofs the manufacturing asset against tightening ecological standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ionic liquid-based synthesis. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this green chemistry approach into existing production lines. The answers reflect the specific advantages of using acidic ionic liquids over traditional mineral acid catalysts in the context of benzothiazole derivative synthesis.
Q: What are the primary advantages of using acidic ionic liquids over concentrated sulfuric acid in this synthesis?
A: Acidic ionic liquids serve a dual role as both solvent and catalyst, eliminating the need for large volumes of corrosive concentrated sulfuric acid. This significantly reduces waste acid generation, lowers equipment corrosion risks, and simplifies the post-reaction separation process compared to traditional nitration methods.
Q: Can the ionic liquid catalyst be recovered and reused in this process?
A: Yes, one of the key features of this technology is the recyclability of the ionic liquid. After the product is extracted with organic solvents like toluene, the ionic liquid phase remains and can be reused for subsequent batches, contributing to substantial long-term cost reductions and environmental compliance.
Q: What is the typical purity and yield achievable with this method?
A: According to the patent data, this method consistently achieves yields around 88% to 89% with a product content (purity) of ≥98%. The melting point matches literature values (252-253°C), indicating high structural integrity suitable for downstream dye synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-6-Nitrobenzothiazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green catalytic technologies like the one described in patent CN101906083B for the production of high-value intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the exacting standards required by the global dye and pharmaceutical industries. We understand that transitioning to new synthetic routes requires confidence in both technical capability and supply reliability, which is why we invest heavily in process optimization and capacity expansion.
We invite you to collaborate with us to leverage these advanced manufacturing capabilities for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this ionic liquid-based route for your supply chain. Please contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your volume requirements. By partnering with us, you secure not just a product, but a strategic advantage through superior process technology and dedicated support.
