Advanced Synthesis of Halogenated Benzothiepin Oxide for Scalable Pharmaceutical Production
Advanced Synthesis of Halogenated Benzothiepin Oxide for Scalable Pharmaceutical Production
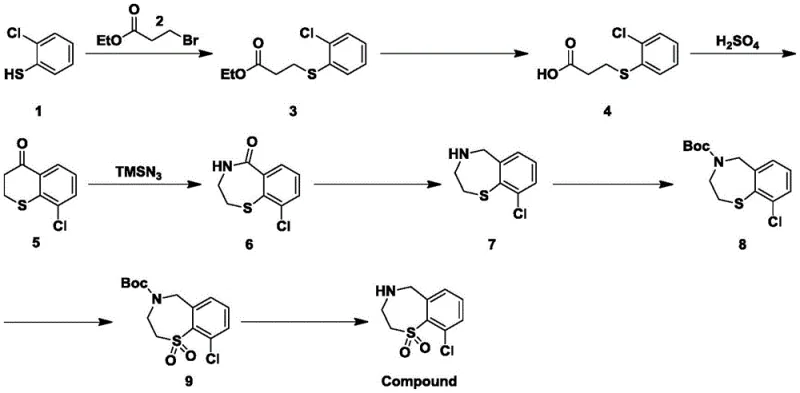
The pharmaceutical industry is constantly seeking robust synthetic routes for complex heterocyclic intermediates that can enhance the efficacy of antiviral medications. Patent CN111574475B discloses a sophisticated preparation method for halogenated benzothiepin oxide, specifically 9-chloro-2,3,4,5-tetrahydro-1,4-benzothiepin-1,1-dioxide, which serves as a critical building block for active components in drugs designed to prevent and treat Respiratory Syncytial Virus (RSV) infections. This technical breakthrough addresses longstanding challenges in heterocyclic chemistry by enabling the precise introduction of polar functional groups onto the benzene ring, a modification that was previously difficult to achieve with high regioselectivity. The disclosed methodology not only streamlines the production of these valuable intermediates but also ensures that the final medicinal products exhibit superior physicochemical properties, particularly regarding water solubility and absorption rates in biological systems. For R&D directors and process chemists, understanding the nuances of this eight-step synthesis is essential for developing next-generation antiviral therapies that meet stringent regulatory standards for purity and performance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing benzothiepin derivatives, such as those described in prior art like CN106414436A, often rely on starting materials that lack specific polar substituents on the aromatic ring. These conventional methods typically generate 2,3,4,5-tetrahydro-1,4-benzothiazepine-1,1-dioxide intermediates which suffer from inherently poor water solubility due to the absence of electron-withdrawing or polar functional groups. This physicochemical limitation translates directly into reduced bioavailability when these intermediates are incorporated into final drug formulations, necessitating higher dosages or complex formulation strategies to achieve therapeutic levels in patients. Furthermore, older synthetic pathways frequently involve harsh reaction conditions, including extreme temperatures and the use of aggressive reagents that can compromise the integrity of sensitive functional groups, leading to lower overall yields and a more complex impurity profile that requires extensive and costly purification efforts to resolve.
The Novel Approach
The innovative strategy outlined in the patent data fundamentally shifts the paradigm by utilizing o-chlorothiophenol as the initial raw material, thereby embedding a chlorine atom at a specific position early in the synthetic sequence. This strategic choice facilitates a series of transformations including thio-compound alkylation, ethyl propionate hydrolysis, and a crucial ring-closing reaction that preserves the halogen functionality throughout the process. By integrating an azide reaction followed by carbonyl reduction and a carefully sequenced oxidation step, the novel approach successfully constructs the seven-membered benzothiepin ring system with the desired 1,1-dioxide oxidation state while maintaining the chlorine substituent. This method not only simplifies the introduction of polar groups but also results in a final intermediate that significantly improves the water solubility of the derived medicament, enhancing its absorption by the human body and ultimately boosting the pharmacological effect against RSV without the need for excessive structural modifications later in the drug development pipeline.
Mechanistic Insights into Azide-Mediated Ring Expansion and Selective Oxidation
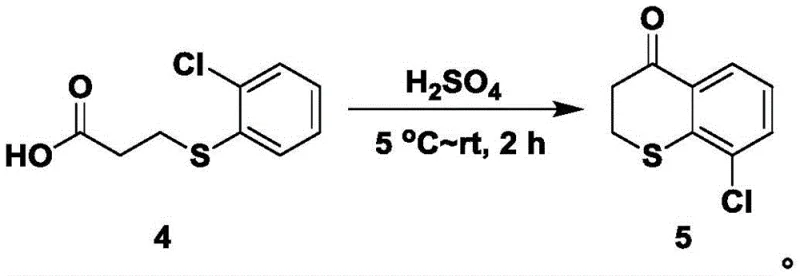
The core of this synthetic innovation lies in the mechanistic elegance of the ring expansion and functionalization steps, particularly the reaction involving trimethylsilyl azide (TMSN3). In step S4, the ketone intermediate, 9-chloro-3,4-dihydro-2H-1-benzothiopyran-4-one, undergoes a Schmidt-type rearrangement where the azide species attacks the carbonyl carbon, leading to the insertion of a nitrogen atom and the expansion of the six-membered ring to a seven-membered 1,4-thiazepine structure. This transformation is conducted in trifluoroacetic acid at room temperature, a condition that promotes the formation of the acyl azide intermediate and its subsequent rearrangement to the lactam with high efficiency. The presence of the chlorine atom on the aromatic ring does not hinder this process; rather, it stabilizes the transition state through electronic effects, ensuring that the nitrogen is inserted regioselectively to form the 9-chloro-3,4-dihydrobenzo-1,4-thiazepin-5(2H)-one scaffold which is pivotal for the subsequent biological activity.
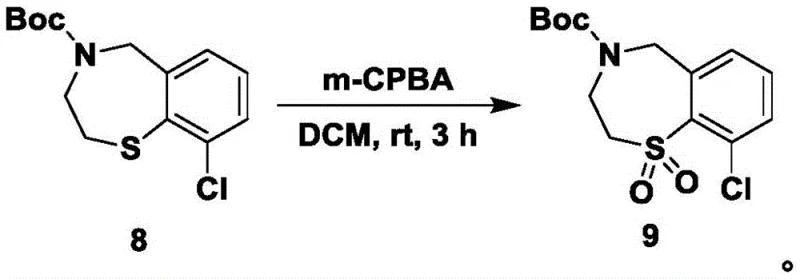
Following the ring expansion, the synthesis employs a highly chemoselective oxidation strategy in step S7 to convert the sulfide moiety into a sulfone (1,1-dioxide). Crucially, this oxidation is performed only after the secondary amine has been protected with a tert-butoxycarbonyl (Boc) group in step S6. This sequence is mechanistically vital because unprotected amines are susceptible to oxidation themselves, potentially forming N-oxides or other degradation products that would complicate purification and reduce yield. By using m-chloroperoxybenzoic acid (m-CPBA) as the oxidant under mild room temperature conditions, the process achieves exclusive oxidation of the sulfur atom to the sulfone state without affecting the protected nitrogen or the chlorinated aromatic ring. This level of control over the oxidation state is essential for producing high-purity intermediates that meet the rigorous specifications required for pharmaceutical applications, minimizing the formation of toxic byproducts and ensuring batch-to-batch consistency.
How to Synthesize 9-chloro-2,3,4,5-tetrahydro-1,4-benzothiepin-1,1-dioxide Efficiently
The synthesis of this complex heterocyclic intermediate requires precise control over reaction parameters to maximize yield and purity at every stage. The process begins with the alkylation of o-chlorothiophenol and proceeds through hydrolysis, cyclization, and nitrogen insertion before concluding with reduction, protection, oxidation, and deprotection steps. Each transformation has been optimized to utilize commercially available reagents and standard laboratory equipment, making the route highly adaptable for scale-up operations. For detailed operational procedures, including specific solvent volumes, stirring rates, and purification techniques such as column chromatography conditions, please refer to the standardized synthesis guide provided below which encapsulates the critical process parameters defined in the patent examples.
- Perform thioalkylation of o-chlorothiophenol with ethyl 3-bromopropionate using potassium carbonate at 80°C.
- Execute hydrolysis and subsequent acid-catalyzed ring closure to form the benzothiopyranone core.
- Conduct azide reaction with TMSN3 followed by carbonyl reduction, amine protection, sulfur oxidation, and final deprotection.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, this synthetic route offers substantial strategic benefits by leveraging widely available starting materials and minimizing the reliance on exotic or prohibitively expensive catalysts. The use of o-chlorothiophenol and ethyl 3-bromopropionate as primary feedstocks ensures a stable and cost-effective supply base, as these chemicals are produced in large volumes by multiple global manufacturers, reducing the risk of single-source bottlenecks. Furthermore, the ability to conduct several key steps, including hydrolysis, ring closure, and oxidation, at or near room temperature significantly lowers the energy footprint of the manufacturing process, translating into direct operational cost savings and a reduced carbon footprint which aligns with modern sustainability goals in chemical production.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the use of mild reaction conditions drastically simplify the downstream processing requirements. By avoiding the need for expensive heavy metal scavengers or complex high-pressure equipment, the overall cost of goods sold (COGS) is significantly optimized. The high atom economy of the ring-closing and azide-insertion steps further contributes to waste reduction, ensuring that raw material costs are efficiently converted into valuable product mass rather than lost in side reactions or extensive purification losses.
- Enhanced Supply Chain Reliability: The robustness of this synthetic pathway enhances supply chain resilience by reducing the sensitivity to minor fluctuations in reaction conditions. Since critical steps like the Schmidt rearrangement and sulfur oxidation proceed effectively at ambient temperatures, the process is less prone to thermal runaways or equipment failures that can halt production lines. This reliability ensures consistent lead times for high-purity pharmaceutical intermediates, allowing downstream drug manufacturers to maintain steady production schedules for vital antiviral medications without the fear of unexpected supply disruptions.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on liquid-phase reactions in common organic solvents like ethanol, THF, and dichloromethane, which are easily handled in standard stainless steel reactors. The avoidance of cryogenic conditions or extremely high pressures simplifies the engineering requirements for commercial scale-up from kilograms to metric tons. Additionally, the generation of benign byproducts and the potential for solvent recovery systems make this route compliant with increasingly stringent environmental regulations, facilitating smoother regulatory approvals for manufacturing sites globally.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the synthesis and application of halogenated benzothiepin oxide, derived directly from the experimental data and technical specifications provided in the patent documentation. These insights are intended to clarify the mechanistic advantages and practical considerations for teams evaluating this technology for integration into their own R&D pipelines or manufacturing portfolios. Understanding these details is crucial for assessing the feasibility of adopting this route for large-scale production of RSV therapeutics.
Q: What is the primary advantage of introducing a chlorine atom in this benzothiepin oxide structure?
A: The introduction of a chlorine atom at the 9-position significantly enhances the water solubility of the final pharmaceutical active ingredient, thereby improving bioavailability and therapeutic efficacy against Respiratory Syncytial Virus (RSV) compared to non-halogenated analogs.
Q: How does this synthesis route improve process safety and scalability?
A: The process utilizes mild reaction conditions for critical steps such as hydrolysis and oxidation, often proceeding at room temperature, which reduces energy consumption and minimizes the risks associated with high-temperature exothermic reactions common in traditional heterocyclic synthesis.
Q: Why is the specific sequence of amine protection before sulfur oxidation critical?
A: Protecting the secondary amine with a Boc group prior to oxidizing the sulfur atom prevents unwanted side reactions and over-oxidation of the nitrogen center, ensuring high chemoselectivity and purity of the final sulfone product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9-chloro-2,3,4,5-tetrahydro-1,4-benzothiepin-1,1-dioxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving antiviral drugs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the complex chemistry required for halogenated benzothiepin oxides is executed with precision and consistency. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch meets the exacting standards required for pharmaceutical applications, providing our partners with the confidence they need to advance their clinical programs without delay.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next-generation RSV treatments. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized processes can enhance your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us support your journey from bench-scale discovery to global commercial success with our reliable supply of premium pharmaceutical intermediates.
