Revolutionizing Succinate Production: A Green One-Step Catalytic Approach for Commercial Scale
Revolutionizing Succinate Production: A Green One-Step Catalytic Approach for Commercial Scale
The chemical industry is currently witnessing a paradigm shift towards greener, more efficient synthetic routes, particularly in the production of critical pharmaceutical intermediates like succinic acid diesters. Patent CN102863335A introduces a groundbreaking methodology that fundamentally alters the traditional landscape of succinate synthesis by leveraging carbon dioxide not merely as an inert medium, but as an active participant in the catalytic cycle. This innovation addresses long-standing inefficiencies in the production of compounds such as dimethyl succinate, which serves as a vital precursor for BDO, gamma-butyrolactone, and various high-grade polymer additives. By integrating esterification and hydrogenation into a single, seamless operation, this technology offers a compelling value proposition for manufacturers seeking to optimize their supply chains while adhering to increasingly stringent environmental regulations.
At the heart of this technological advancement is the utilization of supercritical or subcritical carbon dioxide as a dual-function agent, acting simultaneously as a solvent and an acid catalyst. This approach effectively mitigates the severe equipment corrosion and complex waste treatment issues associated with conventional sulfuric acid-catalyzed processes. The structural integrity of the target molecule, dimethyl succinate, shown below, is preserved and enhanced through this gentle yet highly effective reaction environment, ensuring that the final product meets the rigorous purity specifications demanded by the global pharmaceutical and fine chemical sectors.
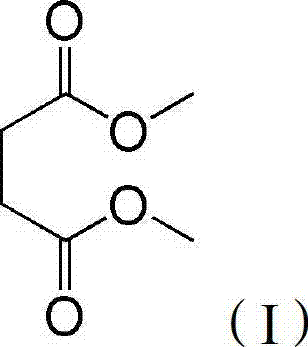
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of succinic acid diesters has relied heavily on the direct esterification of succinic acid with alcohols, typically employing concentrated sulfuric acid as a catalyst. This legacy approach is fraught with significant operational drawbacks that impact both the economic viability and the environmental footprint of the manufacturing process. The use of liquid strong acids necessitates the use of specialized, corrosion-resistant reactor materials, driving up capital expenditure and maintenance costs substantially. Furthermore, the reaction kinetics are often sluggish, requiring prolonged heating and resulting in incomplete conversion rates that complicate downstream purification.
Beyond the immediate reaction challenges, the post-reaction processing in traditional methods is notoriously cumbersome. The neutralization of excess acid generates large volumes of saline wastewater, creating a heavy burden on waste treatment facilities and increasing the overall cost of compliance. Additionally, the separation of the product from the catalyst and by-products often requires multiple distillation steps, which not only consumes excessive energy but also leads to product loss through thermal degradation. These cumulative inefficiencies render conventional methods less competitive in a market that increasingly prioritizes sustainability and cost-effectiveness in the supply of reliable pharmaceutical intermediate suppliers.
The Novel Approach
In stark contrast to these archaic methods, the technology disclosed in CN102863335A utilizes maleic anhydride as the primary starting material, capitalizing on its high reactivity due to the conjugated system within its ring structure. This novel route bypasses the need for pre-formed succinic acid, thereby eliminating a separate production step and its associated energy costs. By introducing hydrogen and a hydrogenation catalyst directly into the esterification mixture, the process achieves a concurrent reduction of the double bond and esterification of the anhydride, streamlining the entire synthetic pathway into a single pot operation.
The introduction of carbon dioxide into this system is the true game-changer, providing a unique reaction medium that enhances mass transfer and catalytic activity without the corrosive effects of mineral acids. As the reaction proceeds, the water generated in situ reacts with CO2 to form carbonic acid, which provides the necessary acidic environment for esterification while remaining easily removable by depressurization. This self-regulating mechanism ensures that the reaction equilibrium is constantly driven towards the product side, facilitated by the favorable solubility of the succinate esters in the compressed gas phase, ultimately leading to superior conversion efficiencies.
Mechanistic Insights into CO2-Assisted Esterification Hydrogenation
To fully appreciate the technical sophistication of this process, one must examine the intricate interplay between the reactants and the catalytic system under elevated pressure. The reaction mechanism begins with the activation of maleic anhydride, where the electron-deficient carbonyl carbons are susceptible to nucleophilic attack by the alcohol molecules. In the presence of the in-situ generated carbonic acid, the electrophilicity of the anhydride is further enhanced, accelerating the ring-opening esterification step. Simultaneously, the hydrogenation catalyst, typically composed of transition metals like Pd, Ni, or Cu supported on oxides or carbon, facilitates the heterolytic cleavage of molecular hydrogen.
The activated hydrogen species then add across the carbon-carbon double bond of the intermediate mono-ester or the unreacted anhydride, saturating the molecule to form the stable succinate backbone. This tandem catalysis is crucial; if esterification were too slow, polymerization might occur, while slow hydrogenation could lead to the accumulation of unstable unsaturated intermediates. The precise control of temperature between 70°C and 160°C and pressure parameters ensures that both reaction rates are synchronized, maximizing the yield of the desired saturated diester while minimizing the formation of oligomers or other side products that could compromise the purity of the final API intermediate.
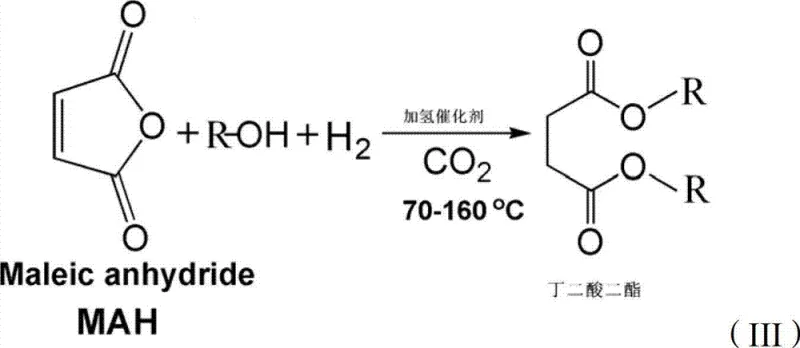
Impurity control in this system is inherently robust due to the homogeneous nature of the reaction medium provided by the dense CO2 phase. Unlike heterogeneous solid acid catalysts which can suffer from pore blockage or deactivation by water, the CO2-mediated system maintains high catalytic activity throughout the reaction duration. The solubility of the product in the supercritical fluid prevents it from precipitating onto the catalyst surface, thereby preserving active sites for continued turnover. This results in a cleaner crude product profile, significantly reducing the load on subsequent purification columns and ensuring that the final material meets the stringent quality standards required for high-purity OLED material or pharmaceutical applications.
How to Synthesize Dimethyl Succinate Efficiently
Implementing this advanced synthesis route requires careful attention to the stoichiometry of reactants and the selection of appropriate catalytic systems to ensure optimal performance. The process is designed to be flexible, accommodating various alcohol chain lengths from methanol to higher alcohols, making it versatile for producing a range of succinate esters. Operators must ensure that the molar ratio of maleic anhydride to alcohol is maintained within the recommended range of 2:1 to 6:1 to drive the equilibrium effectively while preventing excessive dilution of the reaction mixture.
- Mix maleic anhydride, alcohol compound, hydrogenation catalyst, carbon dioxide, and hydrogen in a reactor.
- Maintain reaction temperature between 70°C and 160°C with CO2 pressure of 1-14 MPa and H2 pressure of 0.1-5 MPa.
- Perform esterification and hydrogenation simultaneously in a single step to obtain high-purity succinic acid diester with yields exceeding 99%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this CO2-assisted technology translates into tangible strategic benefits that extend far beyond simple yield improvements. The elimination of corrosive liquid acids fundamentally changes the maintenance schedule and lifecycle costs of production equipment, allowing for the use of standard stainless steel reactors instead of expensive lined vessels. This reduction in capital intensity lowers the barrier to entry for scaling production and improves the overall return on investment for manufacturing facilities dedicated to fine chemical intermediates.
- Cost Reduction in Manufacturing: The consolidation of esterification and hydrogenation into a single step drastically reduces energy consumption by eliminating the need for intermediate isolation and reheating. Furthermore, the removal of sulfuric acid negates the costs associated with neutralization agents and the disposal of hazardous saline waste streams. The high selectivity of the reaction minimizes raw material waste, ensuring that every kilogram of maleic anhydride is converted into valuable product rather than lost to side reactions or purification losses.
- Enhanced Supply Chain Reliability: Maleic anhydride is a widely available commodity chemical with a stable global supply, reducing the risk of raw material shortages that can plague more exotic precursor markets. The robustness of the supported metal catalysts used in this process means they can be regenerated or replaced infrequently, ensuring continuous operation without frequent shutdowns for catalyst change-outs. This reliability is critical for maintaining consistent delivery schedules to downstream customers in the pharmaceutical and agrochemical sectors.
- Scalability and Environmental Compliance: The use of carbon dioxide as a solvent aligns perfectly with modern green chemistry principles, significantly lowering the volatile organic compound (VOC) emissions associated with traditional organic solvents. This environmental advantage simplifies the permitting process for new production lines and future-proofs the facility against tightening environmental regulations. The mild reaction conditions also enhance safety profiles, reducing the risk of thermal runaways and making the scale-up from pilot plant to commercial tonnage a predictable and manageable engineering challenge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this succinate synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing production portfolios.
Q: What are the primary advantages of using CO2 as a solvent in succinate synthesis?
A: Using CO2 eliminates the need for corrosive liquid strong acids like sulfuric acid, significantly reducing equipment maintenance costs and environmental pollution while acting as both a solvent and an acid catalyst.
Q: How does this one-step process improve yield compared to traditional methods?
A: The simultaneous esterification and hydrogenation prevent intermediate degradation and side reactions, driving the equilibrium forward through the solubility of products in CO2, resulting in yields greater than 99%.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the mild reaction conditions (70-160°C) and the use of robust supported metal catalysts make this method highly suitable for large-scale commercial manufacturing with simplified downstream processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dimethyl Succinate Supplier
As the demand for high-quality succinic acid derivatives continues to grow across the pharmaceutical and polymer industries, partnering with an experienced CDMO becomes a strategic imperative. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped to handle the specific pressure and temperature requirements of this advanced CO2-catalyzed process, guaranteeing stringent purity specifications and rigorous QC labs testing for every batch released.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific application requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the potential economic benefits for your organization. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of technical excellence and supply chain security.
