Revolutionizing Prohexadione Calcium Production via Continuous Flow Michael Addition
Revolutionizing Prohexadione Calcium Production via Continuous Flow Michael Addition
The global demand for efficient plant growth regulators continues to surge, driven by the agricultural sector's need for crops with enhanced lodging resistance and stress tolerance. At the heart of synthesizing Prohexadione Calcium, a premier growth inhibitor, lies the critical intermediate diethyl acetonyl succinate. A recent technological breakthrough detailed in patent CN115073291A introduces a transformative production process that shifts from traditional batch processing to a sophisticated continuous flow methodology. This innovation not only addresses the longstanding bottlenecks of high energy consumption and lengthy reaction cycles but also establishes a new benchmark for reliable agrochemical intermediate supplier capabilities. By leveraging a tubular reactor system coupled with a novel benzylamine-DMF catalytic体系, manufacturers can now achieve substantial operational efficiencies. This report delves into the technical nuances of this patent, offering R&D directors and procurement leaders a comprehensive analysis of how this process optimizes the supply chain for high-value agricultural chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
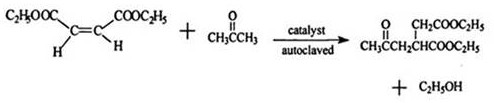
Historically, the industrial synthesis of diethyl acetonyl succinate has been plagued by inefficiencies inherent to batch processing in high-pressure autoclaves. The traditional route involves reacting diethyl maleate with acetone using ethylenediamine as a catalyst under harsh conditions, typically requiring heating to 150°C and maintaining a pressure of approximately 1MPa. This batch operation is notoriously time-consuming, often necessitating reaction times exceeding 20 to 24 hours to reach completion. Such prolonged cycles create significant bottlenecks in the overall production of Prohexadione Calcium, limiting throughput and inflating capital expenditure due to the need for large, pressure-rated vessels. Furthermore, the intermittent nature of batch production leads to inconsistencies in quality and complicates heat management, resulting in higher energy costs per unit of product. The reliance on high-pressure equipment also introduces elevated safety risks and maintenance burdens, making the conventional method increasingly untenable for modern, scalable agrochemical intermediate manufacturing.
The Novel Approach
In stark contrast, the novel approach outlined in the patent utilizes a continuous flow strategy that fundamentally redefines the reaction kinetics and operational parameters. By employing a tubular reactor with a steam jacket, the process allows for precise temperature control between 110°C and 150°C, significantly lower than the peak demands of the old method. The introduction of benzylamine as a catalyst, synergized with N,N-dimethylformamide (DMF) as a co-solvent, dramatically accelerates the Michael addition reaction. This catalytic system reduces the residence time within the reactor to a mere 3 to 5 hours, representing a massive improvement in temporal efficiency. The shift to continuous production eliminates the downtime associated with charging and discharging batch reactors, enabling a seamless, uninterrupted workflow. This transition not only enhances cost reduction in plant growth regulator manufacturing by slashing energy usage but also improves the safety profile by avoiding the complexities of high-pressure batch containment.
Mechanistic Insights into Benzylamine-Catalyzed Michael Addition
The core chemical transformation in this process is a Michael addition reaction between diethyl maleate and acetone, facilitated by a unique catalytic environment. The substitution of ethylenediamine with benzylamine is not merely a change of reagent but a strategic modification of the electronic landscape of the reaction. Benzylamine, possessing a benzene ring structure, offers superior stabilization for the carbanion intermediates formed during the nucleophilic attack compared to the aliphatic ethylenediamine. This structural feature ensures that the reactive species remain stable long enough to effectively target the beta-carbon of the unsaturated ester, thereby driving the equilibrium towards the desired product. Additionally, the inclusion of DMF, a strong polar aprotic solvent, plays a dual role: it increases the steric hindrance around the carbonyl groups, minimizing unwanted 1,2-addition side reactions, and acts as a weak organic base to further promote the addition rate. This synergistic effect allows the reaction to proceed vigorously at lower temperatures, preserving the integrity of the sensitive ester functionalities while maximizing yield.
From an impurity control perspective, the continuous flow nature of the tubular reactor provides unparalleled consistency in reaction conditions, which is critical for minimizing byproduct formation. In batch systems, gradients in temperature and concentration often lead to localized hot spots that foster degradation or polymerization. The plug-flow characteristics of the tubular reactor ensure that every volume element of the reaction mixture experiences the exact same thermal history and residence time. This uniformity is essential for achieving the reported high purity levels, with gas chromatography data indicating content levels reaching 94.2%. For R&D teams focused on high-purity agrochemical intermediates, this mechanistic advantage translates directly into simplified downstream purification processes, reducing the load on distillation columns and lowering solvent consumption during workup.
How to Synthesize Diethyl Acetonyl Succinate Efficiently
Implementing this continuous synthesis route requires careful attention to the mixing and pumping stages to ensure the catalytic activation occurs prior to entering the heated zone. The process begins with a low-temperature mixing step where acetone and diethyl maleate are combined with the benzylamine catalyst and DMF solvent at temperatures between 5°C and 15°C. This pre-mixing is crucial to prevent premature reaction or exotherms before the mixture enters the controlled environment of the tubular reactor. Once homogenized, the stream is pumped into the reactor where steam heating rapidly brings the mixture to the optimal reaction window of 110°C to 150°C. The detailed standardized synthesis steps, including specific flow rates and distillation parameters, are outlined below to guide process engineers in replicating this high-efficiency protocol.
- Mix acetone and diethyl maleate in a low-temperature kettle with benzylamine catalyst and DMF solvent at 5-15°C.
- Pump the mixture into a preheated steam-jacketed tubular reactor, maintaining temperature between 110-150°C for 3-5 hours residence time.
- Distill off acetone at atmospheric pressure followed by vacuum distillation at 11-18mmHg to collect the product at 125-130°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this continuous flow technology represents a strategic opportunity to optimize both cost structures and supply reliability. The drastic reduction in reaction time from over a day to just a few hours means that the same physical footprint of production equipment can generate significantly higher volumes of output annually. This intensification of production capacity directly addresses the challenge of reducing lead time for high-purity agrochemical intermediates, allowing suppliers to respond more agilely to market fluctuations. Moreover, the elimination of high-pressure autoclaves reduces the capital intensity and maintenance overhead associated with the production facility, translating into long-term economic benefits that can be passed down the supply chain. The ability to run the process continuously also mitigates the risk of batch-to-batch variability, ensuring a consistent supply of quality material for downstream formulation.
- Cost Reduction in Manufacturing: The implementation of the benzylamine-DMF catalytic system allows for operation at lower temperatures and pressures, which significantly lowers the energy consumption per kilogram of product produced. By removing the need for expensive high-pressure containment vessels and reducing the cycle time by a factor of roughly five, the overall manufacturing cost structure is drastically simplified. The continuous nature of the process also minimizes solvent loss and improves atom economy, contributing to substantial cost savings without compromising on the quality of the final intermediate.
- Enhanced Supply Chain Reliability: Continuous manufacturing inherently offers greater predictability compared to batch processing, as it removes the start-stop cycles that often lead to delays. The robustness of the tubular reactor system ensures that production can be maintained steadily over long periods, providing a reliable stream of diethyl acetonyl succinate to meet the rigorous demands of the agrochemical industry. This stability is critical for securing long-term contracts and ensuring that the downstream synthesis of Prohexadione Calcium is never interrupted by raw material shortages.
- Scalability and Environmental Compliance: Scaling a continuous flow process is often more straightforward than scaling batch reactions, as it primarily involves running the reactor for longer durations or numbering up identical reactor units rather than building massively larger vessels. This modularity facilitates rapid capacity expansion to meet growing market needs. Furthermore, the improved selectivity and reduced energy footprint align with increasingly stringent environmental regulations, making the process more sustainable and compliant with green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits for potential partners and technical stakeholders. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production lines.
Q: What are the key advantages of the tubular reactor method over the traditional autoclave process?
A: The tubular reactor method replaces the traditional high-pressure autoclave batch process, reducing reaction time from over 24 hours to less than 5 hours. It eliminates the need for high-pressure equipment (1MPa), operates at milder temperatures (110°C vs 150°C), and enables continuous production, significantly improving throughput and safety.
Q: Why is benzylamine preferred over ethylenediamine as a catalyst in this synthesis?
A: Benzylamine, combined with N,N-dimethylformamide (DMF), provides a more stable carbanion environment due to the benzene ring structure. This combination minimizes 1,2-addition competition and accelerates the Michael addition rate, allowing for lower reaction temperatures and shorter residence times compared to ethylenediamine.
Q: What purity levels can be achieved with this new continuous process?
A: According to the patent examples, the process yields diethyl acetonyl succinate with a content of 94.2% as detected by gas chromatography, with an isolated yield of 92.3%. The continuous nature of the flow chemistry ensures consistent quality control compared to batch variations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diethyl Acetonyl Succinate Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that high-quality intermediates play in the efficacy of final agrochemical products. Our expertise in process development allows us to master complex synthetic pathways, such as the continuous flow Michael addition described in patent CN115073291A, ensuring that we can deliver materials that meet the most stringent specifications. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our rigorous QC labs to guarantee batch consistency and purity. Our commitment to technological advancement means we are uniquely positioned to offer high-purity diethyl acetonyl succinate produced via this efficient, cost-effective method.
We invite global partners to collaborate with us to leverage these technical advancements for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your production of Prohexadione Calcium is built on a foundation of reliability and innovation.
