Scalable Synthesis of 3-Hydroxy-4-Amino-5-Nitro-N,N-Dimethyl Benzamide for Tegoprazan Production
The pharmaceutical industry is constantly seeking robust and scalable synthetic routes for novel gastrointestinal therapeutics, particularly for Potassium-Competitive Acid Blockers (P-CABs) like Tegoprazan. Patent CN111018736A discloses a groundbreaking methodology for the preparation of 3-hydroxy-4-amino-5-nitro-N,N-dimethyl benzamide, a pivotal intermediate in the value chain of this advanced drug. This specific compound serves as the structural backbone required for the final assembly of Tegoprazan, and its efficient production is critical for meeting global demand. The disclosed technology offers a distinct departure from traditional, often erratic synthesis paths by introducing a sophisticated protection-deprotection sequence that guarantees high regio-selectivity. By leveraging solid phosgene for simultaneous amino and hydroxyl protection, the process creates a stable benzoxazole scaffold that withstands harsh nitration conditions. This technical breakthrough not only simplifies the operational workflow but also ensures that the final intermediate meets the stringent purity profiles demanded by modern regulatory bodies for API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of highly functionalized aminophenol derivatives has been plagued by significant chemoselectivity challenges, particularly when introducing electron-withdrawing groups like nitro functionalities. Conventional direct nitration of 3-hydroxy-4-amino benzamide derivatives often results in a complex mixture of oxidation byproducts and regio-isomers due to the high reactivity of the free amino and phenolic hydroxyl groups. The unprotected amino group is susceptible to oxidation by nitric acid, leading to tar formation and a drastic reduction in overall yield. Furthermore, without a directing group or protective mask, the nitro group may attach to undesired positions on the aromatic ring, creating difficult-to-remove impurities that compromise the safety profile of the final drug substance. These inefficiencies necessitate extensive and costly purification steps, such as repeated recrystallizations or preparative chromatography, which are economically unsustainable for large-scale commercial production. The instability of the starting materials under acidic nitration conditions further exacerbates supply chain risks, making consistent batch-to-batch quality difficult to achieve.
The Novel Approach
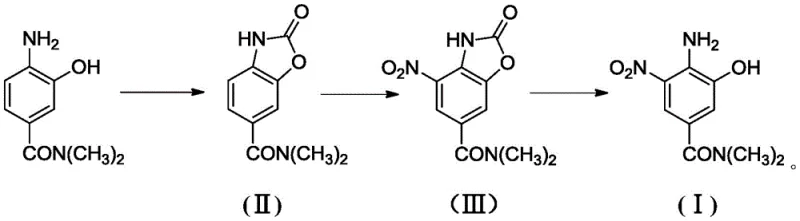
The innovative route described in the patent data fundamentally resolves these issues by employing a strategic cyclization step to form an N,N-dimethyl-2-oxa-2,3-dihydrobenzo[d]oxazole-6-carboxamide intermediate. This transformation effectively locks the reactive amino and hydroxyl functionalities into a rigid heterocyclic ring system, rendering them inert to oxidative degradation during the subsequent nitration phase. This protective architecture allows for the precise introduction of the nitro group at the 4-position relative to the carboxamide, driven by the electronic properties of the oxazole ring. The process utilizes readily available industrial reagents such as solid phosgene and mixed acid, operating under controlled thermal conditions that range from cryogenic temperatures for nitration to moderate heating for cyclization. By decoupling the protection, functionalization, and deprotection stages, the method achieves exceptional selectivity and minimizes side reactions. This streamlined approach not only enhances the chemical yield but also drastically simplifies the downstream processing, making it an ideal candidate for reliable pharmaceutical intermediate supplier operations aiming for cost-effective manufacturing.
Mechanistic Insights into Benzoxazole-Mediated Selective Nitration
The core of this synthetic advancement lies in the formation and utilization of the benzoxazole ring as a temporary protecting group. In the initial step, 3-hydroxy-4-amino-N,N-dimethyl benzamide reacts with solid phosgene in a solvent like 1,1,2-trichloroethane. The mechanism involves the nucleophilic attack of the amino nitrogen and the phenolic oxygen on the carbonyl carbon of the phosgene equivalent, resulting in the elimination of hydrogen chloride and the closure of the five-membered oxazole ring. This cyclization is thermodynamically favorable and proceeds efficiently at temperatures between 0°C and 80°C. The resulting intermediate (II) possesses a unique electronic distribution where the electron density of the benzene ring is modulated by the fused heterocycle. This modulation is crucial because it deactivates the ring towards electrophilic attack just enough to prevent runaway reactions while still allowing substitution at the specific position para to the electron-donating amide group. The stability of this intermediate is key; it prevents the formation of quinone-imine type oxidation products that typically plague unprotected aminophenols.
Following the protection, the nitration step introduces the nitro group using a mixture of concentrated nitric and sulfuric acids. The reaction is conducted at low temperatures, specifically between -10°C and 0°C, to maintain kinetic control over the electrophilic aromatic substitution. The benzoxazole ring directs the incoming nitronium ion to the 4-position (relative to the original amide), which corresponds to the 5-position in the final product numbering. This regio-selectivity is enforced by the steric and electronic environment of the oxazole moiety. Once the nitro group is installed, the final step involves hydrolytic ring opening. Treatment with aqueous sodium hydroxide cleaves the carbamate-like linkage within the oxazole ring, regenerating the free phenolic hydroxyl and amino groups. The pH is carefully adjusted first to acidic conditions to remove insolubles and then to neutral or slightly basic conditions to precipitate the pure product. This sequence ensures that the sensitive functional groups are only exposed once the potentially damaging nitration environment has been completely removed.
How to Synthesize 3-Hydroxy-4-Amino-5-Nitro-N,N-Dimethyl Benzamide Efficiently
The execution of this synthesis requires precise adherence to the thermal and stoichiometric parameters outlined in the patent to ensure optimal yield and purity. The process begins with the charging of the starting aminophenol derivative and 1,1,2-trichloroethane into a reactor equipped with efficient cooling and heating capabilities. Solid phosgene is added in batches while maintaining the temperature below 10°C to manage the exotherm, followed by a prolonged heating period at 80°C to drive the cyclization to completion. The subsequent nitration demands rigorous temperature control, utilizing an ice-salt bath to keep the reaction mixture below 0°C during the addition of mixed acid. Finally, the hydrolysis is performed under mild alkaline conditions at room temperature, avoiding the need for extreme heat or pressure. For a detailed breakdown of the specific equipment setup, reagent grades, and workup procedures, please refer to the standardized protocol below.
- Cyclization Protection: React 3-hydroxy-4-amino-N,N-dimethyl benzamide with solid phosgene in 1,1,2-trichloroethane at 0-10°C, then heat to 80°C to form the benzoxazole intermediate (II).
- Selective Nitration: Treat intermediate (II) with mixed acid (nitric/sulfuric) at -10 to 0°C to introduce the nitro group selectively, yielding intermediate (III).
- Hydrolytic Deprotection: Hydrolyze intermediate (III) using 5% sodium hydroxide solution at room temperature, followed by pH adjustment to isolate the final product (I).
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this novel synthetic route offers substantial advantages over legacy methods by fundamentally altering the cost structure and reliability of the manufacturing process. The elimination of complex purification steps and the use of commodity chemicals significantly lower the barrier to entry for production, translating into tangible economic benefits for downstream API manufacturers. The robustness of the chemistry means that production schedules are less likely to be disrupted by failed batches or inconsistent quality, thereby enhancing the overall security of supply for critical gastrointestinal medications. Furthermore, the scalability of the process allows for flexible production volumes, enabling suppliers to respond rapidly to fluctuations in market demand without requiring massive capital investment in specialized equipment. This adaptability is crucial in the dynamic pharmaceutical landscape where speed to market can determine commercial success.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the dramatic improvement in chemical selectivity, which eliminates the need for expensive and time-consuming purification techniques such as column chromatography or multiple recrystallizations. By preventing the formation of oxidation byproducts through the benzoxazole protection strategy, the process achieves high crude purity, allowing for direct isolation of the product through simple filtration and washing. Additionally, the raw materials employed, including solid phosgene and common mineral acids, are widely available in the global chemical market at competitive prices, avoiding the reliance on exotic or proprietary catalysts. The high yield reported in the patent examples, often exceeding 85% per step, further contributes to cost efficiency by maximizing the throughput of valuable starting materials and minimizing waste disposal costs associated with low-yielding reactions.
- Enhanced Supply Chain Reliability: The reliance on mature, off-the-shelf reagents ensures that the supply chain is resilient against shortages of specialized precursors. Unlike processes that depend on custom-synthesized building blocks with long lead times, this method utilizes chemicals that are produced in multi-ton quantities by major chemical manufacturers worldwide. The operational simplicity of the reaction steps, which involve standard unit operations like stirring, heating, cooling, and filtration, means that the process can be transferred easily between different manufacturing sites without significant re-engineering. This flexibility reduces the risk of supply disruption caused by site-specific issues and allows for the establishment of a diversified supplier base. Moreover, the mild reaction conditions reduce the wear and tear on equipment, leading to lower maintenance costs and higher asset availability for continuous production runs.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with reaction conditions that are safe and manageable even at multi-ton scales. The use of 1,1,2-trichloroethane as a solvent, while requiring proper handling, allows for efficient recovery and recycling, aligning with modern green chemistry principles of solvent management. The absence of heavy metal catalysts or hazardous oxidants simplifies the waste treatment process, reducing the environmental footprint of the manufacturing facility. The high atom economy of the cyclization and nitration steps ensures that a maximum proportion of the reactant mass is incorporated into the final product, minimizing the generation of hazardous waste streams. This environmental compatibility facilitates smoother regulatory approvals and reduces the long-term liability associated with waste disposal, making it a sustainable choice for long-term commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of adopting this technology. Understanding these details is essential for R&D teams evaluating the feasibility of this route and for procurement officers assessing the reliability of the supply source. The answers reflect the optimized conditions and observed outcomes from the pilot-scale examples provided in the intellectual property disclosure.
Q: Why is the benzoxazole protection strategy superior for this synthesis?
A: The benzoxazole protection strategy simultaneously masks both the amino and hydroxyl groups, preventing oxidation and ensuring high regio-selectivity during the subsequent nitration step, which significantly reduces impurity formation compared to direct nitration methods.
Q: What are the critical control points for the nitration reaction?
A: Temperature control is critical; the reaction mixture must be maintained between -10°C and 0°C during the addition of mixed acid to prevent over-nitration or decomposition of the sensitive benzoxazole ring structure.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes common industrial solvents like 1,1,2-trichloroethane and standard reagents like solid phosgene and sulfuric acid, with mild reaction conditions that facilitate easy scale-up from kilogram to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Hydroxy-4-Amino-5-Nitro-N,N-Dimethyl Benzamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development and commercialization of novel therapeutics like Tegoprazan. Our technical team has thoroughly analyzed the synthetic pathway disclosed in CN111018736A and possesses the expertise to execute this chemistry with precision and consistency. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met regardless of the project phase. Our facilities are equipped with state-of-the-art reactors capable of handling the specific thermal requirements of the cyclization and nitration steps, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets your exacting standards. We are committed to being a partner that not only supplies materials but also provides technical solutions to optimize your overall manufacturing process.
We invite you to collaborate with us to leverage this advanced synthetic route for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. Our technical procurement team is ready to provide specific COA data from our pilot runs and comprehensive route feasibility assessments to demonstrate how we can enhance your production efficiency. Contact us today to discuss how our capabilities align with your strategic goals for Tegoprazan intermediate sourcing and to initiate a dialogue on securing a stable, high-quality supply for your future commercial needs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
