Scalable Synthesis of Dicyclopenteno Perylene Derivatives for Next-Gen Organic Semiconductors
Scalable Synthesis of Dicyclopenteno Perylene Derivatives for Next-Gen Organic Semiconductors
The rapid evolution of organic electronics demands materials that combine high charge carrier mobility with solution processability, a balance that traditional silicon-based technologies often struggle to achieve in flexible applications. Patent CN110937967A introduces a groundbreaking methodology for the preparation of dicyclopenteno[cd,lm]perylene derivatives, a class of compounds poised to redefine the performance metrics of organic field effect transistors (OFETs). This technology leverages a robust two-step synthetic strategy that transforms readily available tetrahydrodicyclopentano precursors into highly functionalized semiconductor cores. By integrating cyclopentene rings into the perylene scaffold, the invention significantly enhances the electron-withdrawing properties and planarity of the molecule, which are critical factors for improving intermolecular stacking and carrier migration. As a reliable organic semiconductor material supplier, understanding the nuances of this patent allows us to offer clients superior materials that bridge the gap between laboratory innovation and commercial viability.
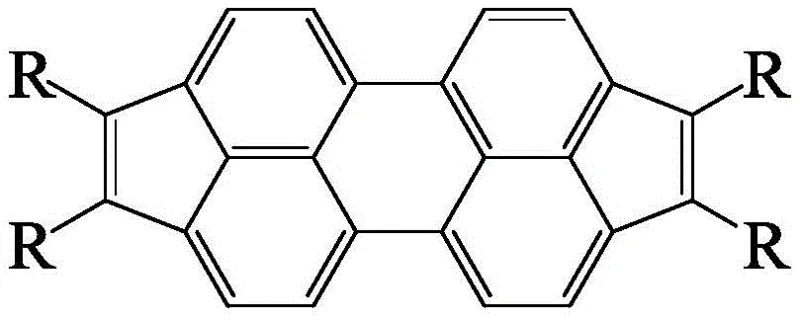
The core innovation lies in the versatility of the molecular architecture, where the 'R' groups can be systematically varied to include alkyl, aryl, or thienyl moieties. This modularity is not merely a chemical curiosity but a strategic advantage for R&D directors seeking to fine-tune the HOMO-LUMO energy levels of their devices. The patent explicitly details how introducing halogen atoms serves as a pivotal bridge for structural adjustment, enabling the precise installation of diverse functional groups through subsequent cross-coupling reactions. This level of control over the impurity profile and molecular geometry is essential for achieving the high purity standards required in display and optoelectronic applications. Consequently, this synthesis route represents a significant leap forward in the design of high-performance organic micromolecule field effect materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex perylene derivatives has been plagued by harsh reaction conditions, poor regioselectivity, and limited scope for functionalization. Traditional methods often rely on direct functionalization of the perylene core, which can lead to mixtures of isomers that are notoriously difficult to separate, thereby drastically reducing the overall yield and purity of the final semiconductor material. Furthermore, many conventional routes require expensive or hazardous reagents that pose significant challenges for environmental compliance and operator safety in a manufacturing setting. The lack of a generalized protocol for introducing diverse substituents has also restricted the ability of material scientists to rapidly iterate on molecular designs to optimize device performance. These bottlenecks have slowed the commercial adoption of high-mobility organic semiconductors, as the cost of goods sold remains prohibitively high for mass-market applications like wearable electronics.
The Novel Approach
In stark contrast, the methodology disclosed in CN110937967A offers a streamlined and universally applicable pathway that circumvents these historical hurdles. The process initiates with a highly selective radical bromination of 1,2,7,8-tetrahydrodicyclopentano[cd,lm]perylene, utilizing benign peroxide catalysts and standard brominating agents like N-bromosuccinimide (NBS) or dibromohydantoin (DBDMH). This step efficiently generates a reactive tetrabromo intermediate with excellent fidelity, setting the stage for precise downstream modifications. The subsequent Suzuki coupling reaction allows for the introduction of a wide array of substituents under relatively mild thermal conditions, typically ranging from 60°C to 90°C. This approach not only simplifies the purification process but also ensures that the delicate perylene backbone remains intact, preserving the electronic properties essential for device function.
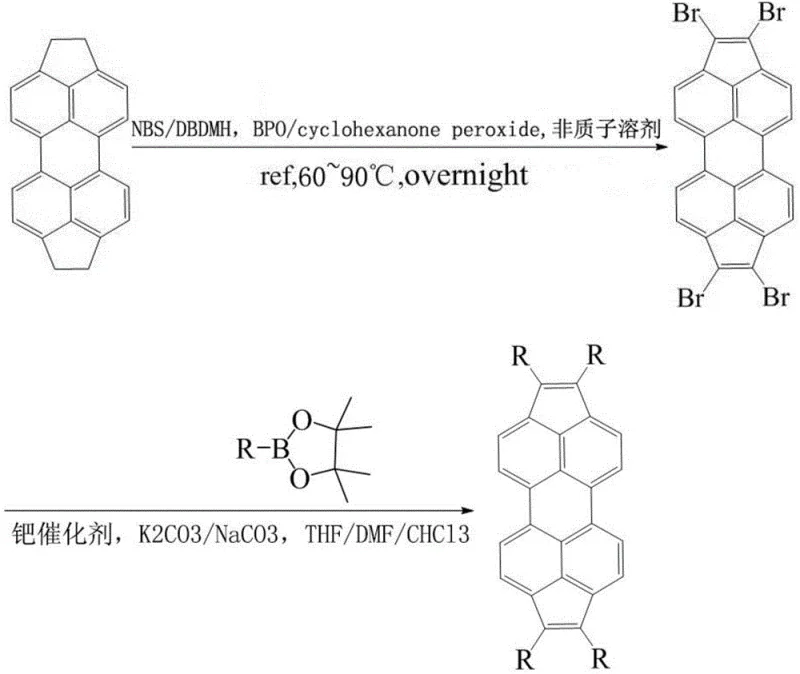
The beauty of this novel approach is its adaptability; by simply swapping the boronic acid pinacol ester reagent in the second step, manufacturers can produce a library of derivatives tailored for specific electronic applications without overhauling the entire production line. This modularity translates directly into cost reduction in electronic chemical manufacturing, as the same infrastructure can be used to produce multiple SKUs. The use of common organic solvents such as carbon tetrachloride, chloroform, THF, and DMF further enhances the practicality of the method, ensuring that supply chains remain resilient and raw material sourcing is straightforward. For procurement managers, this means a stable supply of high-quality intermediates with predictable lead times and reduced risk of production stoppages due to reagent scarcity.
Mechanistic Insights into Radical Bromination and Suzuki Coupling
The first stage of the synthesis involves a free-radical substitution mechanism driven by the decomposition of peroxides such as dibenzoyl peroxide (BPO) or cyclohexanone peroxide. These initiators generate radical species that abstract hydrogen atoms from the benzylic positions of the tetrahydrodicyclopentano precursor, creating reactive carbon radicals. These radicals then react with the brominating agent to form the carbon-bromine bonds necessary for the subsequent cross-coupling. The patent specifies a molar ratio of precursor to NBS between 1:4 and 1:10, ensuring complete tetra-bromination while minimizing side reactions. The choice of solvent plays a crucial role here; non-polar or moderately polar aprotic solvents facilitate the radical propagation steps while maintaining the solubility of the large polycyclic aromatic hydrocarbon substrate. This careful optimization of reaction parameters ensures that the intermediate 1,2,7,8-tetrabromobicyclopenteno[cd,lm]perylene is obtained with high purity, which is a prerequisite for the success of the catalytic step that follows.
The second stage employs a palladium-catalyzed Suzuki-Miyaura cross-coupling, a cornerstone reaction in modern organic synthesis known for its tolerance of functional groups and mild conditions. In this cycle, the palladium catalyst, such as tetrakis(triphenylphosphine)palladium or palladium acetate derivatives, undergoes oxidative addition with the aryl bromide intermediate. This is followed by transmetallation with the organoboron species in the presence of a base like potassium carbonate or sodium carbonate. The final reductive elimination step releases the coupled product and regenerates the active palladium catalyst. The patent highlights the use of various boronic acid pinacol esters, including phenyl, thienyl, and benzothiophene derivatives, demonstrating the reaction's broad scope. From an impurity control perspective, the use of pinacol esters is advantageous as they are generally more stable and less prone to protodeboronation than free boronic acids, leading to cleaner reaction profiles and easier downstream processing.
How to Synthesize Dicyclopenteno Perylene Derivatives Efficiently
Implementing this synthesis route requires strict adherence to the optimized conditions outlined in the patent to ensure maximum yield and reproducibility. The process begins with the preparation of the tetrabromo intermediate, where temperature control between 60°C and 90°C is critical to drive the reaction to completion overnight without degrading the sensitive perylene core. Following isolation and purification of the intermediate, the Suzuki coupling is performed under an inert argon atmosphere to prevent catalyst deactivation by oxygen. The detailed standardized synthesis steps below provide a roadmap for scaling this chemistry from gram-scale laboratory experiments to multi-kilogram pilot runs.
- Perform radical bromination of 1,2,7,8-tetrahydrodicyclopentano[cd,lm]perylene using NBS or DBDMH with a peroxide catalyst at 60-90°C to obtain the tetrabromo intermediate.
- Conduct a Suzuki coupling reaction between the tetrabromo intermediate and various boronic acid pinacol ester derivatives using a palladium catalyst and base.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the transition to this novel synthesis platform offers tangible benefits that extend beyond mere technical performance. The reliance on commodity chemicals such as NBS, peroxides, and standard palladium catalysts means that the raw material supply chain is robust and less susceptible to the volatility often seen with exotic reagents. This stability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of the consumer electronics sector. Furthermore, the simplicity of the workup procedures, which involve standard filtration, solvent removal, and recrystallization steps, reduces the operational complexity and labor costs associated with manufacturing. By eliminating the need for complex chromatographic separations often required by older methods, the process inherently supports a leaner and more efficient production model.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the high atom economy of the Suzuki coupling and the use of inexpensive starting materials. By avoiding multi-step protection and deprotection sequences common in other synthetic routes, the overall number of unit operations is significantly reduced. This consolidation of steps leads to substantial savings in utility consumption, solvent usage, and waste disposal costs. Additionally, the high yields reported for both the bromination and coupling steps minimize the loss of valuable intermediates, directly improving the cost per kilogram of the final active pharmaceutical ingredient or electronic material. The ability to recycle solvents like THF and chloroform further enhances the financial attractiveness of the method for large-scale operations.
- Enhanced Supply Chain Reliability: The universality of the synthetic method allows for the production of a wide range of derivatives using a common intermediate. This flexibility enables manufacturers to respond rapidly to changes in market demand by switching the boronic acid reagent without retooling the entire facility. Such agility is a key competitive advantage in the fast-paced organic electronics industry, where product lifecycles are short and customization is key. Moreover, the use of stable boronic acid pinacol esters ensures that reagents can be stocked for longer periods without degradation, reducing the risk of production delays caused by expired or compromised raw materials. This reliability fosters stronger partnerships between suppliers and downstream device manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and mixing, but the moderate temperatures (60-90°C) and ambient pressure conditions of this reaction make it highly amenable to scale-up. The process does not require specialized high-pressure reactors or cryogenic cooling, allowing it to be implemented in standard glass-lined or stainless steel vessels. From an environmental standpoint, the reduction in solvent volume and the potential for solvent recovery align with green chemistry principles. The avoidance of heavy metal contaminants, aside from the trace palladium which can be scavenged, ensures that the final product meets the stringent purity specifications required for electronic applications, thereby reducing the regulatory burden associated with hazardous substance management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these advanced semiconductor materials. Our answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details is vital for making informed decisions about material selection and process integration.
Q: What are the key advantages of this synthesis route for organic semiconductors?
A: The route utilizes commercially available starting materials and mild reaction conditions (60-90°C), ensuring high reproducibility and ease of scale-up for industrial manufacturing of organic field effect transistors.
Q: Can the substituent groups on the perylene core be customized?
A: Yes, the method is highly universal. By varying the boronic acid pinacol ester reagent in the second step, diverse substituents such as alkyl, aryl, and thienyl groups can be introduced to tune electronic properties.
Q: Is this process suitable for large-scale commercial production?
A: Absolutely. The use of standard solvents like THF, DMF, and chloroform, along with robust catalysts like Pd(PPh3)4, makes the process adaptable for multi-kilogram to ton-scale production without specialized equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dicyclopenteno Perylene Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent to product requires more than just chemical knowledge; it demands a partner with deep expertise in process engineering and quality assurance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We understand that high-purity dicyclopenteno perylene derivatives require stringent purity specifications and rigorous QC labs to detect trace impurities that could compromise device performance. Our state-of-the-art facilities are equipped to handle the specific solvent systems and catalytic requirements of this synthesis, guaranteeing a product that meets the highest industry standards for organic semiconductor applications.
We invite you to collaborate with us to unlock the full potential of this technology for your specific application needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to validate the feasibility of a new derivative, our technical procurement team is ready to assist. Please contact us today to request specific COA data and route feasibility assessments tailored to your project requirements. Let us help you accelerate your time-to-market with reliable, high-performance materials designed for the next generation of flexible electronics.
