Scalable Production of 2-Triisopropylsilyl Indoles via Novel Palladium Catalysis for Advanced Drug Discovery
Scalable Production of 2-Triisopropylsilyl Indoles via Novel Palladium Catalysis for Advanced Drug Discovery
The pharmaceutical and material science industries continuously demand efficient access to complex heterocyclic scaffolds, particularly 2-alkynyl indoles, which serve as critical building blocks for bioactive molecules and functional materials. Patent CN106279236B, published in April 2020, introduces a groundbreaking methodology for synthesizing 2-triisopropylsilyl acetylene indole compounds through a streamlined palladium-catalyzed C-H activation strategy. This innovation represents a significant departure from traditional synthetic routes that often rely on harsh cyclization conditions or expensive transition metal catalysts. By leveraging a pyrimidine-directed approach, the disclosed technology enables the direct functionalization of the indole core at the C2-position with high precision and atom economy. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, this patent offers a compelling pathway to reduce manufacturing complexity while maintaining stringent purity standards required for downstream drug development applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-alkynyl indole derivatives has been plagued by significant synthetic challenges that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Traditional approaches often involve the cyclization of 2-(2,2-dibromovinyl)aniline or similar precursors, which are not only difficult to source but also require multi-step preparation involving hazardous reagents. Furthermore, earlier transition metal-catalyzed dehydrocoupling reactions frequently utilized expensive cobalt or rhodium catalysts, such as [Cp*CoI2]2, which impose substantial cost burdens on large-scale production. Another prevalent issue with prior art methods, such as those employing high-valent iodine reagents, is the inherent instability and safety risks associated with these oxidants, alongside their limited substrate scope. These conventional pathways often suffer from poor regioselectivity, leading to mixtures of C2 and C3 substituted products that require extensive and yield-reducing purification processes, ultimately impacting the overall economic viability of the manufacturing process.
The Novel Approach
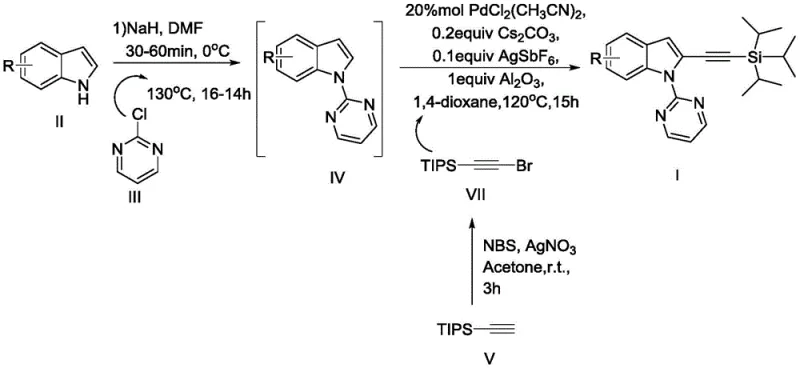
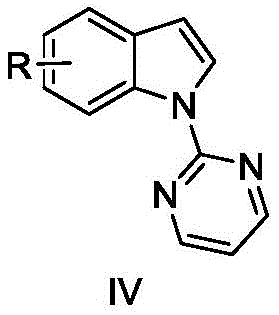
In stark contrast to these legacy methods, the technology disclosed in CN106279236B utilizes a sophisticated yet practical palladium-catalyzed direct C-H activation mechanism that fundamentally simplifies the synthetic landscape. The core innovation lies in the strategic installation of a pyrimidine directing group at the nitrogen atom of the indole ring, which effectively guides the palladium catalyst to the desired C2-position for alkynylation. This method employs readily available and cost-effective palladium bis(acetonitrile) dichloride as the catalyst, coupled with triisopropylsilylacetylene bromide as the alkynylating agent. The reaction proceeds under relatively mild thermal conditions in 1,4-dioxane, utilizing cesium carbonate and silver hexafluoroantimonate as essential additives to facilitate the catalytic cycle. This novel approach not only bypasses the need for pre-functionalized substrates but also demonstrates excellent compatibility with various functional groups, including halogens and esters, thereby expanding the chemical space accessible for medicinal chemistry campaigns.
Mechanistic Insights into Pd-Catalyzed C-H Activation and Alkynylation
The mechanistic elegance of this synthesis relies on the cooperative interaction between the pyrimidine directing group and the palladium center to achieve high regioselectivity. Initially, the indole substrate is converted into a 1-(pyrimidinyl-2-yl)-1H-indole intermediate, where the nitrogen atoms of the pyrimidine ring coordinate with the palladium species. This coordination brings the metal center into close proximity with the C2-H bond of the indole moiety, facilitating the cleavage of this specific carbon-hydrogen bond through a concerted metalation-deprotonation (CMD) or electrophilic palladation pathway. The presence of aluminum oxide in the reaction mixture is believed to play a crucial role as a heterogeneous additive, potentially assisting in the removal of acidic byproducts or stabilizing the active catalytic species, although its exact mechanistic function remains a subject of detailed kinetic study. The subsequent oxidative addition of the triisopropylsilylacetylene bromide to the palladated intermediate forms a high-valent palladium complex, which then undergoes reductive elimination to forge the new carbon-carbon bond, releasing the desired 2-alkynylated product and regenerating the active Pd(II) catalyst for the next turnover.
Controlling impurity profiles is paramount for any process intended for high-purity pharmaceutical intermediate manufacturing, and this method offers distinct advantages in this regard. The use of a directing group inherently suppresses the formation of C3-alkynylated byproducts, which are thermodynamically favored in many electrophilic substitution reactions of indoles. Furthermore, the choice of triisopropylsilyl (TIPS) as the protecting group for the alkyne provides steric bulk that further enhances selectivity and stability during the reaction. The reaction conditions, specifically the use of silver hexafluoroantimonate, assist in halide abstraction, ensuring that the palladium center remains coordinatively unsaturated and reactive towards the alkyne coupling partner. Experimental data from the patent indicates that omitting key components like the silver salt or the aluminum oxide leads to a drastic reduction in yield, highlighting the delicate balance of the catalytic system. This precise control over reaction parameters ensures that the final crude product contains minimal structural isomers, simplifying the downstream purification workload and enhancing the overall process mass intensity.
How to Synthesize 2-Triisopropylsilyl Indoles Efficiently
The practical implementation of this synthesis route involves a sequential two-step protocol that begins with the preparation of the directing group precursor followed by the key alkynylation event. The initial step requires the reaction of the parent indole or substituted indole with 2-chloropyrimidine in the presence of sodium hydride in DMF, establishing the necessary N-pyrimidyl linkage. Once this intermediate is isolated and purified, it serves as the substrate for the palladium-catalyzed coupling reaction. The detailed operational parameters, including specific molar ratios of catalysts and additives, are critical for achieving the reported yields ranging from 37% to 80% depending on the substrate electronics. For process chemists looking to replicate or adapt this methodology, understanding the precise stoichiometry and temperature controls is essential for success. The detailed standardized synthesis steps see the guide below.
- Preparation of the directing group intermediate by reacting indole with 2-chloropyrimidine under basic conditions (NaH/DMF) at elevated temperatures.
- Execution of the alkynylation reaction using PdCl2(CH3CN)2 catalyst, Cs2CO3 base, and triisopropylsilylacetylene bromide in 1,4-dioxane at 120°C.
- Purification of the crude reaction mixture via silica gel column chromatography to isolate the high-purity 2-triisopropylsilyl indole derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented methodology offers transformative benefits for procurement managers and supply chain heads focused on cost reduction in pharmaceutical intermediate manufacturing. The shift from expensive and scarce cobalt or rhodium catalysts to widely available palladium salts represents a significant optimization of the bill of materials. Additionally, the avoidance of hazardous high-valent iodine reagents eliminates the need for specialized handling protocols and waste disposal procedures associated with explosive or unstable oxidants. This simplification of the chemical inventory directly translates to reduced operational overheads and enhanced workplace safety, which are critical metrics for modern chemical manufacturing facilities. The robustness of the reaction conditions also implies a lower risk of batch failures due to sensitive reagent degradation, thereby ensuring more predictable production schedules and reliable delivery timelines for downstream clients.
- Cost Reduction in Manufacturing: The utilization of inexpensive palladium catalysts and commercially sourced indole starting materials drastically lowers the raw material expenditure compared to traditional cyclization routes. By eliminating the pre-functionalization steps required for aniline-based precursors, the overall number of synthetic operations is reduced, leading to substantial savings in labor, energy, and solvent consumption. The high atom economy of the direct C-H activation approach ensures that a greater proportion of the input mass is converted into the valuable product, minimizing waste generation and associated disposal costs. Furthermore, the ability to tolerate diverse functional groups without the need for additional protection and deprotection steps streamlines the synthesis, further driving down the cost of goods sold for complex heterocyclic intermediates.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as indoles, 2-chloropyrimidine, and standard palladium salts ensures a resilient supply chain that is less susceptible to geopolitical disruptions or single-source bottlenecks. Unlike specialized organometallic catalysts that may have long lead times, the reagents for this process are readily available from multiple global suppliers, guaranteeing continuity of supply. The mild reaction conditions reduce the stress on reactor equipment and minimize the formation of tarry byproducts that can foul processing lines, thereby extending campaign lengths and reducing maintenance downtime. This operational stability allows manufacturers to respond more agilely to fluctuating market demands, securing their position as a reliable pharmaceutical intermediate supplier in a competitive landscape.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents like 1,4-dioxane that, while requiring careful management, are standard in industrial organic synthesis and have established recovery protocols. The reduction in reaction steps and the avoidance of heavy metal contaminants from exotic catalysts simplify the purification workflow, making it easier to meet stringent regulatory limits for residual metals in API intermediates. The improved selectivity reduces the volume of organic waste generated per kilogram of product, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing process. This alignment with sustainability goals is increasingly important for multinational corporations seeking to optimize their Scope 3 emissions and adhere to strict environmental, social, and governance (ESG) criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and experimental data. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into their existing production pipelines. The answers provided reflect the specific capabilities and limitations observed during the development of this method, ensuring transparency and accuracy for potential partners. We encourage stakeholders to review these points carefully to assess the fit for their specific project requirements.
Q: What are the primary advantages of this Pd-catalyzed method over conventional cyclization routes?
A: This method utilizes direct C-H bond activation, eliminating the need for complex pre-functionalized starting materials like 2-(2,2-dibromovinyl)aniline. It employs inexpensive palladium catalysts instead of costly cobalt or rhodium systems, significantly improving atom economy and reducing raw material costs.
Q: How does the pyrimidine directing group influence the reaction selectivity?
A: The pyrimidine group introduced at the N1-position of the indole acts as a powerful directing group, guiding the palladium catalyst specifically to the C2-position. This ensures high regioselectivity for the alkynylation, minimizing the formation of unwanted C3-substituted byproducts common in non-directed electrophilic substitutions.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process operates under relatively mild conditions (120°C) using commercially available solvents like 1,4-dioxane and stable reagents. The use of robust catalysts and the avoidance of hazardous high-valent iodine reagents make it highly amenable to scale-up for industrial pharmaceutical intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Triisopropylsilyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development timelines. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory techniques like the one described in CN106279236B can be successfully translated into robust industrial processes. We are committed to delivering high-purity pharmaceutical intermediates that meet rigorous quality standards, supported by our state-of-the-art analytical infrastructure and stringent purity specifications. Our rigorous QC labs employ advanced chromatographic and spectroscopic techniques to verify the identity and purity of every batch, guaranteeing that our clients receive materials that are ready for immediate use in sensitive downstream transformations.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. By partnering with our technical procurement team, you can gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in C-H activation chemistry can drive efficiency and value for your organization. Let us be your trusted partner in navigating the complexities of modern chemical synthesis.
