Advanced CAN-Catalyzed Synthesis of E-Alkenyl Diphenylphosphines for Commercial Scale-Up
The landscape of organophosphorus chemistry is undergoing a significant transformation driven by the urgent need for sustainable and economically viable synthetic routes. Patent CN108659043B introduces a groundbreaking methodology for the preparation of alkenyl diphenylphosphine compounds, which are critical building blocks in the synthesis of advanced ligands and functional materials. This innovation leverages the unique oxidative properties of ceric ammonium nitrate (CAN) to facilitate a direct coupling between aryl vinyl derivatives and diphenylphosphine oxide. Unlike traditional methods that rely on complex catalytic systems, this approach streamlines the reaction architecture by utilizing a single, inexpensive inorganic oxidant. For R&D directors and process chemists, this represents a paradigm shift towards simpler, more robust reaction systems that minimize the risk of heavy metal contamination in high-value pharmaceutical intermediates. The protocol operates under relatively mild thermal conditions and demonstrates exceptional versatility across a broad spectrum of substituted styrenes, ensuring that the production of these vital compounds can be scaled with confidence and precision.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alkenyl phosphine compounds has been fraught with significant economic and operational challenges that hinder large-scale adoption. Conventional strategies often depend heavily on the addition of precious metal catalysts such as palladium, platinum, or rhodium, which not only inflate the raw material costs but also introduce severe toxicity concerns that complicate downstream processing. Furthermore, existing literature describes methods requiring a cocktail of additives, including transition metal catalysts, stoichiometric oxidants, and specialized ligands, to achieve acceptable conversion rates. For instance, comparative methodologies often necessitate the use of silver nitrate combined with TEMPO and persulfates, creating a complex reaction matrix that is difficult to control and purify. 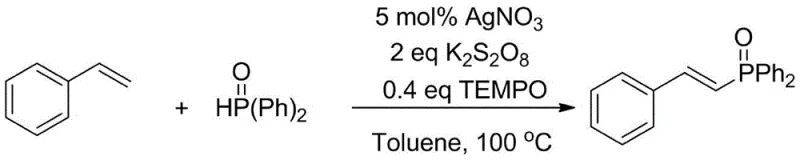 This complexity leads to lower atom economy, as significant amounts of reagents are wasted in side reactions or remain as difficult-to-remove impurities. The reliance on multiple reagents also increases the logistical burden on supply chain managers, who must source and quality-control a wider array of sensitive chemicals, thereby increasing the risk of production delays and batch failures.
This complexity leads to lower atom economy, as significant amounts of reagents are wasted in side reactions or remain as difficult-to-remove impurities. The reliance on multiple reagents also increases the logistical burden on supply chain managers, who must source and quality-control a wider array of sensitive chemicals, thereby increasing the risk of production delays and batch failures.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent simplifies the entire synthetic workflow by replacing the complex catalytic ensemble with a single, cost-effective oxidant: ceric ammonium nitrate. This approach fundamentally alters the reaction dynamics, allowing for the direct oxidative coupling of readily available styrene derivatives with diphenylphosphine oxide without the need for exogenous metal catalysts. 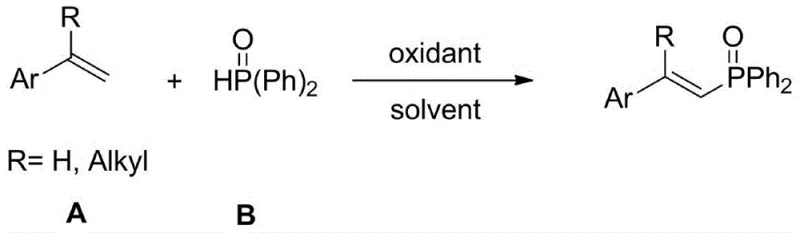 The reaction system is remarkably robust, operating effectively in common organic solvents like 1,4-dioxane, and proceeds through a controlled temperature gradient that optimizes yield and selectivity. By eliminating the need for expensive noble metals and reducing the number of additive components, this method drastically lowers the barrier to entry for manufacturing these compounds. The result is a process that is not only chemically elegant but also commercially superior, offering a clear pathway for cost reduction in pharmaceutical intermediate manufacturing while maintaining high standards of product purity and stereochemical control.
The reaction system is remarkably robust, operating effectively in common organic solvents like 1,4-dioxane, and proceeds through a controlled temperature gradient that optimizes yield and selectivity. By eliminating the need for expensive noble metals and reducing the number of additive components, this method drastically lowers the barrier to entry for manufacturing these compounds. The result is a process that is not only chemically elegant but also commercially superior, offering a clear pathway for cost reduction in pharmaceutical intermediate manufacturing while maintaining high standards of product purity and stereochemical control.
Mechanistic Insights into CAN-Mediated Oxidative Coupling
The success of this synthesis lies in the multifaceted role played by ceric ammonium nitrate (CAN), which acts as more than just a simple oxidant. The mechanism initiates with the strong oxidative power of the Ce(IV) species, which abstracts a hydrogen atom from the diphenylphosphine oxide to generate a reactive phosphorus-centered radical. This radical species then undergoes a regioselective addition to the terminal carbon of the aryl vinyl double bond, forming a stabilized benzylic carbon radical intermediate. 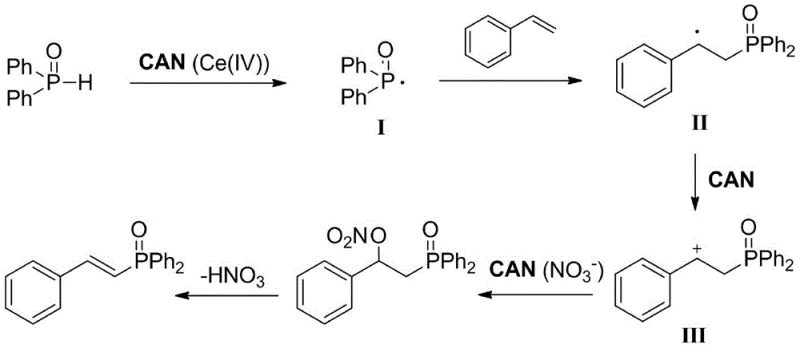 Crucially, the CAN continues to participate in the cycle by oxidizing this carbon radical into a carbocation species. This cationic intermediate is then subjected to elimination, facilitated by the nitrate ions provided by the CAN itself, ultimately yielding the desired E-alkenyl diphenylphosphine oxide with high stereoselectivity. This intricate dance of single-electron transfers and ionic eliminations ensures that the reaction proceeds with high efficiency and minimal formation of unwanted isomers. For the R&D team, understanding this mechanism is vital for troubleshooting and optimization, as it highlights the importance of maintaining the correct oxidant stoichiometry to drive the radical propagation and subsequent oxidation steps to completion without over-oxidizing the sensitive phosphine moiety.
Crucially, the CAN continues to participate in the cycle by oxidizing this carbon radical into a carbocation species. This cationic intermediate is then subjected to elimination, facilitated by the nitrate ions provided by the CAN itself, ultimately yielding the desired E-alkenyl diphenylphosphine oxide with high stereoselectivity. This intricate dance of single-electron transfers and ionic eliminations ensures that the reaction proceeds with high efficiency and minimal formation of unwanted isomers. For the R&D team, understanding this mechanism is vital for troubleshooting and optimization, as it highlights the importance of maintaining the correct oxidant stoichiometry to drive the radical propagation and subsequent oxidation steps to completion without over-oxidizing the sensitive phosphine moiety.
From an impurity control perspective, this mechanism offers distinct advantages over transition-metal catalyzed pathways. Because the reaction does not involve the formation of stable metal-phosphine complexes, the risk of catalyst poisoning or the generation of metal-bound by-products is virtually eliminated. The primary by-products are inorganic cerium salts and nitric acid derivatives, which are water-soluble and can be easily separated from the organic product during the aqueous workup phase. This inherent cleanliness of the reaction profile translates directly into higher crude purity, reducing the load on purification columns and minimizing product loss during isolation. Furthermore, the high E-selectivity observed suggests that the elimination step is highly stereospecific, likely driven by the steric bulk of the diphenylphosphine oxide group and the stability of the trans-alkene product. This level of control is essential for applications where the geometric configuration of the double bond dictates the biological activity or material properties of the final derivative.
How to Synthesize (E)-Diphenyl(styryl)phosphine Oxide Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized thermal profile and reagent ratios established in the patent data. The process begins with the careful preparation of the reaction mixture under an inert atmosphere to prevent premature oxidation of the phosphine reagent by atmospheric oxygen. The specific molar ratios of aryl vinyl derivative to diphenylphosphine oxide and CAN are critical; typically, an excess of the alkene is used to drive the reaction forward while ensuring the oxidant is present in sufficient quantity to complete the redox cycle. The two-stage heating protocol, starting at a lower temperature to initiate radical formation and ramping up to facilitate the elimination step, is key to maximizing yield. This structured approach ensures reproducibility and safety, allowing technical teams to scale the reaction from gram-scale experiments to multi-kilogram batches with predictable outcomes.
- Combine aryl vinyl derivatives and diphenylphosphine oxide in an organic solvent such as 1,4-dioxane under nitrogen protection.
- Add ceric ammonium nitrate (CAN) as the sole oxidant and heat the mixture initially at 40°C for 6 hours.
- Increase the temperature to 90°C for an additional 12 hours, then concentrate and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this CAN-mediated synthesis offers tangible strategic benefits that extend far beyond the laboratory bench. The most immediate impact is seen in the drastic simplification of the bill of materials. By removing the requirement for precious metal catalysts like palladium or silver, the direct material costs are significantly reduced, and the volatility associated with the pricing of these rare earth metals is completely avoided. Additionally, the use of ceric ammonium nitrate, a commodity chemical with stable pricing and widespread availability, ensures a secure and reliable supply chain for the core reagents. This stability allows for more accurate long-term budgeting and reduces the risk of production stoppages due to reagent shortages. The simplified reagent list also means fewer vendor qualifications and less administrative overhead in managing chemical inventory, freeing up resources to focus on other critical supply chain initiatives.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and complex ligand systems results in substantial cost savings per kilogram of product. Since the oxidant serves a dual purpose and no additional additives are required, the overall reagent consumption is minimized. Furthermore, the simplified workup procedure reduces the consumption of solvents and stationary phases during purification, lowering the operational expenditure associated with waste disposal and solvent recovery. These cumulative efficiencies contribute to a leaner manufacturing process that enhances the overall profit margin for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like styrenes and ceric ammonium nitrate mitigates the risks associated with sourcing specialized or proprietary catalysts. These starting materials are produced on a massive industrial scale, ensuring consistent quality and availability even during periods of market fluctuation. This robustness is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for downstream clients. The ability to source materials from multiple suppliers further strengthens the supply chain resilience, preventing single points of failure that could disrupt the manufacturing of critical API precursors.
- Scalability and Environmental Compliance: The reaction conditions are mild and do not require extreme pressures or temperatures, making the process inherently safer and easier to scale in standard stainless steel reactors. The absence of heavy metals simplifies the environmental compliance landscape, as there is no need for costly and rigorous heavy metal scavenging steps or specialized waste treatment for toxic metal residues. This aligns perfectly with modern green chemistry principles and regulatory requirements, facilitating smoother audits and reducing the environmental footprint of the manufacturing facility. The high atom economy also means less chemical waste is generated, supporting sustainability goals and reducing disposal costs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical aspects of the technology. Understanding these nuances helps stakeholders make informed decisions about integrating this methodology into their existing production workflows.
Q: What are the primary advantages of using Ceric Ammonium Nitrate (CAN) over traditional precious metal catalysts?
A: The use of CAN eliminates the need for expensive and toxic transition metals like Palladium, Platinum, or Silver. It acts as a multifunctional reagent that serves as both the oxidant and the nitrate source, significantly reducing reagent costs and simplifying the removal of heavy metal impurities from the final pharmaceutical intermediate.
Q: Does this method support a wide range of substrate substituents?
A: Yes, the protocol demonstrates excellent functional group tolerance. It successfully accommodates various substituted styrenes, including those with electron-donating groups like methyl and tert-butyl, as well as electron-withdrawing groups such as fluoro, chloro, and bromo, maintaining high stereoselectivity for the E-isomer.
Q: How does this process improve atom economy compared to coupling reactions?
A: Unlike traditional coupling reactions that generate stoichiometric amounts of salt by-products, this direct oxidative coupling utilizes the hydrogen atoms from the reactants efficiently. The only by-products are inorganic salts and water/nitric acid derivatives, leading to a much cleaner reaction profile and reduced waste treatment burden.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkenyl Diphenylphosphine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the CAN-mediated oxidative coupling technology for the production of high-value organophosphorus compounds. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific thermal and safety requirements of oxidative reactions, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards required for pharmaceutical applications. We are committed to delivering not just a product, but a comprehensive solution that optimizes your supply chain and reduces your time to market.
We invite you to collaborate with us to leverage this advanced synthesis for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this metal-free route can improve your bottom line. Please contact us today to request specific COA data for our alkenyl diphenylphosphine portfolio and to discuss route feasibility assessments for your custom synthesis needs. Together, we can drive innovation and efficiency in the production of next-generation chemical intermediates.
