Advanced Purification Technology for Lesinurad: Ensuring Clinical Grade Purity and Scalability
Introduction to Advanced Lesinurad Refining Technologies
The global pharmaceutical landscape for gout management has been significantly reshaped by the introduction of URAT1 inhibitors, with Lesinurad standing out as a pivotal therapeutic agent. However, the commercial viability of this active pharmaceutical ingredient (API) hinges critically on the ability to manufacture it with exceptional purity profiles that meet stringent regulatory standards. Patent CN111153862B addresses a critical bottleneck in the supply chain by disclosing a robust refining method that transforms crude Lesinurad into a high-purity finished product suitable for clinical application. This innovation is not merely a laboratory curiosity but represents a substantial leap forward in process chemistry, offering a reliable pathway for manufacturers to overcome the persistent challenges of impurity control. By leveraging specific solvent systems and controlled crystallization dynamics, this technology ensures that the final API is free from genotoxic alerts and structural analogs that could compromise patient safety.
For procurement leaders and supply chain directors, the implications of this patent extend far beyond simple chemical optimization. The ability to consistently produce Lesinurad with purity levels exceeding 99.9% directly correlates to reduced batch rejection rates and streamlined regulatory filings. In an era where supply chain resilience is paramount, adopting a purification strategy that relies on common, non-proprietary solvents mitigates the risk of raw material shortages. This report delves deep into the technical nuances of Patent CN111153862B, analyzing how this refining methodology serves as a cornerstone for establishing a dependable Lesinurad supply chain. We will explore the mechanistic underpinnings of the purification process, contrast it with legacy methods, and outline the tangible commercial advantages it offers to forward-thinking pharmaceutical enterprises seeking a competitive edge in the anti-gout medication market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
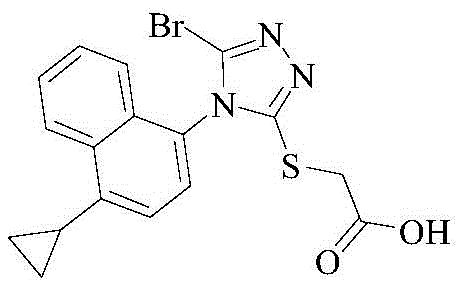
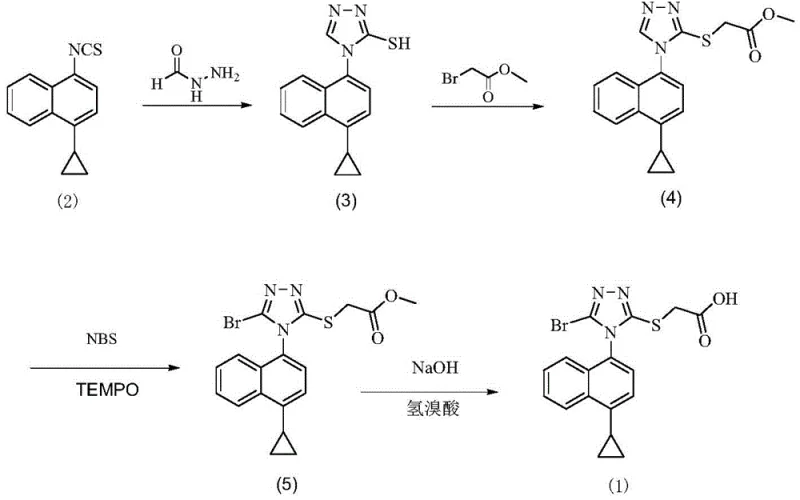
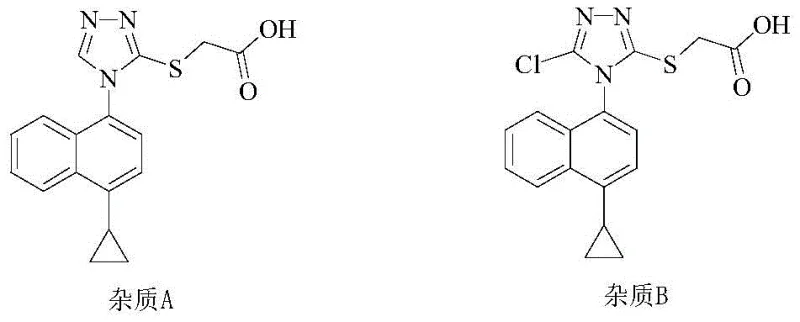
To fully appreciate the breakthrough offered by the new refining method, one must first understand the inherent deficiencies in the prior art synthesis routes, specifically those outlined in patent CN104736522A. The traditional synthetic pathway involves a multi-step sequence culminating in a bromination reaction using N-bromosuccinimide (NBS). While chemically effective for constructing the core scaffold, this approach invariably introduces stubborn impurities that resist removal through standard workup procedures. The most problematic contaminants are identified as Impurity A, which arises from the hydrolysis of residual intermediate compounds, and Impurity B, a chlorinated byproduct resulting from chloride contamination within the NBS reagent. These impurities are structurally similar to the target molecule, making their separation via conventional washing or simple recrystallization exceptionally difficult and often inefficient.
The presence of these impurities poses a severe threat to the commercial feasibility of the manufacturing process. From a regulatory perspective, the accumulation of halogenated impurities like Impurity B can trigger extensive toxicological studies, delaying time-to-market and inflating development costs. Furthermore, the variability in NBS quality from different suppliers can lead to inconsistent impurity profiles across batches, creating a nightmare for quality assurance teams striving for reproducibility. The legacy methods often require repeated recrystallizations or expensive preparative chromatography to achieve acceptable purity, which drastically reduces overall yield and increases the environmental footprint of the process. This inefficiency creates a fragile supply chain where small deviations in reaction conditions can render entire batches unusable, highlighting the urgent need for a more robust downstream processing solution.
The Novel Approach
Patent CN111153862B introduces a paradigm shift in downstream processing by focusing on a highly optimized recrystallization protocol that effectively purges these recalcitrant impurities in a single operational sequence. Instead of relying on complex chemical transformations to fix the impurity profile, this method leverages the subtle differences in solubility and crystal lattice energy between Lesinurad and its contaminants. The process utilizes a strategic combination of a primary solvent, such as ethyl acetate or dichloromethane, and an anti-solvent, specifically n-hexane. By dissolving the crude material at elevated temperatures and subsequently introducing the anti-solvent under controlled conditions, the system creates a supersaturated environment that favors the nucleation of pure Lesinurad crystals while keeping the impurities in the mother liquor. This thermodynamic selectivity is the key to achieving purity levels that were previously unattainable with standard techniques.
Moreover, the inclusion of an activated carbon treatment step prior to crystallization adds an additional layer of purification, effectively adsorbing colored bodies and trace organic particulates that could act as nucleation sites for impure crystals. The result is a finished product that not only meets but exceeds the rigorous purity specifications required for global pharmaceutical markets. The method is remarkably versatile, accommodating various solvent ratios to fine-tune the crystallization kinetics based on the specific impurity load of the incoming crude material. This flexibility ensures that even batches with higher initial impurity concentrations can be salvaged and upgraded to premium grade, significantly enhancing the overall material throughput and economic efficiency of the manufacturing plant. It represents a turnkey solution for transforming a chemically complex crude mixture into a pristine API ready for formulation.
Mechanistic Insights into Solvent-Mediated Recrystallization
The efficacy of the refining method described in Patent CN111153862B is rooted in the precise manipulation of solubility parameters and crystallization kinetics. The core mechanism relies on the principle that the target molecule, Lesinurad, exhibits a distinct solubility curve in the chosen solvent system compared to its structural analogs. By heating the crude product in a solvent like ethyl acetate to a temperature range of 35-80°C, the process ensures complete dissolution of the API while leaving behind insoluble particulate matter. The subsequent addition of activated carbon serves a dual purpose: it decolorizes the solution by adsorbing conjugated organic impurities and provides a clean surface that minimizes heterogeneous nucleation of unwanted phases. This step is critical for ensuring that the subsequent crystal growth is driven purely by the thermodynamics of the Lesinurad molecule itself, rather than being seeded by foreign particles.
The addition of n-hexane as an anti-solvent is the pivotal moment in the process, fundamentally altering the polarity of the medium. As the volume fraction of the non-polar hexane increases, the solubility of the polar Lesinurad molecule decreases sharply, driving the system into a state of supersaturation. However, the patent specifies a controlled cooling regimen—first to 35-45°C and then to 0-10°C—which is essential for controlling the rate of nucleation versus crystal growth. Rapid cooling would lead to the precipitation of amorphous material or the trapping of impurities within the crystal lattice (occlusion). In contrast, the slow, staged cooling allows for the orderly arrangement of molecules into a stable crystal lattice, effectively excluding the structurally distinct Impurity A and Impurity B. This kinetic control ensures that the impurities remain dissolved in the supernatant, allowing for their physical separation via filtration, thereby delivering a product of exceptional chemical and physical integrity.
How to Synthesize Lesinurad Efficiently
The implementation of this purification technology requires a disciplined approach to process parameters to ensure consistent results across different scales of operation. The patent outlines a clear protocol that balances solvent volumes, temperature gradients, and agitation rates to maximize yield and purity. For R&D teams looking to replicate or adapt this process, understanding the interplay between the solvent ratio and the cooling rate is paramount. The following guide summarizes the critical operational steps derived from the patent examples, providing a foundational framework for process development. It is important to note that while the specific ratios provided are optimal for the disclosed examples, slight adjustments may be necessary depending on the specific impurity profile of the crude starting material sourced from different synthetic routes.
- Dissolve crude Lesinurad in a heated organic solvent such as ethyl acetate or dichloromethane.
- Add activated carbon to adsorb colored impurities and filter the hot solution.
- Dropwise add n-hexane as an anti-solvent and control cooling rates to crystallize high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this refining technology translates directly into enhanced operational stability and significant cost optimization opportunities. The primary advantage lies in the drastic simplification of the purification workflow. Unlike legacy methods that might require multiple recrystallization cycles or expensive chromatographic separations to remove chlorinated impurities, this method achieves high purity in a single pass. This reduction in unit operations directly correlates to lower utility consumption, reduced labor hours, and decreased solvent usage per kilogram of finished product. Furthermore, the solvents employed—ethyl acetate, n-hexane, and isopropanol—are commodity chemicals with stable global supply chains, insulating the manufacturing process from the volatility associated with exotic or specialized reagents. This accessibility ensures that production schedules remain uninterrupted, even during periods of broader chemical market fluctuation.
From a yield perspective, the method demonstrates remarkable efficiency in recovering the target API from the crude matrix. High recovery rates mean that less raw material is wasted in the mother liquor, effectively lowering the cost of goods sold (COGS) for the final API. The robustness of the process also implies a higher success rate for batch production, reducing the financial risk associated with batch failures and re-processing. Additionally, the elimination of complex purification steps reduces the generation of hazardous waste, simplifying environmental compliance and disposal costs. For supply chain planners, this translates to a more predictable and reliable production timeline, enabling better inventory management and the ability to respond swiftly to market demand surges without the fear of quality-related bottlenecks. The scalability of the process ensures that these benefits are retained whether producing pilot batches or multi-ton commercial campaigns.
- Cost Reduction in Manufacturing: The elimination of expensive chromatographic purification steps and the reduction in solvent consumption per batch lead to substantial operational expenditure savings. By utilizing common, low-cost solvents and minimizing the number of processing stages, the overall manufacturing overhead is significantly decreased, allowing for more competitive pricing strategies in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: The reliance on widely available commodity solvents mitigates the risk of supply disruptions caused by raw material shortages. The robustness of the purification protocol ensures consistent output quality regardless of minor variations in crude input, providing a stable and predictable supply of high-purity Lesinurad to downstream formulation partners.
- Scalability and Environmental Compliance: The process is designed for seamless scale-up from laboratory to industrial reactors without the need for specialized equipment. The simplified waste stream, characterized by common organic solvents, facilitates easier recycling and treatment, aligning with modern green chemistry initiatives and reducing the environmental compliance burden on the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. These insights are derived directly from the experimental data and claims presented in Patent CN111153862B, offering clarity on the method's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into their existing manufacturing infrastructure. The answers provided reflect the specific advantages of the solvent-system approach over traditional purification techniques.
Q: What are the primary impurities found in conventional Lesinurad synthesis?
A: Conventional synthesis often leaves behind hydrolyzed precursor residues (Impurity A) and chlorinated byproducts generated during NBS bromination (Impurity B), which are difficult to remove without specialized purification.
Q: How does the patented recrystallization method improve yield?
A: By optimizing the solvent-to-anti-solvent ratio and implementing a staged cooling protocol, the method maximizes crystal recovery while maintaining a purity profile exceeding 99.9%.
Q: Is this purification method suitable for industrial scale-up?
A: Yes, the process utilizes common industrial solvents like ethyl acetate and n-hexane and relies on standard filtration and drying equipment, making it highly scalable for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lesinurad Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires more than just chemical knowledge; it demands engineering excellence and unwavering commitment to quality. As a premier CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated purification protocols described in CN111153862B are executed with precision. Our facilities are equipped with state-of-the-art crystallization and filtration units capable of handling the specific solvent systems required for Lesinurad refinement. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Lesinurad we produce meets the highest international pharmacopoeial standards, effectively eliminating the risks associated with Impurity A and Impurity B.
We invite pharmaceutical companies and procurement specialists to collaborate with us to leverage this advanced purification technology for their supply chains. Our team is prepared to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this refined manufacturing route for your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data from our recent pilot runs and to discuss detailed route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you secure not just a supplier, but a strategic ally dedicated to optimizing your API sourcing strategy through innovation and operational excellence.
