Optimizing Oxybuprocaine Hydrochloride Production: A Novel 4-Step Industrial Route
Optimizing Oxybuprocaine Hydrochloride Production: A Novel 4-Step Industrial Route
The pharmaceutical landscape for local anesthetics is constantly evolving, driven by the need for safer, more efficient manufacturing processes that comply with stringent regulatory standards. Patent CN113185419B introduces a significant advancement in the synthesis of oxybuprocaine hydrochloride, a critical benzoate ester used extensively in ophthalmology and otolaryngology for surface anesthesia. This innovative methodology addresses long-standing challenges associated with genotoxic impurities and complex equipment requirements found in legacy production routes. By streamlining the synthesis into a concise four-step sequence, the patent offers a robust framework for producing high-purity intermediates suitable for commercial drug substance manufacturing. The structural integrity of the final product, as depicted below, is maintained through mild reaction conditions that preserve the delicate ester linkage while ensuring complete conversion of the nitro group.
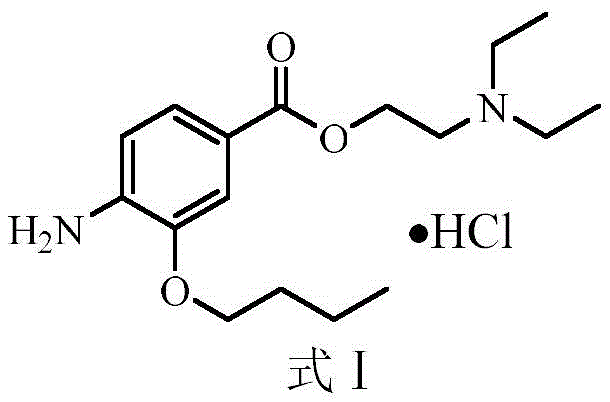
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of oxybuprocaine hydrochloride has been plagued by safety hazards and supply chain vulnerabilities inherent to older synthetic strategies. For instance, the route disclosed in Chinese patent CN106810463A relies heavily on diethylaminoethyl chloride, a monitored chemical that is difficult to procure and tightly regulated due to its potential misuse. Furthermore, this conventional pathway necessitates the use of N,N-dimethylformamide (DMF) as a reaction solvent, which poses a severe risk of generating N-nitrosodimethylamine (NDMA), a potent genotoxic impurity that is strictly controlled by global health authorities. The reliance on hydrazine hydrate for the final reduction step further exacerbates safety concerns, introducing another genotoxic hazard that requires extensive and costly purification protocols to remove from the final active pharmaceutical ingredient. These factors collectively render traditional methods unsuitable for modern, compliant large-scale manufacturing.
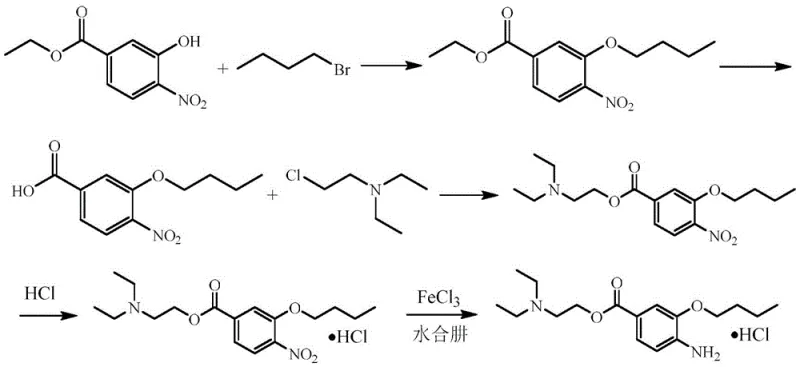
The Novel Approach
In stark contrast to these problematic legacy methods, the synthesis method described in CN113185419B offers a streamlined, four-step pathway that prioritizes safety and operational simplicity without compromising yield. The process initiates with a straightforward etherification using readily available 1-bromobutane, followed by a transesterification that cleverly integrates salt formation in a single operational sequence. This integration reduces the overall number of isolation steps, thereby minimizing material loss and solvent consumption. Crucially, the reduction of the nitro group is achieved using iron powder in an aqueous medium, a classic yet highly effective green chemistry technique that avoids the need for high-pressure hydrogenation equipment or pyrophoric catalysts. The final salification is conducted under controlled low-temperature conditions to ensure optimal crystal morphology and purity, resulting in a process that is not only chemically efficient but also economically superior for industrial application.
Mechanistic Insights into Iron-Mediated Reduction and Transesterification
The core of this synthetic innovation lies in the strategic selection of reagents that facilitate high conversion rates while mitigating side reactions. In the transesterification step, the use of sodium hydroxide as a catalyst in n-heptane creates a heterogeneous environment that drives the equilibrium towards the formation of the desired ester bond between the benzoate intermediate and 2-(diethylamino)ethanol. The choice of n-heptane is particularly astute, as it allows for easy phase separation during workup and avoids the solubility issues often encountered with polar aprotic solvents. Following this, the reduction mechanism utilizes iron powder in water at 80°C, where the iron acts as an electron donor to reduce the nitro group to an amine. This aqueous-based reduction is inherently safer than organic solvent-based reductions and generates iron oxide byproducts that are easily filtered off, leaving a clean organic phase for subsequent processing. The entire sequence is designed to minimize the formation of des-alkylated byproducts, ensuring a clean impurity profile.
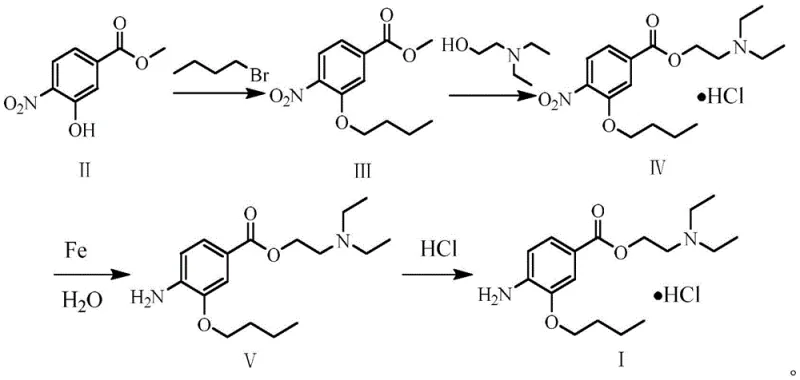
Furthermore, the control of impurities is rigorously managed through the specific pH adjustments and solvent choices detailed in the patent. During the transesterification and subsequent salification, adjusting the pH to 2-3 ensures the precipitation of the intermediate hydrochloride salt, effectively purifying the compound before the reduction step. This intermediate isolation is critical for removing unreacted starting materials and preventing them from carrying over into the final reduction stage, where they could form difficult-to-remove impurities. The final crystallization from butanone at sub-zero temperatures (-5 to 0°C) further refines the product quality by excluding soluble impurities from the crystal lattice. This multi-stage purification strategy embedded within the synthesis route demonstrates a deep understanding of process chemistry, ensuring that the final oxybuprocaine hydrochloride meets the stringent purity specifications required for ophthalmic applications.
How to Synthesize Oxybuprocaine Hydrochloride Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and safety. The process begins with the etherification of methyl 3-hydroxy-4-nitrobenzoate, where maintaining the temperature at 70°C in butanone is essential for optimal kinetics. Subsequent steps involve careful stoichiometry, particularly in the transesterification where a slight excess of the amino alcohol drives the reaction to completion. The reduction step demands attention to the particle size of the iron powder to ensure sufficient surface area for reaction without creating filtration difficulties. Finally, the introduction of hydrogen chloride gas must be carefully monitored to prevent local overheating and ensure uniform salt formation. For a detailed breakdown of the specific molar ratios, solvent volumes, and operational timelines required to replicate this high-yielding process, please refer to the standardized synthesis guide below.
- Perform etherification of methyl 3-hydroxy-4-nitrobenzoate with 1-bromobutane using potassium carbonate in butanone at 70°C.
- Conduct transesterification with 2-(diethylamino)ethanol using sodium hydroxide catalyst in n-heptane, followed by salification with hydrochloric acid.
- Reduce the nitro group using iron powder in water at 80°C to obtain the free base oxybuprocaine.
- Finalize the process by reacting the free base with hydrogen chloride gas in butanone at -5 to 0°C to crystallize the hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this novel synthesis route offers transformative advantages that directly impact the bottom line and operational resilience. By eliminating the dependence on monitored precursors like diethylaminoethyl chloride, manufacturers can bypass complex regulatory hurdles and lengthy lead times associated with controlled substance procurement. The substitution of hazardous solvents like DMF with common hydrocarbons like n-heptane and ketones like butanone significantly reduces waste disposal costs and environmental compliance burdens. Moreover, the avoidance of high-pressure hydrogenation equipment lowers the barrier to entry for contract manufacturing organizations, allowing for more flexible and distributed production networks. These factors combine to create a supply chain that is not only more cost-effective but also far more robust against regulatory disruptions and raw material shortages.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the reaction sequence and the use of commodity-grade reagents. By integrating the ester exchange and salt formation into a single operational step, the process reduces labor hours, energy consumption, and solvent usage compared to multi-step legacy routes. The elimination of expensive noble metal catalysts in favor of inexpensive iron powder for reduction drastically lowers the cost of goods sold (COGS). Additionally, the high yields reported in the patent examples indicate minimal raw material waste, further enhancing the overall cost efficiency of the manufacturing process.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals such as 1-bromobutane, potassium carbonate, and sodium hydroxide ensures a stable and predictable supply chain. Unlike specialized reagents that may be sourced from a single vendor, these commodities are produced globally, mitigating the risk of supply disruptions due to geopolitical issues or manufacturer outages. The robustness of the chemistry also means that the process is less sensitive to minor variations in raw material quality, allowing procurement teams to source from a broader range of qualified suppliers without compromising product consistency.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard stirred-tank reactors and filtration equipment found in most multipurpose pharmaceutical facilities. The absence of high-pressure operations and pyrophoric reagents simplifies the safety validation process, accelerating the timeline from pilot plant to commercial production. Environmentally, the use of aqueous reduction and recyclable organic solvents aligns with green chemistry principles, reducing the facility's carbon footprint and simplifying the permitting process for wastewater treatment. This alignment with sustainability goals is increasingly important for securing contracts with major pharmaceutical companies committed to environmental stewardship.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. They are derived from a detailed analysis of the patent's experimental data and comparative advantages over prior art. Understanding these nuances is crucial for R&D directors and process engineers evaluating the feasibility of technology transfer. The answers provided reflect the specific conditions and outcomes documented in the patent literature, offering a realistic expectation of performance.
Q: How does this new synthesis route address genotoxic impurity concerns?
A: Unlike prior art methods that utilize N,N-dimethylformamide (DMF) and hydrazine hydrate, this novel process employs n-heptane and iron powder reduction. This substitution completely eliminates the risk of introducing N-nitrosodimethylamine (NDMA) and hydrazine residues, ensuring a safer impurity profile for the final API.
Q: What are the equipment requirements for scaling this process?
A: The process is designed for standard industrial equipment, operating at atmospheric pressure and moderate temperatures (max 90°C). It avoids the need for high-pressure hydrogenation reactors or specialized corrosion-resistant vessels required for thionyl chloride processes, significantly lowering capital expenditure.
Q: Is the raw material supply chain stable for this method?
A: Yes, the route relies on commodity chemicals such as 1-bromobutane, methyl 3-hydroxy-4-nitrobenzoate, and iron powder. These materials are widely available globally, unlike monitored precursors like diethylaminoethyl chloride used in older patents, ensuring robust supply chain continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxybuprocaine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent examples are faithfully reproduced at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to monitor every critical quality attribute, from residual solvents to genotoxic impurities. Our dedication to process optimization means we can adapt this novel route to fit your specific capacity and timeline requirements while maintaining full regulatory compliance.
We invite you to collaborate with us to leverage this advanced synthesis technology for your oxybuprocaine hydrochloride needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data from our pilot batches and comprehensive route feasibility assessments to demonstrate how this method can enhance your supply chain efficiency and product quality.
