Revolutionizing D,L-Pantolactone Production: A Green, Telescoped Synthetic Route for Global Vitamin B5 Supply Chains
Introduction to Advanced D,L-Pantolactone Synthesis
The global demand for Vitamin B5 (D-calcium pantothenate) continues to surge across the pharmaceutical, animal nutrition, and cosmetic sectors, driving an urgent need for more efficient and sustainable synthetic routes for its key chiral precursor, D,L-pantolactone. Traditional manufacturing pathways have long been plagued by excessive wastewater generation, high energy consumption, and reliance on expensive raw materials like glyoxylic acid. However, a groundbreaking technological advancement detailed in patent CN110498781B introduces a novel, green synthesis method that fundamentally restructures the production landscape. This innovative approach leverages an alcohol-based solvent system to facilitate a telescoped aldol condensation and cyanohydrin reaction, effectively bypassing the cumbersome isolation and extraction steps that have historically burdened the industry. By integrating the reaction and purification stages, this method not only achieves exceptional product purity exceeding 98.5% but also aligns perfectly with modern green chemistry principles, offering a compelling solution for manufacturers seeking to optimize their operational expenditures and environmental footprint simultaneously.
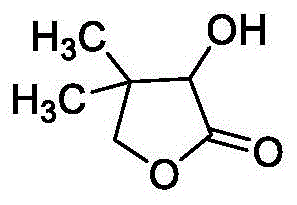
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of D,L-pantolactone has relied heavily on three primary routes, each carrying significant logistical and economic burdens that hinder optimal supply chain efficiency. The earliest method involving glyoxylic acid and isobutyraldehyde suffers from the prohibitive cost of glyoxylic acid and generates massive quantities of inorganic salt byproducts, necessitating complex wastewater treatment infrastructure to handle the high salinity effluent. Another common pathway utilizes hydroxyacetonitrile, which is chemically unstable under alkaline conditions, leading to polymerization side reactions that darken the reaction mass and complicate downstream purification. Perhaps the most widely used conventional technique involves the condensation of formaldehyde and isobutyraldehyde in an aqueous medium followed by cyanidation; however, this process is severely limited by the poor water solubility of the intermediate hydroxypivalaldehyde. To overcome this, manufacturers are forced to use vast amounts of water and energy for evaporation, followed by extraction with approximately 5 tons of ethyl acetate per ton of product. This reliance on organic extraction not only inflates raw material costs due to solvent loss but also creates a hazardous waste stream that is difficult and expensive to treat, creating a bottleneck for scalable and compliant manufacturing.
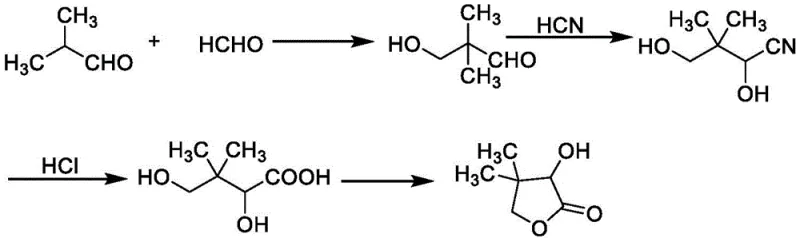
The Novel Approach
In stark contrast to these legacy processes, the method disclosed in patent CN110498781B introduces a paradigm shift by replacing the aqueous reaction medium with an alcohol solvent system, such as methanol, ethanol, or n-butanol. This strategic substitution dramatically enhances the solubility of the intermediate hydroxypivalaldehyde, allowing the aldol condensation product to remain in solution and proceed directly to the cyanohydrin reaction without any intermediate isolation or phase separation. This telescoping of steps eliminates the need for the energy-intensive evaporation of water and completely removes the requirement for organic solvent extraction with ethyl acetate. Instead, the process utilizes the alcohol solvent as a recyclable medium, which is recovered via distillation in the final stage. The reaction sequence culminates in an acid-catalyzed lactonization followed by neutralization with ammonia, which precipitates ammonium salts that are easily filtered off. This streamlined workflow not only simplifies the equipment requirements but also ensures a high-yield transformation with minimal waste generation, representing a significant leap forward in process intensification for fine chemical intermediates.
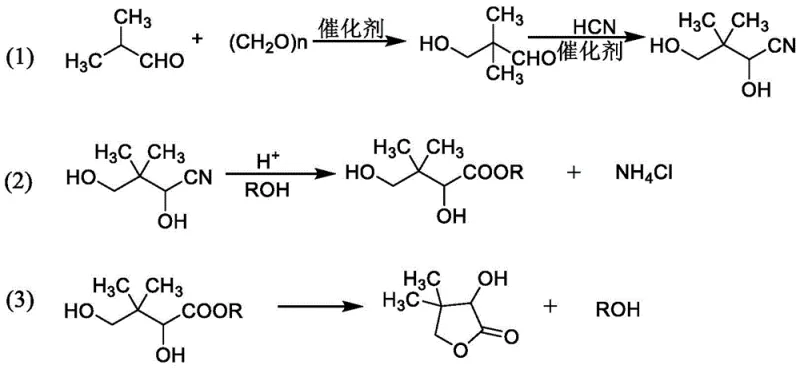
Mechanistic Insights into Alcohol-Mediated Telescoped Synthesis
The core innovation of this synthesis lies in the mechanistic advantages provided by the alcoholic medium during the initial aldol condensation between paraformaldehyde and isobutyraldehyde. In traditional aqueous systems, the equilibrium often favors the reactants or leads to hydration byproducts due to the polarity of water, whereas the alcohol solvent stabilizes the transition state and solubilizes the resulting hydroxypivalaldehyde effectively. This homogeneous environment ensures that when hydrocyanic acid is introduced in the subsequent step, the cyanohydrin reaction proceeds rapidly and uniformly throughout the bulk liquid, minimizing localized concentration gradients that could lead to oligomerization or decomposition. The use of a basic catalyst, which can range from organic amines like triethylamine to inorganic bases like potassium hydroxide, further accelerates the deprotonation steps necessary for carbon-carbon bond formation. Following the cyanohydrin formation, the introduction of strong acid catalyzes the hydrolysis of the nitrile group and the simultaneous intramolecular esterification (lactonization) to form the five-membered lactone ring. Crucially, the final neutralization with ammonia rather than sodium hydroxide prevents the formation of non-volatile sodium salts, instead generating ammonium chloride or sulfate which can be physically removed by filtration, leaving the organic phase clean for final distillation.
From an impurity control perspective, this mechanism offers superior selectivity by avoiding the harsh thermal conditions often required to drive aqueous reactions to completion. The avoidance of high-temperature water evaporation reduces the risk of thermal degradation of the sensitive lactone ring, which can otherwise open up or polymerize under stress. Furthermore, the direct use of the reaction liquid without workup minimizes exposure to air and moisture, reducing oxidative impurities that typically contribute to product discoloration. The precipitation of ammonium salts serves as an effective purification step, scavenging residual acid and ionic impurities from the organic matrix before the final rectification. This results in a crude product with significantly higher purity compared to extraction-based methods, where trace amounts of extraction solvent and emulsified water often persist. The robustness of this catalytic cycle ensures consistent batch-to-batch reproducibility, a critical factor for regulatory compliance in pharmaceutical intermediate manufacturing where impurity profiles must be tightly controlled to meet stringent pharmacopoeia standards.
How to Synthesize D,L-Pantolactone Efficiently
The implementation of this advanced synthetic route requires precise control over reaction parameters to maximize the benefits of the telescoped design. The process begins with the careful addition of isobutyraldehyde to a mixture of paraformaldehyde and alcohol solvent under basic catalysis, maintaining temperatures around 40°C to 94°C depending on the specific alcohol used to ensure complete condensation. Once the hydroxypivalaldehyde is formed, the reaction mixture is cooled slightly before the controlled addition of hydrocyanic acid, a step that demands rigorous safety protocols due to the toxicity of HCN. Following the cyanohydrin formation, the mixture is subjected to acidic reflux to drive lactonization, after which ammonia is introduced to neutralize the system. The resulting slurry is filtered to remove ammonium salts, and the filtrate is distilled to recover the alcohol solvent for reuse, leaving behind the crude lactone which is finally rectified to achieve pharmaceutical grade purity. For detailed operational parameters, safety guidelines, and specific stoichiometric ratios, please refer to the standardized synthesis protocol below.
- Perform aldol condensation between paraformaldehyde and isobutyraldehyde in an alcohol solvent (methanol, ethanol, or butanol) using a basic catalyst, utilizing the crude reaction liquid directly for the next step.
- Add hydrocyanic acid to the reaction mixture to generate 2,4-dihydroxy-3,3-dimethylbutyronitrile, followed by acidic esterification and hydrolysis to induce lactonization.
- Neutralize excess acid with ammonia to precipitate ammonium salts, filter the byproducts, recover the alcohol solvent via distillation, and rectify the residue to obtain high-purity D,L-pantolactone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this alcohol-based synthesis route presents a transformative opportunity to drastically reduce manufacturing costs and mitigate supply risks associated with traditional methods. The elimination of the organic solvent extraction step is perhaps the most significant economic driver; by removing the need for thousands of liters of ethyl acetate per ton of product, companies can achieve substantial cost savings on raw material procurement and solvent recovery infrastructure. Additionally, the ability to recycle the alcohol reaction solvent directly within the process loop creates a closed-loop system that minimizes waste disposal fees and reduces the overall consumption of volatile organic compounds. This efficiency translates directly into a lower cost of goods sold (COGS), providing a competitive pricing advantage in the global market for Vitamin B5 intermediates. Furthermore, the simplified process flow reduces the number of unit operations, which in turn lowers labor costs and decreases the likelihood of operational errors or batch failures, ensuring a more reliable and consistent supply of high-purity material for downstream customers.
- Cost Reduction in Manufacturing: The structural simplification of the process eliminates the capital and operational expenditures associated with large-scale liquid-liquid extraction units and the subsequent solvent recovery columns required for ethyl acetate. By utilizing the reaction solvent as the crystallization and distillation medium, the energy load for heating and cooling is significantly optimized, as there is no need to evaporate large volumes of water or separate immiscible phases. The switch to ammonia for neutralization also reduces the cost of acid and base consumption compared to the sodium hydroxide/hydrochloric acid cycles used in older methods, while the resulting ammonium salt byproducts are easier and cheaper to handle than massive quantities of sodium sulfate waste. These cumulative efficiencies result in a leaner manufacturing process that delivers significant margin improvements without compromising on product quality or yield.
- Enhanced Supply Chain Reliability: Dependence on complex multi-step extractions often introduces bottlenecks that can delay production schedules and extend lead times, particularly when solvent quality or availability fluctuates. This novel route mitigates such risks by relying on commodity chemicals like methanol or ethanol and paraformaldehyde, which are widely available and stable in the global market. The robustness of the telescoped reaction means that production cycles are shorter and more predictable, allowing manufacturers to respond more agilely to spikes in demand for D-calcium pantothenate. Moreover, the reduced generation of hazardous wastewater simplifies environmental compliance, reducing the risk of regulatory shutdowns or fines that could disrupt the supply chain. This stability makes the manufacturer a more dependable partner for long-term contracts with major pharmaceutical and nutraceutical companies.
- Scalability and Environmental Compliance: As global regulations on industrial emissions and wastewater discharge become increasingly stringent, the environmental profile of a chemical process is a critical factor in its long-term viability. This synthesis method inherently aligns with green chemistry principles by preventing waste at the source rather than treating it post-generation. The absence of ethyl acetate emissions and the reduction in saline wastewater load make it easier to obtain and maintain environmental permits, facilitating smoother capacity expansions. The process is inherently scalable because it avoids the physical limitations of extraction columns, which can be difficult to scale up linearly; instead, the reaction and distillation steps scale predictably with reactor volume. This scalability ensures that suppliers can meet growing global demand for Vitamin B5 precursors without facing the diminishing returns often associated with scaling up complex, multi-phase separation processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis technology. These insights are derived directly from the experimental data and process descriptions provided in the patent literature, offering a transparent view into the feasibility and advantages of adopting this route for industrial production. Understanding these details is crucial for technical teams evaluating the transition from legacy aqueous processes to this more efficient alcohol-based methodology.
Q: How does the alcohol-based solvent system improve yield compared to traditional aqueous methods?
A: The use of alcohol solvents like methanol or ethanol significantly improves the solubility of intermediate hydroxypivalaldehyde, eliminating the need for large volumes of water and subsequent energy-intensive evaporation steps required in aqueous processes. This homogeneous reaction environment facilitates higher conversion rates and allows for a telescoped process where the intermediate is not isolated, thereby minimizing mechanical losses and boosting overall yield to over 90%.
Q: What are the environmental benefits of replacing ethyl acetate extraction with this new method?
A: Traditional methods often require massive amounts of organic solvents like ethyl acetate (up to 5 tons per ton of product) for extraction, leading to significant solvent loss and difficult wastewater treatment due to solvent solubility in water. The patented method eliminates the extraction step entirely by utilizing the reaction solvent for crystallization and distillation, drastically reducing volatile organic compound (VOC) emissions and simplifying wastewater treatment protocols.
Q: Can this synthesis route be scaled for commercial production of Vitamin B5 precursors?
A: Yes, the process is highly scalable due to its simplified unit operations. By removing the complex extraction and multiple separation stages found in glyoxylic acid or hydroxyacetonitrile routes, the equipment footprint is reduced. Furthermore, the ability to recycle the alcohol solvent directly within the process loop enhances economic viability and supply chain stability for large-scale manufacturing of D-calcium pantothenate precursors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D,L-Pantolactone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the shift towards greener, more efficient synthetic routes is not just a regulatory necessity but a strategic imperative for maintaining competitiveness in the fine chemical sector. Our technical team has extensively analyzed the potential of the alcohol-based telescoped synthesis described in CN110498781B and possesses the expertise to implement this technology at an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of high yield and low waste are realized in actual manufacturing operations. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of D,L-pantolactone meets the exacting standards required for Vitamin B5 synthesis. By leveraging our CDMO capabilities, clients can accelerate their time-to-market while benefiting from a supply chain that is both cost-effective and environmentally responsible.
We invite procurement leaders and R&D directors to engage with us to explore how this advanced synthesis route can be tailored to your specific production needs. Whether you require a Customized Cost-Saving Analysis to quantify the potential reductions in OPEX or need to evaluate the technical feasibility of integrating this intermediate into your existing value chain, our technical procurement team is ready to assist. Please contact us to request specific COA data from our pilot runs or to discuss route feasibility assessments that can help secure your supply of high-purity D,L-pantolactone for the future.
