Advanced Fermentation-Based Synthesis of Thibetolide for Commercial Fragrance Manufacturing
The global demand for high-quality macrocyclic musks in the fine fragrance and personal care sectors continues to drive innovation in synthetic methodologies, particularly those that balance ecological sustainability with economic efficiency. Patent CN101486704A introduces a robust synthetic method for Thibetolide (also known as Muscolide), a valuable 15-membered lactone renowned for its warm, musky odor and excellent fixative properties. This technology leverages 1,15-pentadecanedioic acid, produced via biological fermentation, as the foundational raw material, marking a significant departure from traditional petrochemical-dependent routes. By integrating esterification, selective hydrolysis, reduction, and cyclization steps, this process achieves a streamlined production flow that addresses the critical pain points of yield stability and environmental impact. For R&D directors and procurement specialists seeking a reliable fragrance intermediate supplier, understanding the mechanistic advantages of this bio-based approach is essential for securing long-term supply chain resilience.
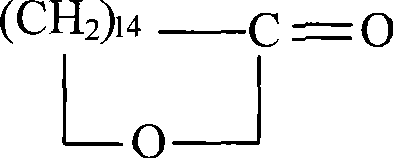
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of macrocyclic musks like Thibetolide has been plagued by complex multi-step sequences that rely heavily on expensive transition metal catalysts and harsh reaction conditions. Traditional routes often involve the use of acetylenic compounds or long-chain halogenated precursors that require rigorous purification to remove trace metallic impurities, which can be detrimental to the olfactory profile of the final fragrance. Furthermore, many existing industrial methods suffer from low atom economy and generate substantial quantities of hazardous waste, leading to increased disposal costs and regulatory scrutiny. The reliance on scarce raw materials, such as specific unsaturated fatty acids or complex organometallic reagents, also introduces volatility into the supply chain, making cost reduction in flavor & fragrance manufacturing difficult to achieve consistently. Additionally, the cyclization steps in older methodologies often require high dilution techniques to prevent polymerization, which drastically reduces volumetric productivity and increases solvent recovery burdens.
The Novel Approach
In contrast, the methodology described in the patent utilizes a fermentation-derived dicarboxylic acid to create a more direct and environmentally benign pathway to the target lactone. This novel approach simplifies the synthetic architecture by employing a sequence of esterification, partial hydrolysis, and borohydride reduction that avoids the need for precious metal catalysts entirely. The process operates under moderate thermal conditions, typically ranging from ambient temperature to 150°C, which significantly lowers energy consumption compared to high-temperature pyrolysis or high-pressure hydrogenation methods used in legacy processes. By starting with a bio-based feedstock, the manufacturer gains access to a renewable carbon source that is less susceptible to the price fluctuations of the crude oil market. The final cyclization step is optimized to favor intramolecular ring closure over intermolecular polymerization, thereby enhancing the overall yield and reducing the formation of linear oligomers that complicate downstream purification.
Mechanistic Insights into Bio-based Macrolactonization
The core of this synthetic strategy lies in the precise control of functional group transformations, beginning with the selective mono-esterification of the 1,15-pentadecanedioic acid. The process initiates by converting the diacid into a diester using a solid acid catalyst, which facilitates easy separation and reuse, followed by a controlled saponification to generate the monoester salt. This intermediate is crucial as it sets the stage for the subsequent reduction phase, where borohydride reagents selectively reduce the ester functionality to a primary alcohol while preserving the carboxylic acid moiety after acidification. The resulting 15-hydroxypentadecanoic acid serves as the direct precursor for the macrocyclization, ensuring that the carbon chain length is perfectly preserved for the formation of the 15-membered ring. This level of chemoselectivity is vital for maintaining high-purity macrocyclic musks standards, as it minimizes the generation of side products that could alter the scent profile.
Following the formation of the hydroxy acid, the mechanism proceeds through a bromination step to activate the hydroxyl group, converting it into a good leaving group for the final nucleophilic substitution. The cyclization is executed under basic conditions using carbonate salts in polar aprotic solvents, which promotes the intramolecular attack of the carboxylate anion on the terminal carbon bearing the bromine atom. This SN2-type ring closure is kinetically favored by the specific chain length of the C15 backbone, which possesses the ideal flexibility to adopt the conformation required for ring formation without excessive strain. The reaction conditions are carefully tuned to prevent intermolecular esterification, which would lead to unwanted polyesters, ensuring that the reaction trajectory remains focused on the formation of the monomeric lactone. This mechanistic precision allows for a robust process that is highly reproducible and suitable for the commercial scale-up of complex fragrance ingredients.
How to Synthesize Thibetolide Efficiently
The synthesis of Thibetolide via this patented route involves a logical progression of eight distinct operational units, starting from the fermentation broth processing to the final vacuum distillation. The protocol emphasizes the importance of strict temperature control during the reduction and bromination phases to ensure safety and maximize conversion rates. Operators must pay close attention to the stoichiometry of the base used in the cyclization step, as an excess can lead to hydrolysis of the newly formed lactone, while insufficient base may result in incomplete conversion. The detailed standardized operating procedures for each unit operation, including specific solvent ratios and agitation speeds, are critical for maintaining batch-to-batch consistency. For technical teams looking to implement this chemistry, the following guide outlines the critical process parameters derived from the patent examples.
- Esterify 1,15-pentadecanedioic acid with organic alcohol using a solid acid catalyst to form the diester.
- Perform partial hydrolysis using alkali metal hydroxides to obtain the monoester salt, followed by acidification.
- Reduce the monoester to a hydroxy ester using borohydride reagents, then hydrolyze to 15-hydroxypentadecanoic acid.
- Brominate the hydroxy acid and perform intramolecular cyclization in the presence of carbonate bases to yield crude Thibetolide.
- Purify the crude product via vacuum distillation to achieve final purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this bio-based synthetic route offers compelling strategic advantages that extend beyond simple unit cost metrics. By shifting the raw material base from petrochemical derivatives to fermentation products, manufacturers can decouple their production costs from the volatility of the oil market, leading to more predictable pricing structures over long-term contracts. The elimination of expensive transition metal catalysts not only reduces the direct material cost but also simplifies the purification train, as there is no need for specialized resin columns or complex extraction protocols to remove trace metals to ppm levels. This simplification translates directly into reduced processing time and lower utility consumption, contributing to a leaner and more agile manufacturing operation. Furthermore, the use of greener chemistry aligns with the increasing corporate sustainability mandates of major global fragrance houses, enhancing the marketability of the final product.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing readily available fermentation acids and avoiding the use of noble metals like palladium or platinum which are common in hydrogenation steps of alternative routes. The simplified workup procedures, which rely on standard filtration and distillation rather than chromatography, drastically reduce the consumption of silica gel and specialized solvents. Additionally, the high selectivity of the reaction minimizes the loss of valuable intermediates to byproduct streams, thereby improving the overall mass balance and yield efficiency. These factors combined result in a substantially lower cost of goods sold (COGS) without compromising the quality of the final musk.
- Enhanced Supply Chain Reliability: Sourcing 1,15-pentadecanedioic acid from biological fermentation provides a stable and renewable supply chain that is less prone to the geopolitical disruptions often affecting petrochemical feedstocks. The synthetic route utilizes commodity chemicals such as sodium borohydride, potassium carbonate, and common organic solvents, all of which have robust global supply networks with multiple qualified vendors. This diversification of raw material sources mitigates the risk of single-supplier dependency and ensures continuous production capability even during market shortages. Consequently, this reliability aids in reducing lead time for high-purity musks and ensures timely delivery to downstream formulation customers.
- Scalability and Environmental Compliance: The technology is inherently designed for industrial scalability, utilizing reaction conditions that are easily managed in standard stainless steel reactors without the need for exotic high-pressure vessels. The waste profile of this process is significantly cleaner than traditional methods, as it avoids the generation of heavy metal sludge and reduces the volume of organic waste requiring incineration. This environmental compatibility simplifies the permitting process for new manufacturing facilities and reduces the ongoing costs associated with wastewater treatment and regulatory compliance. The ability to scale from pilot batches to multi-ton production with minimal process re-engineering makes this an ideal candidate for rapid capacity expansion.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Thibetolide synthesized via this advanced fermentation-based route. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this material into existing fragrance portfolios or new product developments.
Q: What are the primary advantages of the bio-fermentation route for Thibetolide compared to petrochemical methods?
A: The bio-fermentation route utilizes 1,15-pentadecanedioic acid derived from renewable resources, significantly reducing reliance on volatile petrochemical feedstocks. This method shortens the synthetic pathway, lowers energy consumption during reaction phases, and minimizes environmental pollution associated with heavy metal catalysts often found in traditional macrolactone synthesis.
Q: How does this synthesis method ensure high purity for fragrance applications?
A: The process incorporates a rigorous purification stage involving vacuum distillation at controlled pressures (130-140°C/2mmHg), which effectively separates the target macrocyclic lactone from oligomeric byproducts. The use of selective reduction and controlled cyclization conditions minimizes the formation of impurities, resulting in a product with consistent organoleptic properties suitable for fine fragrance formulations.
Q: Is this manufacturing process scalable for industrial production volumes?
A: Yes, the technology is designed with strong industrial operability, utilizing common solvents and reagents like sodium borohydride and potassium carbonate that are readily available at scale. The reaction conditions operate within moderate temperature ranges and do not require exotic high-pressure equipment, facilitating easy transfer from laboratory pilot runs to multi-ton commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thibetolide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of delivering high-performance fragrance ingredients that meet the rigorous standards of the global luxury market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistent quality. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of Thibetolide delivers the characteristic warm, musky note expected by perfumers. Our commitment to process excellence means we can offer a supply solution that is both economically competitive and environmentally responsible.
We invite you to collaborate with us to explore how this optimized synthetic route can enhance your product lineup and improve your margin structure. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals in the fine fragrance sector.
