Advanced Manufacturing of Brivudine Intermediates: A Technical Breakthrough for Scalable Pharmaceutical Production
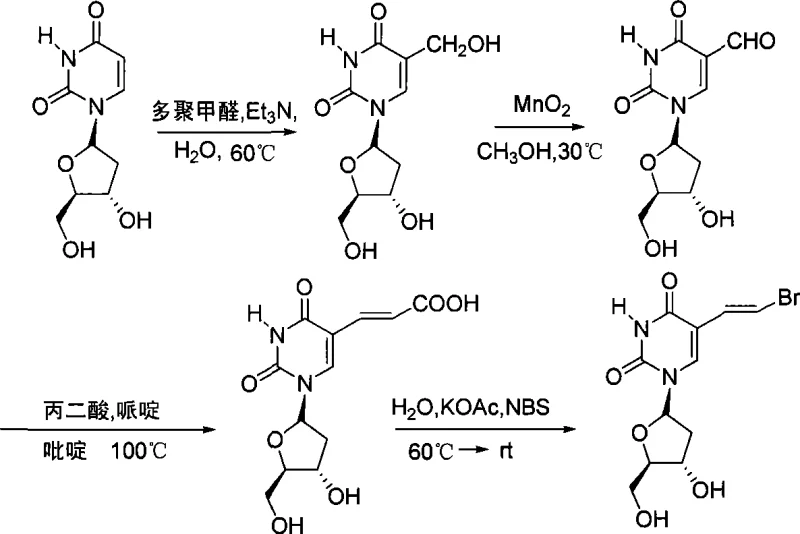
The pharmaceutical industry continuously seeks robust synthetic pathways for high-potency antiviral agents, and Brivudine ((E)-5-(2-bromovinyl)-2'-deoxyuridine) stands out as a critical therapeutic for treating Varicella-Zoster Virus infections. The technical landscape for manufacturing this compound has evolved significantly, moving away from hazardous and costly legacy methods toward more sustainable chemical engineering solutions. Patent CN101066987A introduces a transformative four-step synthesis strategy that begins with the readily available industrial feedstock 2'-deoxyuridine. This methodology addresses long-standing challenges in nucleoside chemistry, specifically the control of stereochemistry during vinyl group installation and the elimination of toxic heavy metal residues. By leveraging a sequence involving hydroxymethylation, selective oxidation, Knoevenagel condensation, and decarboxylative bromination, this process establishes a new benchmark for efficiency. For global procurement teams and R&D directors, understanding this specific intellectual property is vital, as it represents a viable pathway for cost reduction in pharmaceutical intermediates manufacturing while ensuring the stringent purity profiles required for clinical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Brivudine and related vinyl-nucleosides relied heavily on organometallic chemistry that posed significant barriers to industrial scale-up. Prior art, such as the methods described by Jones and Walker, utilized organomercury intermediates followed by catalysis with lithium tetrachloropalladate (Li2PdCl4). These conventional approaches suffered from multiple critical deficiencies that hindered commercial viability. Firstly, the use of mercury and palladium introduced severe environmental and safety liabilities, requiring complex waste treatment protocols that inflated operational expenditures. Secondly, the reliance on precious metal catalysts like palladium created a volatile cost structure dependent on fluctuating commodity markets. Furthermore, the purification of intermediates in these legacy routes frequently necessitated column chromatography, a technique that is notoriously difficult to translate from laboratory benchtop to multi-ton reactor scales. The combination of toxic reagents, expensive catalysts, and labor-intensive purification resulted in low overall yields and inconsistent product quality, making the commercial scale-up of complex antiviral intermediates economically unfeasible for many manufacturers.
The Novel Approach
In stark contrast, the methodology disclosed in CN101066987A presents a streamlined, metal-free alternative that fundamentally restructures the synthetic logic. This novel approach bypasses the need for organomercury species and precious metal catalysts entirely, opting instead for a sequence driven by common organic reagents such as paraformaldehyde, manganese dioxide, malonic acid, and N-bromosuccinimide (NBS). The strategic decision to start with 2'-deoxyuridine, a commodity chemical already produced at industrial scales globally, immediately stabilizes the supply chain and reduces raw material lead times. The process achieves the critical carbon-carbon bond formation through a Knoevenagel condensation followed by a decarboxylative bromination, which inherently favors the formation of the biologically active (E)-isomer without requiring difficult separation of cis-trans mixtures. By replacing column chromatography with simple recrystallization steps using solvents like methanol, this route dramatically simplifies downstream processing. This shift not only enhances the throughput capacity but also ensures that the final high-purity pharmaceutical intermediate meets regulatory standards without the risk of heavy metal contamination.
Mechanistic Insights into Manganese Dioxide Oxidation and Stereoselective Condensation
The core chemical innovation in this pathway lies in the selective oxidation step utilizing activated manganese dioxide (MnO2). In nucleoside chemistry, differentiating between primary and secondary hydroxyl groups on the sugar ring is often challenging and typically requires protecting group strategies that add steps and reduce yield. However, manganese dioxide exhibits a unique chemoselectivity for oxidizing allylic or benzylic alcohols, and in this specific substrate context, it effectively converts the 5-hydroxymethyl group to the corresponding aldehyde without disturbing the 3'-hydroxyl or the glycosidic bond. This selectivity is paramount because it allows the synthesis to proceed without protective groups on the sugar moiety, thereby shortening the synthetic timeline and reducing solvent consumption. The reaction conditions, typically conducted in methanol at moderate temperatures around 30°C, ensure that the sensitive uracil base remains intact while generating the reactive aldehyde intermediate necessary for the subsequent chain extension.
Following oxidation, the stereochemical integrity of the final vinyl bromide is secured through the Knoevenagel condensation with malonic acid. This reaction, catalyzed by piperidine in pyridine at elevated temperatures (100°C), proceeds through a mechanism that thermodynamically favors the formation of the (E)-isomer of the acrylic acid derivative. The subsequent decarboxylative bromination using NBS and potassium acetate is a concerted process where the carboxylic acid group serves as a leaving group, replaced by bromine with retention of the double bond geometry. This mechanistic pathway effectively solves the cis-trans isomerization problem that plagued earlier synthetic attempts. By controlling the reaction environment and utilizing specific reagents like potassium acetate to buffer the acidity, the process minimizes side reactions such as hydration of the double bond or degradation of the glycosidic linkage. This level of mechanistic control is essential for R&D directors aiming to validate the impurity profile and ensure batch-to-batch consistency in a GMP environment.
How to Synthesize Brivudine Efficiently
Implementing this four-step synthesis requires precise control over reaction parameters to maximize yield and minimize impurity formation. The process begins with the hydroxymethylation of 2'-deoxyuridine using paraformaldehyde in an aqueous triethylamine solution, followed by the critical manganese dioxide oxidation which must be monitored to prevent over-oxidation. The subsequent condensation and bromination steps demand strict temperature management to maintain stereoselectivity. While the general chemical transformations are well-defined, the specific operational details regarding stoichiometry, addition rates, and workup procedures are critical for successful technology transfer. The detailed standardized synthesis steps, including specific molar ratios and isolation techniques, are outlined in the guide below to assist technical teams in replicating this efficient route.
- React 2'-deoxyuridine with paraformaldehyde in aqueous triethylamine at 60°C to obtain 5-hydroxymethyl-2'-deoxyuridine.
- Selectively oxidize the hydroxymethyl intermediate using manganese dioxide in methanol at 30°C to yield 5-formyl-2'-deoxyuridine.
- Perform Knoevenagel condensation with malonic acid in pyridine/piperidine at 100°C to form the (E)-5-(2-carboxyvinyl) derivative.
- Execute decarboxylative bromination using N-bromosuccinimide (NBS) and potassium acetate in water to finalize Brivudine production.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers substantial strategic advantages beyond mere technical feasibility. The primary value driver is the drastic simplification of the bill of materials. By eliminating the need for lithium tetrachloropalladate and organomercury precursors, manufacturers can insulate themselves from the volatility of precious metal markets and the regulatory burdens associated with toxic heavy metals. This substitution translates directly into a more predictable cost structure and reduced expenditure on specialized waste disposal services. Furthermore, the reliance on 2'-deoxyuridine as a starting material leverages an existing, mature global supply chain, ensuring that raw material availability is not a bottleneck for production scaling. The ability to source key inputs from multiple established vendors enhances supply security and mitigates the risk of single-source dependency.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from the removal of expensive catalytic systems and the simplification of purification workflows. Traditional methods requiring column chromatography are labor-intensive and solvent-heavy, driving up both direct material costs and utility expenses. In contrast, this novel route utilizes recrystallization as the primary purification method, which is significantly cheaper and faster to execute on an industrial scale. Additionally, the avoidance of precious metal catalysts eliminates the need for costly metal scavenging resins and the associated analytical testing for residual metals, further streamlining the cost base. These factors combine to create a manufacturing process with a substantially lower cost of goods sold (COGS), allowing for more competitive pricing in the generic antiviral market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commoditized reagents such as paraformaldehyde, malonic acid, and NBS, which are produced in high volumes by numerous chemical suppliers worldwide. Unlike specialized organometallic reagents that may have long lead times or limited suppliers, these basic chemicals are readily accessible, reducing the risk of production delays due to raw material shortages. The robustness of the synthetic route also means that minor fluctuations in reagent quality can be accommodated without compromising the final product specification. This flexibility allows supply chain planners to optimize inventory levels and negotiate better terms with vendors, knowing that the production process is not fragile or overly sensitive to specific reagent grades.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process represents a significant improvement over legacy methods. The absence of mercury and palladium reduces the toxicity profile of the waste stream, simplifying compliance with increasingly stringent environmental regulations. The shift from chromatography to crystallization reduces the volume of organic solvent waste generated per kilogram of product, aligning with green chemistry principles. This environmental efficiency not only lowers disposal costs but also enhances the corporate sustainability profile of the manufacturer. Moreover, the simplicity of the unit operations—primarily stirring, heating, filtering, and crystallizing—makes the process highly scalable from pilot plant to full commercial production without the need for specialized equipment, facilitating rapid capacity expansion to meet market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Brivudine synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on yield expectations, purification strategies, and regulatory considerations. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their own manufacturing portfolios.
Q: Why is the manganese dioxide oxidation step critical in this Brivudine synthesis?
A: Manganese dioxide offers high selectivity for oxidizing allylic alcohols to aldehydes without affecting other sensitive functional groups on the nucleoside sugar moiety, eliminating the need for protection-deprotection sequences.
Q: How does this patent method improve upon traditional palladium-catalyzed routes?
A: Traditional methods often rely on expensive lithium tetrachloropalladate and organomercury intermediates. This novel route replaces them with inexpensive reagents like NBS and MnO2, significantly lowering raw material costs and simplifying purification.
Q: What purification methods are suitable for scaling up this Brivudine process?
A: The process is designed to avoid column chromatography. Instead, it utilizes recrystallization from solvents like methanol and simple filtration techniques, which are far more amenable to large-scale industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Brivudine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires a partner with deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics of patent CN101066987A can be realized in a practical manufacturing setting. We maintain stringent purity specifications and operate rigorous QC labs equipped to detect trace impurities and residual solvents, guaranteeing that every batch of Brivudine intermediate meets the highest international pharmacopeial standards. Our commitment to quality assurance extends throughout the entire production lifecycle, from raw material qualification to final product release.
We invite pharmaceutical companies and contract research organizations to collaborate with us to leverage this advanced synthesis technology for their antiviral drug pipelines. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage potential partners to contact us directly to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete performance metrics rather than theoretical projections. Let us help you secure a stable, cost-effective supply of high-quality Brivudine intermediates for your global operations.
