Optimizing Ranitidine Intermediate Production: A Technical Breakthrough in Catalytic Condensation and Commercial Scalability
The pharmaceutical industry is constantly seeking more efficient pathways for synthesizing critical gastric drug intermediates, and recent advancements detailed in patent CN114605360B offer a compelling solution for the production of 2-[[[5-(dimethylamino)methyl-2-furyl]methyl]thio]ethylamine. This specific compound serves as a vital precursor in the manufacture of Ranitidine hydrochloride, a widely utilized H2 receptor antagonist. The disclosed technology represents a significant departure from conventional synthetic routes by introducing a transition metal-catalyzed condensation process that fundamentally alters the impurity profile and downstream processing requirements. By leveraging nanoscale copper catalysts and optimizing reaction atmospheres with dimethylamine and formaldehyde, this method achieves a total yield of more than 92.1% while maintaining a purity level exceeding 99.1%. For a reliable pharmaceutical intermediate supplier, understanding these mechanistic shifts is crucial for evaluating long-term supply chain stability and cost structures in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of this aminoethyl thioether has relied heavily on processes that utilize cysteamine hydrochloride as the primary sulfur-containing nucleophile. While effective in forming the carbon-sulfur bond, this traditional approach introduces a significant stoichiometric burden in the form of inorganic salt byproducts. Specifically, the neutralization steps required to liberate the free amine from its hydrochloride salt result in the generation of massive quantities of sodium chloride; data suggests that producing 1000kg of the target intermediate can yield approximately 700kg of sodium chloride waste. This not only complicates the waste treatment protocols but also necessitates energy-intensive extraction procedures using solvents like dichloromethane to separate the organic product from the aqueous salt phase. Furthermore, the multi-step workup involving alkalization and extraction increases the risk of product loss and environmental contamination, making cost reduction in API manufacturing increasingly difficult under stricter regulatory frameworks.
The Novel Approach
In stark contrast, the innovative methodology described in the patent data eliminates the root cause of salt generation by substituting cysteamine hydrochloride with free cysteamine in the presence of a transition metal catalyst. This strategic modification allows the condensation reaction to proceed without the need for subsequent alkalization, thereby bypassing the formation of sodium chloride entirely. The process utilizes an alkane solvent, such as cyclohexane, which facilitates a homogeneous reaction environment conducive to high conversion rates. By removing the extraction and alkalization steps, the workflow is drastically simplified to a direct distillation purification sequence. This streamlining not only reduces the consumption of organic solvents but also minimizes the operational time and equipment footprint required for production. For supply chain heads, this translates to a more robust and scalable process that reduces lead time for high-purity pharmaceutical intermediates while aligning with green chemistry principles.
Mechanistic Insights into Nano-Copper Catalyzed Condensation
The core of this technological advancement lies in the precise application of nanoscale copper as a transition metal catalyst, which operates effectively within a particle diameter range of 5 to 20nm. The reaction mechanism involves a sequential condensation where furfuryl alcohol first reacts with dimethylamine and formaldehyde to form an intermediate species, which subsequently undergoes thioetherification with cysteamine. The presence of the nanoscale copper significantly lowers the activation energy for these transformations, allowing the reaction to proceed efficiently at moderate temperatures between 75°C and 95°C. Crucially, the system is designed to continuously remove water generated during the condensation, maintaining a water content of less than 0.2wt% to drive the equilibrium towards product formation. This rigorous control over reaction conditions ensures that side reactions are minimized, directly contributing to the observed high purity specifications and consistent batch-to-batch reproducibility required for commercial scale-up of complex pharmaceutical intermediates.
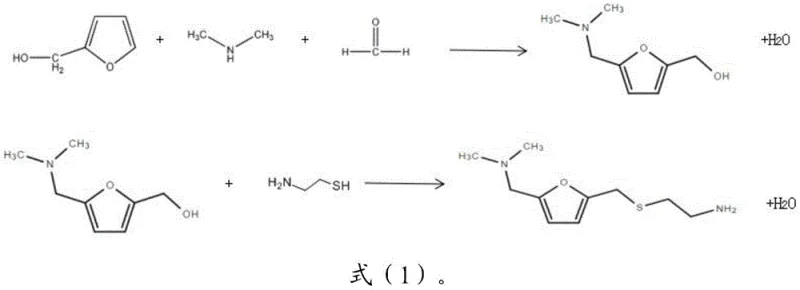
Furthermore, the impurity control mechanism is inherently built into the choice of reagents and the purification strategy. By avoiding the use of hydrochloride salts, the process prevents the introduction of chloride ions that could potentially corrode equipment or contaminate the final active pharmaceutical ingredient. The purification is achieved through a two-stage distillation process: a first distillation at reduced pressure (-0.09 MPa) removes the alkane solvent and unreacted starting materials, followed by a second high-vacuum distillation (50-150 Pa) to isolate the target amine. This physical separation method is far superior to chemical extraction in terms of selectivity, ensuring that trace organic impurities are effectively separated from the main product fraction. For R&D directors focused on purity and impurity profiles, this distillation-only approach offers a cleaner spectrum and simplifies the validation process for regulatory filings.
How to Synthesize 2-[[[5-(Dimethylamino)methyl-2-furyl]methyl]thio]ethylamine Efficiently
Implementing this synthesis route requires careful attention to the order of addition and the maintenance of anhydrous conditions throughout the reaction cycle. The process begins with the preparation of a furfuryl alcohol solution in cyclohexane, into which the nanoscale copper catalyst is dispersed. The reaction atmosphere is carefully controlled by introducing dried dimethylamine and formaldehyde gases, initiating the first condensation stage at temperatures around 78-84°C. Once the intermediate is formed, the temperature is raised to 90-95°C, and cysteamine is added dropwise over a period of several hours to manage the exotherm and ensure complete conversion. The detailed standardized synthesis steps, including specific stirring rates and vacuum parameters for the distillation phases, are outlined in the technical guide below for process engineers to review.
- Mix furfuryl alcohol, nanoscale copper catalyst, and cysteamine in an alkane solvent under a dimethylamine and formaldehyde atmosphere.
- Conduct a two-stage condensation reaction, initially at 75-85°C followed by 90-95°C, while continuously removing water to maintain system dryness below 0.2wt%.
- Filter to recover the catalyst, then purify the crude liquid via sequential vacuum distillation to isolate the high-purity amine product without alkalization.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this catalytic condensation method offers profound economic and logistical benefits that extend beyond simple yield improvements. The elimination of sodium chloride byproducts removes a major waste disposal cost center, significantly reducing the environmental compliance burden associated with salt-laden wastewater treatment. Additionally, the ability to recover and reuse the transition metal catalyst through simple filtration at elevated temperatures enhances the overall atom economy of the process. These factors combine to create a manufacturing protocol that is not only chemically superior but also economically resilient against fluctuating raw material costs and tightening environmental regulations.
- Cost Reduction in Manufacturing: The removal of the alkalization and organic solvent extraction steps results in substantial cost savings by reducing both labor hours and solvent consumption. Without the need to purchase and dispose of large quantities of inorganic bases and extractants, the operational expenditure per kilogram of product is drastically lowered. Furthermore, the high yield exceeding 92% ensures that raw material utilization is maximized, minimizing the cost of goods sold. This efficiency allows for a more competitive pricing structure without compromising on the quality standards required for pharmaceutical applications.
- Enhanced Supply Chain Reliability: Simplifying the post-reaction workup to a direct distillation process reduces the number of unit operations, thereby decreasing the potential points of failure in the production line. The use of commercially available raw materials like furfuryl alcohol and cyclohexane ensures that supply continuity is maintained even during market fluctuations. By shortening the overall production cycle time through the elimination of lengthy extraction and washing phases, manufacturers can respond more agilely to demand spikes, ensuring a steady flow of high-purity intermediates to downstream API producers.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on standard distillation and filtration equipment rather than complex multi-phase extraction setups. The significant reduction in organic solvent usage and the complete avoidance of inorganic salt waste align perfectly with modern green chemistry initiatives. This makes the technology highly attractive for facilities aiming to reduce their carbon footprint and meet stringent environmental discharge limits, facilitating smoother regulatory approvals for capacity expansion projects.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details is essential for technical teams evaluating the feasibility of adopting this method for large-scale production.
Q: How does this new method improve upon traditional Ranitidine intermediate synthesis?
A: Traditional methods utilize cysteamine hydrochloride, which generates substantial sodium chloride byproducts requiring complex extraction. This novel approach uses free cysteamine with a transition metal catalyst, completely avoiding salt formation and enabling direct distillation purification.
Q: What are the purity and yield specifications achievable with this catalytic route?
A: Experimental data indicates a total yield exceeding 92.1% and a final product purity greater than 99.1%, achieved through precise temperature control and nanoscale copper catalysis.
Q: Is the nanoscale copper catalyst recoverable for industrial scale-up?
A: Yes, the process includes a solid-liquid separation step at elevated temperatures (85-95°C) specifically designed to filter and recover the transition metal catalyst for reuse, enhancing process economics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-[[[5-(Dimethylamino)methyl-2-furyl]methyl]thio]ethylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the nano-copper catalyzed condensation are translated into reliable supply solutions. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of intermediate meets the exacting standards required for final drug substance manufacturing. We are committed to bridging the gap between patent innovation and industrial reality.
We invite you to engage with our technical procurement team to discuss how this optimized route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic impact of switching to this salt-free process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring a partnership built on transparency, technical excellence, and mutual growth.
