Revolutionizing Isoeugenol Methyl Ether Production: A Green One-Step Catalytic Strategy
The global demand for high-purity Isoeugenol Methyl Ether, a critical monomeric spice known for its fresh, spicy, and long-lasting floral fragrance, has driven significant innovation in synthetic methodologies. As detailed in the pioneering patent CN109836312B, a transformative approach has been established that fundamentally alters the production landscape for this valuable fine chemical intermediate. Historically, the synthesis of this compound relied on hazardous reagents and multi-step processes that posed severe safety and environmental challenges. However, this new technology introduces a robust, one-step liquid-solid phase reaction conducted under pressure, utilizing Dimethyl Carbonate (DMC) as a green methylating agent. This breakthrough not only eliminates the use of toxic dimethyl sulfate but also integrates the methylation and isomerization steps into a single, efficient operation. For R&D directors and procurement specialists seeking a reliable synthetic flavors & fragrances supplier, this patent represents a paradigm shift towards sustainable manufacturing, offering a pathway to produce high-purity products with drastically simplified downstream processing and enhanced operational safety profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the industrial preparation of Isoeugenol Methyl Ether from eugenol has been plagued by inherent inefficiencies and significant safety risks associated with legacy chemical practices. The conventional route typically necessitates a two-step sequence: first, the methylation of the phenolic hydroxyl group, followed by a separate isomerization of the allyl double bond to the propenyl configuration. The methylation step historically employs dimethyl sulfate or methyl bromide, reagents that are notoriously toxic, carcinogenic, and highly corrosive, generating substantial amounts of acidic waste that require complex and costly neutralization procedures. Furthermore, the subsequent isomerization often demands harsh conditions, such as high-temperature treatment with inorganic strong alkalis under reduced pressure, which frequently leads to unwanted side reactions, polymerization, and darkening of the product. These disjointed processes result in high material consumption, excessive energy usage due to multiple heating and cooling cycles, and difficult post-treatment protocols that hinder the ability to achieve consistent, high-purity specifications required by top-tier flavor and fragrance houses.
The Novel Approach
In stark contrast to these archaic methods, the novel methodology disclosed in patent CN109836312B streamlines the entire synthesis into a singular, pressurized liquid-solid phase reaction. By substituting the hazardous dimethyl sulfate with the environmentally benign Dimethyl Carbonate (DMC), the process immediately mitigates the most severe safety liabilities associated with production. The core innovation lies in the simultaneous execution of phenolic hydroxyl methylation and double bond isomerization within the same reactor, catalyzed by a synergistic system of solid alkali and a phase transfer catalyst. This one-pot strategy effectively collapses the production timeline, eliminating the need for intermediate isolation and the associated yield losses. The reaction is conducted under controlled pressure (0.2-0.3 MPa) and elevated temperatures (160-220°C), which compensates for the lower reactivity of DMC compared to traditional methylating agents, ensuring high conversion rates. This integrated approach not only simplifies the technological process but also aligns perfectly with modern green chemistry principles, offering a clean, energy-saving route that significantly reduces the environmental footprint of flavor & fragrance intermediates manufacturing.
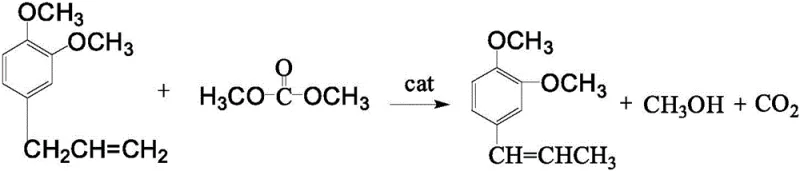
Mechanistic Insights into Solid Base and Phase Transfer Catalysis
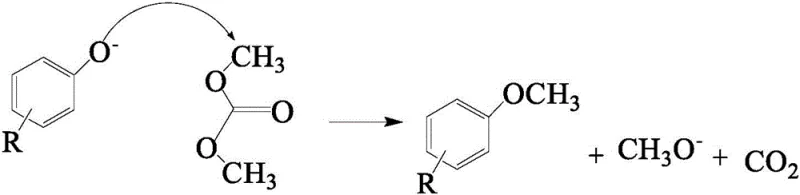
The success of this one-step synthesis relies heavily on a sophisticated understanding of reaction kinetics and the specific roles played by the catalytic system. The mechanism initiates with the deprotonation of the phenolic hydroxyl group on the eugenol molecule by the solid base catalyst, generating a highly reactive phenoxide anion. This nucleophile then attacks the methyl carbon of the Dimethyl Carbonate (DMC) molecule in a classic nucleophilic substitution reaction. Unlike traditional methylating agents, DMC decomposes into methanol and carbon dioxide upon reaction, which are gaseous or volatile byproducts that can be easily vented from the system, driving the equilibrium forward according to Le Chatelier's principle. The continuous removal of these byproducts through periodic venting of the reactor ensures that the reaction proceeds irreversibly towards the formation of the methyl ether intermediate, preventing the reverse reaction and maximizing the utilization of the raw materials.
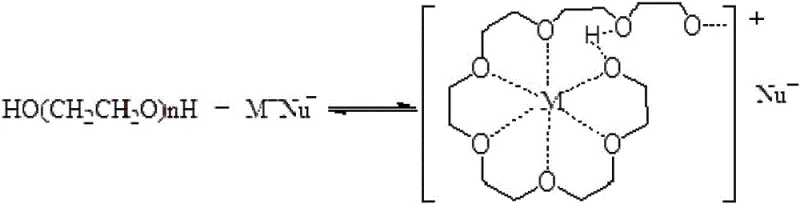
Crucially, the transformation does not stop at methylation; the concurrent isomerization of the double bond is facilitated by the unique properties of the Phase Transfer Catalyst (PTC), specifically polyethylene glycol (PEG). The PEG molecules possess a helical chain structure capable of folding into cavities that complex with metal cations (such as potassium ions from KOH or K2CO3). This complexation solubilizes the basic anions in the organic phase, bringing them into close proximity with the substrate. Under these conditions, the basic environment promotes the formation of a carbanion adjacent to the benzene ring. The subsequent abstraction of a proton and the rearrangement of the electron density lead to the migration of the double bond from the terminal allyl position to the internal propenyl position. This isomerization yields the thermodynamically more stable conjugated system found in Isoeugenol Methyl Ether. The synergy between the solid base, which drives the methylation, and the PTC, which lowers the activation energy for isomerization, allows the entire transformation to occur efficiently at temperatures lower than those required for traditional thermal isomerization, thereby preserving product quality and minimizing degradation.
How to Synthesize Isoeugenol Methyl Ether Efficiently
Implementing this advanced synthetic route requires precise control over reaction parameters to ensure optimal yield and purity. The process begins by charging eugenol and a calculated portion of DMC into a pressure-resistant reactor, followed by the introduction of the solid alkali catalyst and the PEG-400 phase transfer catalyst. The system is then pressurized with nitrogen and heated to the target range of 160-220°C. A critical operational detail is the dropwise addition of the remaining DMC over a period of approximately 2 hours; this controlled feeding prevents excessive pressure spikes caused by the boiling point of DMC and minimizes side reactions. Throughout the reaction, periodic venting is essential to remove the generated methanol and CO2, pushing the equilibrium towards product formation. Following the reaction period, the mixture is cooled, neutralized, and subjected to a standard workup involving extraction and vacuum rectification.
- Charge eugenol and a portion of dimethyl carbonate (DMC) into a pressure reactor, followed by the addition of solid alkali catalyst and PEG-400 phase transfer catalyst.
- Pressurize with nitrogen, heat to 160-220°C, and dropwise add the remaining DMC over 2 hours while maintaining pressure between 0.2-0.3 MPa.
- After reaction completion, cool, neutralize with dilute acid, extract with toluene, and purify via vacuum rectification to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented technology offers profound strategic advantages that extend far beyond simple chemical conversion. The primary value proposition lies in the drastic simplification of the supply chain risk profile by eliminating the dependency on highly regulated and dangerous raw materials like dimethyl sulfate. By switching to Dimethyl Carbonate, a green reagent with a much safer handling profile, facilities can reduce insurance premiums, lower the costs associated with hazardous waste disposal, and minimize the regulatory burden of storing toxic substances. Furthermore, the consolidation of two distinct chemical steps into a single unit operation significantly reduces the capital expenditure required for equipment, as fewer reactors and separation units are needed. This reduction in process complexity translates directly into lower operational expenditures (OPEX) through decreased energy consumption and reduced labor hours per batch, providing a clear path for cost reduction in synthetic flavors & fragrances manufacturing without compromising on the quality of the final output.
- Cost Reduction in Manufacturing: The elimination of the separate isomerization step and the replacement of expensive, hazardous reagents with cost-effective DMC creates a leaner production model. The ability to recover and recycle excess DMC and the extraction solvent (toluene) further enhances the economic viability of the process, ensuring that raw material costs are kept to a minimum while maintaining high throughput efficiency.
- Enhanced Supply Chain Reliability: By utilizing a one-pot synthesis, the overall production cycle time is significantly shortened, allowing for faster turnaround times on customer orders. The robustness of the solid base catalyst system, which tolerates a wider range of operating conditions compared to sensitive liquid acids or bases, ensures consistent batch-to-bquality, reducing the risk of production delays caused by off-spec material. This reliability is crucial for maintaining the continuity of supply for downstream applications in daily chemical essences and edible flavors.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory validation to commercial production scales of 100 kgs to 100 MT annually. The generation of benign byproducts (methanol and CO2) rather than sulfur-containing waste streams simplifies wastewater treatment and ensures compliance with increasingly stringent environmental regulations. This "green" credential not only future-proofs the supply chain against tightening legislation but also appeals to end-consumer brands seeking sustainably sourced ingredients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology. These insights are derived directly from the experimental data and technical disclosures within the patent literature, providing a transparent view of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing infrastructure.
Q: Why is Dimethyl Carbonate (DMC) preferred over Dimethyl Sulfate for this synthesis?
A: DMC is a green, non-toxic reagent that replaces the highly toxic and corrosive dimethyl sulfate, significantly reducing environmental hazards and post-treatment complexity associated with sulfuric acid byproducts.
Q: How does the one-step process improve efficiency compared to traditional methods?
A: Traditional methods require separate methylation and isomerization steps. This patented method combines both reactions in a single pressurized vessel, reducing energy consumption, processing time, and material loss.
Q: What role does the Phase Transfer Catalyst play in this reaction?
A: The PEG-400 catalyst complexes with metal ions from the solid base, facilitating their transfer into the organic phase to enhance the nucleophilic attack on DMC and promote double bond isomerization at lower temperatures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoeugenol Methyl Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is not just a regulatory necessity but a competitive advantage. Our technical team has extensively analyzed the methodology described in patent CN109836312B and possesses the expertise to adapt this pressurized liquid-solid phase reaction for large-scale commercial production. We offer extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this one-step process are fully realized in practice. Our state-of-the-art facilities are equipped with high-pressure reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Isoeugenol Methyl Ether delivers the fresh, spicy aroma profile required by the world's leading fragrance houses.
We invite you to collaborate with us to optimize your supply chain for this critical intermediate. By leveraging our process engineering capabilities, we can help you achieve substantial cost savings and improved safety standards. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis. We are ready to provide specific COA data from our pilot runs and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your long-term growth in the global flavors and fragrances market.
