Advanced Catalytic Hydrogenation for High-Purity Tropinol Production and Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic routes for critical intermediates like Tropinol, a pivotal precursor in the manufacture of Trospium Chloride and other anticholinergic agents. Patent CN115466255B introduces a transformative methodology that replaces traditional, hazardous catalytic systems with a highly efficient heteropolyacid-based approach. This innovation addresses long-standing challenges in purity, safety, and environmental compliance, offering a compelling value proposition for global supply chains. By leveraging the unique properties of the K5PPdW11O39/C catalyst, manufacturers can achieve conversion rates exceeding 96% while operating under relatively mild conditions. This technical advancement not only streamlines the production workflow but also aligns with modern green chemistry principles, making it an attractive option for reliable pharmaceutical intermediates suppliers aiming to optimize their portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Tropinol has relied heavily on Raney Nickel as the primary hydrogenation catalyst. While effective in terms of basic reactivity, Raney Nickel presents severe operational and logistical drawbacks that complicate large-scale manufacturing. The catalyst is notoriously unstable and pyrophoric, meaning it can spontaneously ignite upon exposure to air, necessitating rigorous safety protocols and specialized handling equipment throughout the production facility. Furthermore, post-reaction processing is cumbersome; the spent catalyst must be chemically destroyed before filtration to prevent safety incidents, generating significant volumes of hazardous solid waste containing nickel, aluminum, and organic residues. This waste stream poses substantial environmental liabilities and increases disposal costs, creating a bottleneck for cost reduction in API manufacturing. Additionally, the variability in catalyst activity often leads to inconsistent yields and impurity profiles, requiring extensive downstream purification to meet stringent pharmacopeial standards.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN115466255B utilizes a sophisticated K5PPdW11O39/C heteropolyacid catalyst supported on activated carbon. This novel catalytic system exhibits exceptional stability and activity, allowing for a direct hydrogenation of Tropinone to Tropinol without the inherent dangers associated with pyrophoric materials. The process operates efficiently in methanol at temperatures between 55-60°C and hydrogen pressures of 1-5 MPa, achieving raw material conversion levels where residual content is less than 1.0%. The high selectivity of this catalyst minimizes the formation of side products, thereby simplifying the isolation process to a straightforward distillation step. This shift represents a paradigm change in process chemistry, moving away from brute-force catalysis towards precision engineering that enhances both safety and yield.
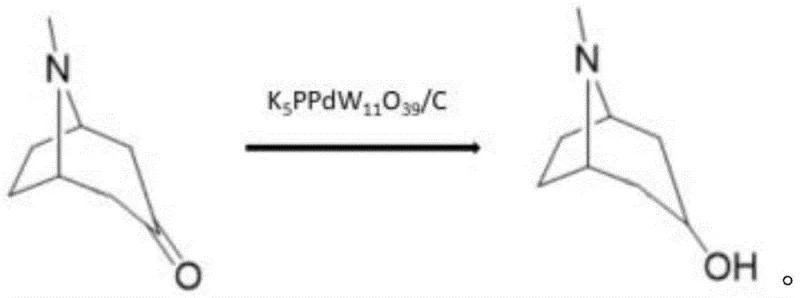
Mechanistic Insights into K5PPdW11O39/C Catalyzed Hydrogenation
The efficacy of the K5PPdW11O39/C catalyst lies in its unique structural composition as a palladium-substituted polyoxometalate. Unlike traditional heterogeneous catalysts where metal particles may leach or aggregate over time, the palladium atoms in this heteropolyacid framework are atomically dispersed and stabilized within the Keggin-type structure. This arrangement facilitates a highly efficient hydrogen activation mechanism, where molecular hydrogen is dissociated and transferred to the carbonyl group of the Tropinone substrate with minimal energy barriers. The support on activated carbon further enhances mass transfer properties, ensuring that the reactants have optimal access to the active sites. This mechanistic precision results in a reaction pathway that favors the formation of the desired alcohol functionality while suppressing over-reduction or ring-opening side reactions that typically plague less selective catalytic systems.
From an impurity control perspective, this catalytic system offers distinct advantages for R&D teams focused on quality by design. The high specificity of the hydrogenation step means that the crude reaction mixture contains significantly fewer by-products compared to Raney Nickel processes. This purity profile reduces the burden on downstream purification units, such as crystallization or chromatography, which are often the most expensive and time-consuming stages of API production. By minimizing the generation of closely related impurities, the process ensures a more consistent quality of the final Tropinol product. This consistency is critical for regulatory filings, as it demonstrates a robust understanding and control of the manufacturing process, thereby reducing the risk of batch failures and ensuring a steady supply of high-purity pharmaceutical intermediates for downstream drug synthesis.
How to Synthesize Tropinol Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this advanced technology in a commercial setting. The process begins with a decolorization step using activated carbon in methanol, which removes colored impurities from the starting Tropinone material before the reaction even commences. Following this pretreatment, the reaction is conducted in a high-pressure reactor under controlled nitrogen and hydrogen atmospheres to ensure safety and efficiency. The detailed operational parameters, including specific pressure cycles and temperature maintenance, are designed to maximize catalyst longevity and reaction completion. For a comprehensive understanding of the exact stoichiometry and workup procedures required to replicate these results, please refer to the standardized synthesis guide below.
- Prepare a decolorized tropinone solution by mixing tropinone and activated carbon in stirred methanol, followed by filtration to remove impurities.
- Charge the high-pressure reactor with the decolorized solution and the K5PPdW11O39/C heteropolyacid catalyst, then perform nitrogen and hydrogen置换 (replacement) cycles to ensure an inert atmosphere.
- Maintain hydrogen pressure at 1-5 MPa and temperature at 55-60°C for 40-50 hours until raw material consumption exceeds 99%, followed by distillation to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this heteropolyacid-catalyzed route offers tangible strategic benefits beyond mere chemical efficiency. The elimination of pyrophoric Raney Nickel fundamentally alters the risk profile of the manufacturing site, potentially lowering insurance premiums and reducing the need for specialized hazardous material storage infrastructure. Furthermore, the simplified waste stream—devoid of heavy metal sludge requiring complex destruction—translates directly into lower environmental compliance costs and faster turnaround times between batches. These operational efficiencies contribute to a more resilient supply chain capable of responding quickly to market demands without the bottlenecks associated with hazardous waste disposal. Consequently, this technology supports significant cost savings in pharmaceutical intermediates manufacturing by optimizing both material utilization and operational overhead.
- Cost Reduction in Manufacturing: The transition to the K5PPdW11O39/C catalyst eliminates the costly and labor-intensive steps associated with the destruction and disposal of spent Raney Nickel. By avoiding the generation of hazardous heavy metal waste, facilities can drastically reduce their expenditure on waste management services and regulatory compliance fees. Additionally, the high yield of over 96% ensures that raw material inputs are converted into valuable product with minimal loss, improving the overall material balance and reducing the cost per kilogram of the final API intermediate. This efficiency gain is compounded by the removal of complex purification steps, which saves on solvent usage and energy consumption during the isolation phase.
- Enhanced Supply Chain Reliability: The stability of the new catalyst allows for more predictable production scheduling, as there is no risk of spontaneous combustion or catalyst deactivation due to air exposure during loading. This reliability ensures consistent batch-to-batch quality and reduces the likelihood of unplanned shutdowns or safety incidents that could disrupt supply continuity. Moreover, the simplified process flow shortens the overall cycle time from raw material charging to final product isolation, enabling manufacturers to increase throughput and reduce lead time for high-purity pharmaceutical intermediates. This agility is crucial for maintaining inventory levels and meeting the just-in-time delivery requirements of major pharmaceutical clients.
- Scalability and Environmental Compliance: The process described is inherently scalable, having been demonstrated effectively from laboratory scales up to multi-hundred kilogram batches without loss of efficiency. The use of methanol as a solvent and the absence of toxic heavy metal residues make the process more environmentally benign, aligning with increasingly strict global regulations on industrial emissions and waste. This 'green' profile not only future-proofs the manufacturing asset against tightening environmental laws but also enhances the corporate sustainability credentials of the supplier. Such compliance is increasingly becoming a prerequisite for inclusion in the supply chains of top-tier multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in patent CN115466255B, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of adopting this technology for commercial production of Tropinol and its derivatives.
Q: What are the safety advantages of using K5PPdW11O39/C over Raney Nickel?
A: Unlike Raney Nickel, which is pyrophoric and requires complex destruction procedures after use, the K5PPdW11O39/C heteropolyacid catalyst is stable in air, significantly reducing fire hazards and simplifying waste disposal protocols.
Q: Does this new catalytic method require extensive purification steps?
A: No, the process described in patent CN115466255B achieves high selectivity and conversion rates (>96%), allowing the reduced product to be isolated directly via distillation without complex purification steps often needed with traditional catalysts.
Q: How does this synthesis route impact the cost of Trospium Chloride manufacturing?
A: By eliminating the need for hazardous catalyst destruction and reducing solid waste generation, this route lowers environmental compliance costs and improves overall material efficiency, leading to substantial cost reductions in API intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tropinol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has extensively evaluated the K5PPdW11O39/C catalytic system and confirmed its potential for delivering superior quality Tropinol. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of this vital intermediate. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch meets the highest international standards for pharmaceutical applications.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be integrated into your supply chain. By collaborating with us, you can leverage our expertise to achieve a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to request specific COA data and route feasibility assessments to verify the commercial viability of this process for your projects. Let us help you secure a sustainable and cost-effective source of high-quality Tropinol for your drug development and manufacturing needs.
