Advanced Manufacturing of Alogliptin Benzoate via Optimized Ester Solvent Systems
Introduction to Patent CN107540656B
The global pharmaceutical landscape for Type 2 diabetes treatments continues to evolve, with Dipeptidyl Peptidase-4 (DPP-4) inhibitors remaining a cornerstone of therapy. Patent CN107540656B introduces a transformative preparation method for Alogliptin Benzoate, addressing critical bottlenecks in existing manufacturing protocols. This intellectual property details a robust synthetic pathway that replaces hazardous reagents and toxic high-boiling solvents with safer, more efficient ester-based systems. For R&D directors and supply chain leaders, this patent represents a significant opportunity to enhance process safety while simultaneously driving down the cost of goods sold (COGS) through improved yield and simplified downstream processing. The technology specifically targets the elimination of carcinogenic methylating agents and the replacement of difficult-to-recycle solvents like NMP, aligning perfectly with modern green chemistry mandates.
Furthermore, the technical disclosure emphasizes the achievement of ultra-high purity standards, with the final active pharmaceutical ingredient (API) demonstrating HPLC purity exceeding 99.95%. This level of quality control is paramount for regulatory compliance in major markets such as the US and EU, where impurity profiles are scrutinized under ICH guidelines. By leveraging this optimized route, manufacturers can mitigate the risks associated with complex purification steps and reduce the likelihood of batch failures due to out-of-specification impurity levels. The following analysis dissects the mechanistic advantages and commercial implications of adopting this superior synthesis strategy for large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing Alogliptin Benzoate have historically been plagued by significant safety and environmental liabilities. As illustrated in the reaction scheme below, early routes (such as those disclosed in WO2005/095381) relied heavily on the use of sodium hydride (NaH) and methyl iodide (MeI). These reagents pose extreme operational hazards; NaH is pyrophoric and requires stringent handling protocols, while methyl iodide is a known carcinogen, necessitating expensive containment engineering and worker safety monitoring. Additionally, these processes frequently utilized polar aprotic solvents like DMF and DMSO, which possess high boiling points that make solvent recovery energy-intensive and economically inefficient.
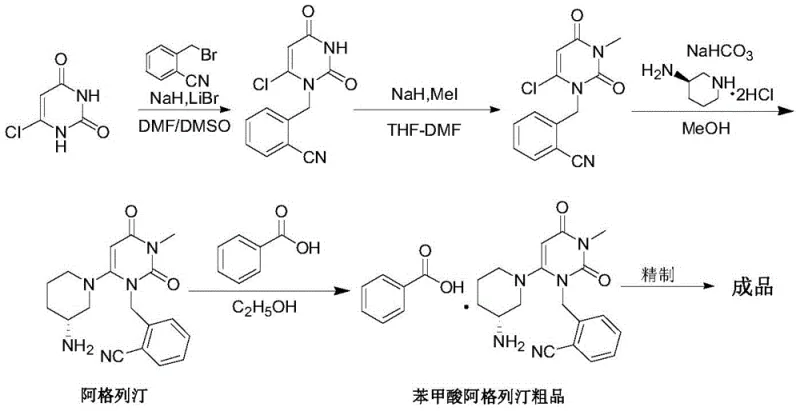
Subsequent improvements, such as those found in WO2007035629A2, attempted to mitigate some risks by switching to N-methyl-2-pyrrolidone (NMP) and toluene systems. However, these solvents introduce their own set of supply chain and regulatory challenges. NMP is subject to increasing regulatory restrictions under REACH due to reproductive toxicity concerns, limiting its long-term viability in sustainable manufacturing. Moreover, the post-treatment in these legacy processes often requires massive volumes of water to precipitate the product, generating substantial wastewater streams that increase environmental compliance costs. The cumulative effect of these factors is a process with low overall yields, typically hovering around 20-25%, which drastically inflates the raw material cost per kilogram of the final API.
The Novel Approach
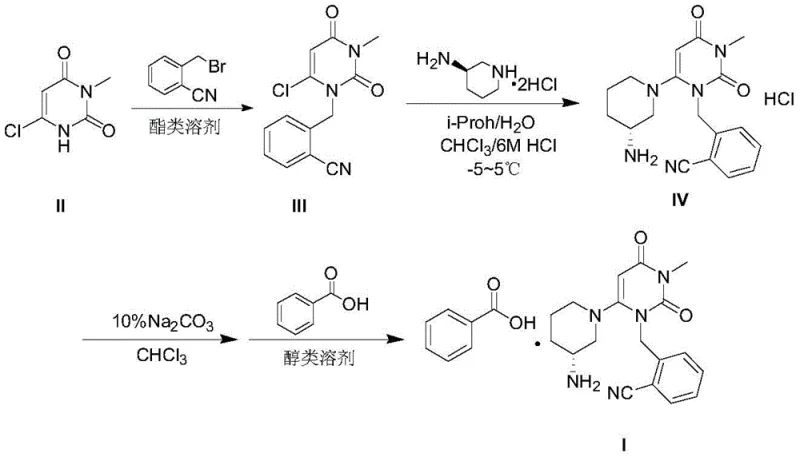
In stark contrast, the methodology disclosed in Patent CN107540656B utilizes a fundamentally safer and more efficient solvent system based on organic esters, such as ethyl acetate or isopropyl acetate. This novel approach replaces the hazardous methylation step entirely by starting with 6-chloro-3-methyluracil, thereby eliminating the need for methyl iodide and strong bases like sodium hydride. The reaction proceeds smoothly in the presence of an organic base, such as N,N-diisopropylethylamine, within the ester solvent matrix. A key innovation of this route is the solubility profile of the intermediate; the product exhibits low solubility in the ester solvent at reduced temperatures (0-5°C), allowing it to precipitate directly from the reaction mixture with high purity (>99.50%) without the need for complex chromatographic purification.
This strategic shift in solvent chemistry not only enhances operator safety but also dramatically improves the economic profile of the synthesis. The total yield of the process has been reported to exceed 70%, a nearly threefold improvement over the earliest conventional methods. By avoiding high-boiling, toxic solvents like NMP and DMF, the process facilitates easier solvent recycling and reduces the energy load required for distillation. For procurement managers, this translates to a more resilient supply chain less dependent on restricted chemicals, while for R&D teams, it offers a streamlined path to clinical-grade material with a significantly cleaner impurity profile, effectively removing the difficult-to-control dimer impurities often associated with older synthetic routes.
Mechanistic Insights into Ester-Mediated Crystallization
The core technical breakthrough of this patent lies in the precise manipulation of solubility parameters using ester solvents to drive both reaction efficiency and purification. In the initial alkylation step, the use of ethyl acetate or similar esters creates a reaction environment where the intermediate, 2-((6-chloro-3-methyl-2,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)methyl)benzonitrile, remains soluble at elevated temperatures (70-80°C) but becomes sparingly soluble upon cooling. This thermodynamic behavior allows for a "reaction-crystallization" coupling, where the product spontaneously precipitates as the system cools to 0-5°C. This phenomenon effectively purifies the intermediate in situ, washing away unreacted starting materials and organic base residues which remain dissolved in the mother liquor or are removed during the subsequent aqueous wash steps.
Furthermore, the subsequent substitution reaction with (R)-3-aminopiperidine dihydrochloride is conducted in a mixed solvent system of isopropanol and water. This biphasic-friendly environment, combined with the use of mild inorganic bases like potassium carbonate, ensures high stereoselectivity and minimizes side reactions. The patent explicitly notes the surprising elimination of dimer impurities, which are structurally related byproducts that are notoriously difficult to separate in traditional processes. By controlling the pH and solvent composition during the workup—specifically the extraction into chloroform followed by acidification—the process selectively isolates the desired Alogliptin Hydrochloride salt. This rigorous control over the physical chemistry of the system ensures that the final salification with benzoic acid yields a product with HPLC purity greater than 99.95%, meeting the most stringent pharmaceutical specifications without requiring recrystallization.
How to Synthesize Alogliptin Benzoate Efficiently
The synthesis of Alogliptin Benzoate via this optimized route involves three distinct operational stages that prioritize safety and yield. The process begins with the alkylation of 6-chloro-3-methyluracil in an ester solvent, followed by a nucleophilic substitution in an alcohol-water mixture, and concludes with salt formation. Each step is designed to maximize material throughput while minimizing waste generation. The detailed standardized operating procedures, including specific stoichiometric ratios, temperature ramping rates, and agitation speeds required for successful replication of this high-yield pathway, are outlined in the technical guide below.
- React 6-chloro-3-methyluracil with 2-cyanobenzyl bromide in an ester solvent (e.g., ethyl acetate) using an organic base to obtain the chloro-intermediate with >99.5% purity.
- Perform nucleophilic substitution with (R)-3-aminopiperidine dihydrochloride in an isopropanol/water mixture using an inorganic base, followed by acidification to isolate Alogliptin Hydrochloride.
- Convert the hydrochloride salt to the free base using sodium carbonate, then react with benzoic acid in an alcohol solvent to crystallize the final Alogliptin Benzoate product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in Patent CN107540656B offers profound strategic advantages beyond mere technical elegance. The primary value driver is the substantial reduction in raw material and waste disposal costs. By eliminating the need for hazardous reagents like sodium hydride and methyl iodide, facilities can significantly lower their insurance premiums and safety infrastructure investments. Furthermore, the replacement of NMP and DMF with common esters like ethyl acetate simplifies solvent procurement, as these are commodity chemicals with stable pricing and abundant global availability, reducing the risk of supply disruptions caused by regulatory bans on specialized solvents.
- Cost Reduction in Manufacturing: The shift to an ester-based solvent system fundamentally alters the cost structure of Alogliptin Benzoate production. Traditional methods require energy-intensive distillation to recover high-boiling solvents like NMP, whereas ethyl acetate can be recovered and reused with significantly lower energy input due to its lower boiling point. Additionally, the dramatic increase in overall yield from approximately 25% to over 70% means that less raw material is required to produce the same amount of API, directly lowering the variable cost per unit. The simplified workup, which relies on filtration rather than complex extractions or chromatography, further reduces labor hours and consumable costs associated with purification media.
- Enhanced Supply Chain Reliability: Relying on restricted substances like NMP poses a long-term risk to supply continuity as global regulations tighten. This new process utilizes widely available, non-restricted solvents and reagents, ensuring a more robust and future-proof supply chain. The ability to source raw materials from multiple vendors without being locked into specialized chemical suppliers enhances negotiating power and mitigates the risk of single-source failure. Moreover, the simplified process flow reduces the lead time for batch production, allowing for more responsive inventory management and faster turnaround times for custom orders.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is markedly superior. The reduction in toxic waste streams simplifies wastewater treatment and lowers the cost of environmental compliance. The process is inherently safer, reducing the potential for catastrophic incidents associated with pyrophoric reagents. This makes the technology highly scalable, as the safety constraints that often limit batch sizes in traditional processes are largely removed. Facilities can confidently scale production from pilot batches to multi-ton commercial campaigns without encountering the diminishing returns often associated with scaling hazardous chemistries.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this optimized synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on the practical benefits of transitioning to this newer methodology. Understanding these nuances is critical for technical teams evaluating the feasibility of technology transfer.
Q: Why is the ester solvent method superior to traditional NMP or DMF routes for Alogliptin synthesis?
A: Traditional routes utilize hazardous reagents like sodium hydride and carcinogenic methyl iodide, or toxic high-boiling solvents like NMP which are difficult to recover. The ester solvent method (Patent CN107540656B) eliminates these hazards, allows for easy product precipitation via temperature control, and significantly reduces environmental compliance costs.
Q: What purity levels can be achieved with this optimized crystallization process?
A: The optimized process achieves exceptional purity levels, with the final Alogliptin Benzoate product reaching HPLC purity greater than 99.95%. Crucially, it effectively eliminates difficult-to-remove dimer impurities often found in prior art methods, meeting strict ICH guidelines for unknown single impurities (<0.10%).
Q: How does this process impact the overall yield compared to conventional methods?
A: Conventional methods reported in earlier patents often suffer from low total yields ranging between 20-25%. In contrast, the improved ester-solvent route described in this patent demonstrates a total yield exceeding 70%, representing a substantial increase in material efficiency and production throughput.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alogliptin Benzoate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced manufacturing processes requires a partner with deep technical expertise and proven scalability. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of Patent CN107540656B are fully realized in a commercial setting. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Alogliptin Benzoate meets or exceeds the >99.95% purity benchmark established by this innovative process.
We invite pharmaceutical partners to engage with our technical procurement team to discuss how this optimized route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the specific economic benefits relevant to your production volume. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of safety, efficiency, and superior quality.
