Scalable Metal-Free Synthesis of Tetrahydroindole Scaffolds for Next-Generation Drug Discovery
The pharmaceutical industry continuously seeks robust synthetic methodologies that balance structural complexity with economic and environmental sustainability. A pivotal advancement in this domain is documented in patent CN103467356A, which discloses a novel preparation method for tetrahydroindole compounds. These heterocyclic scaffolds are ubiquitous in medicinal chemistry, serving as critical cores for agents exhibiting anxiolytic, antihypertensive, and potent antitumor activities. The disclosed technology leverages a domino cyclization strategy catalyzed by L-proline in an aqueous medium under microwave irradiation. This approach represents a paradigm shift from traditional transition-metal-dependent syntheses, offering a pathway to high-purity intermediates with significantly reduced environmental footprints. For R&D directors and procurement strategists, understanding the nuances of this metal-free, water-based protocol is essential for optimizing supply chains and reducing the cost of goods sold (COGS) in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indole nucleus has relied heavily on classical transformations such as the Fischer indole synthesis or various metal-catalyzed cyclizations. While effective in academic settings, these conventional methods present substantial hurdles for industrial application. The Fischer synthesis, for instance, often suffers from poor regioselectivity and low yields when applied to complex, multi-substituted substrates, leading to difficult purification challenges and increased material waste. Furthermore, many modern alternatives utilize expensive and toxic transition metal catalysts, such as palladium, copper, or rhodium complexes. The presence of these metals necessitates rigorous downstream processing to meet stringent regulatory limits for residual metals in pharmaceutical products, typically requiring parts-per-million (ppm) levels. This additional purification step not only inflates production costs but also introduces potential bottlenecks in the supply chain. Additionally, the reliance on volatile organic solvents like dichloromethane or toluene exacerbates environmental compliance issues and increases the hazard profile of the manufacturing facility.
The Novel Approach
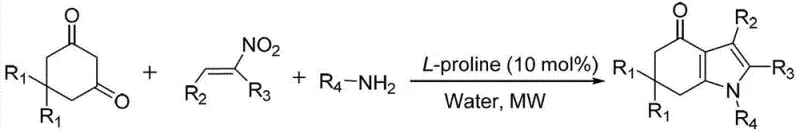
In stark contrast, the methodology outlined in CN103467356A offers a streamlined, green chemistry solution that directly addresses these pain points. By employing a one-pot, three-component domino reaction involving 1,3-cyclohexanedione derivatives, nitroalkenes, and amines, the process achieves rapid assembly of the tetrahydroindole core with high atom economy. The use of L-proline as an inexpensive, non-toxic organocatalyst eliminates the risk of heavy metal contamination entirely, thereby simplifying the quality control workflow. Moreover, the reaction proceeds efficiently in water, a benign and non-flammable solvent, under mild microwave irradiation at 80°C. This combination of factors results in a process that is not only chemically efficient but also operationally superior, minimizing solvent recovery costs and reducing the overall environmental impact of the synthesis.
Mechanistic Insights into L-Proline Catalyzed Domino Cyclization
The mechanistic elegance of this transformation lies in the ability of L-proline to activate carbonyl components through enamine or iminium ion intermediates, facilitating the cascade of bond-forming events without external metallic assistance. The reaction initiates with the condensation of the amine and the 1,3-dicarbonyl compound or the nitroalkene, driven by the organocatalyst. This is followed by a Michael addition and subsequent intramolecular cyclization to forge the pyrrole ring fused to the cyclohexanone moiety. The microwave irradiation plays a critical role in accelerating these steps, providing uniform heating that enhances reaction kinetics and suppresses side reactions often seen in thermal convection heating. This precise control over reaction parameters ensures high conversion rates and minimizes the formation of polymeric by-products, which are common in uncontrolled polycondensation reactions.
From an impurity control perspective, the absence of transition metals is a decisive advantage for pharmaceutical manufacturing. Metal residues can act as catalysts for degradation pathways during drug storage, compromising the stability and shelf-life of the final API. By utilizing an organocatalytic system, the impurity profile of the resulting tetrahydroindole derivatives is significantly cleaner, primarily consisting of unreacted starting materials or simple hydrolysis products that are easily removed via standard extraction or crystallization techniques. The use of water as the reaction medium further aids in impurity management, as many organic by-products are insoluble in the aqueous phase and can be separated via filtration or phase separation. This inherent purity reduces the burden on analytical laboratories and accelerates the release of batches for clinical or commercial use.
How to Synthesize Tetrahydroindole Derivatives Efficiently
The practical implementation of this synthesis route is designed for operational simplicity, making it accessible for both laboratory-scale optimization and pilot-plant production. The process begins with the preparation of the nitroalkene precursor, followed by the one-pot assembly of the final scaffold. The standardized protocol ensures reproducibility across different batches, a critical factor for regulatory compliance. Detailed below is the strategic overview of the synthesis steps, highlighting the critical parameters for maximizing yield and purity while maintaining safety standards.
- Prepare nitroalkene precursors by condensing aromatic aldehydes with nitroethane using ammonium acetate in acetic acid.
- Combine 1,3-cyclohexanedione, the prepared nitroalkene, and an amine in water with 10 mol% L-proline catalyst.
- Subject the mixture to microwave irradiation at 80°C for 8-15 minutes, followed by extraction and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic benefits beyond mere chemical yield. The shift from metal-catalyzed to organocatalytic processes fundamentally alters the cost structure of intermediate manufacturing. By eliminating the need for precious metal catalysts, companies can avoid the volatility associated with the pricing of commodities like palladium or platinum. Furthermore, the simplified workup procedure reduces the consumption of auxiliary chemicals and solvents required for metal scavenging, leading to substantial cost savings in raw material procurement. The use of water as a primary solvent also mitigates the costs associated with solvent recovery systems and hazardous waste disposal, contributing to a leaner and more sustainable operational model.
- Cost Reduction in Manufacturing: The economic advantages of this protocol are driven by the replacement of expensive transition metal catalysts with L-proline, a commodity chemical available at a fraction of the cost. This substitution removes the necessity for specialized metal-scavenging resins or complex filtration units, thereby lowering capital expenditure (CAPEX) and operational expenditure (OPEX). Additionally, the high atom economy of the domino reaction means that a greater proportion of the input mass is converted into the desired product, reducing the cost per kilogram of the active intermediate. The energy efficiency gained through microwave-assisted heating further contributes to lower utility costs compared to prolonged thermal reflux methods.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available, non-hazardous raw materials. 1,3-cyclohexanediones, nitroalkenes, and amines are commercially abundant, reducing the risk of supply disruptions caused by the scarcity of specialized reagents. The robustness of the reaction conditions—tolerant to moisture and oxygen due to the aqueous medium—simplifies logistics and storage requirements. There is no need for inert atmosphere handling or anhydrous solvents, which streamlines the manufacturing workflow and reduces the lead time for high-purity pharmaceutical intermediates. This reliability ensures consistent delivery schedules to downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage levels is facilitated by the simplicity of the reaction setup. The absence of exothermic risks associated with strong Lewis acids or pyrophoric reagents enhances process safety, a key consideration for large-scale production. Environmentally, the process aligns with green chemistry principles by minimizing volatile organic compound (VOC) emissions. Water, being non-toxic and non-flammable, simplifies regulatory compliance regarding air quality and worker safety. The ease of product isolation via extraction or crystallization from the aqueous phase ensures that the process remains efficient even at commercial scales, supporting the continuous supply of complex heterocycles needed for global drug markets.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this synthesis route into their existing portfolios, we have compiled answers to common inquiries regarding the process specifics and its implications for quality and scale. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making.
Q: Does this synthesis method involve heavy metal catalysts?
A: No, the process described in patent CN103467356A utilizes L-proline, an organocatalyst, eliminating the need for toxic transition metals and simplifying purification.
Q: What are the environmental benefits of using water as a solvent?
A: Using water eliminates volatile organic compounds (VOCs), reduces flammability risks, and simplifies waste treatment compared to traditional organic solvents like toluene or DCM.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the one-pot nature, mild conditions (80°C), and absence of sensitive metal catalysts make this route highly adaptable for commercial scale-up and continuous flow processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydroindole Supplier
The technological potential of the L-proline catalyzed domino cyclization represents a significant opportunity for advancing the synthesis of bioactive indole derivatives. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative academic protocols are successfully translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs, guaranteeing that every batch of tetrahydroindole intermediate meets the exacting standards required for pharmaceutical applications. We understand the critical nature of supply continuity and are equipped to handle the complexities of multi-step organic synthesis with precision and reliability.
We invite you to collaborate with us to optimize your supply chain for these valuable scaffolds. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our expertise in green chemistry and process intensification, we can help you achieve significant efficiencies in your API manufacturing. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our capabilities can support your long-term strategic goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
