Advanced Sulfuric Acid Hydrolysis Route for High-Purity DL-Pantolactone Manufacturing
The global demand for Vitamin B5 derivatives, specifically D-Panthenol and Calcium D-Pantothenate, continues to surge across the pharmaceutical, nutritional, and cosmetic sectors, driving an urgent need for more efficient synthesis of their key precursor, DL-Pantolactone. Patent CN114773295A introduces a transformative methodology that addresses the longstanding environmental and economic bottlenecks associated with traditional pantolactone manufacturing. This technical insight report analyzes the novel sulfuric acid-mediated hydrolysis pathway, which replaces conventional hydrochloric acid processes to achieve significant reductions in wastewater generation and energy consumption. By eliminating the need for extensive organic solvent extraction and enabling the recovery of ammonium sulfate as a saleable byproduct, this innovation represents a paradigm shift towards greener chemistry in fine chemical production. For R&D directors and supply chain leaders, understanding the mechanistic advantages of this route is critical for securing a sustainable and cost-effective supply of high-purity vitamin intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of DL-pantolactone has relied heavily on the isobutyraldehyde-formaldehyde-hydrocyanic acid route, which, while chemically sound, suffers from severe downstream processing inefficiencies. The traditional protocol typically employs dilute hydrochloric acid for the hydrolysis of the cyanohydrin intermediate, a step that generates massive quantities of saline wastewater that are notoriously difficult and expensive to treat. Furthermore, the isolation of the target lactone from the acidic aqueous phase necessitates the use of large volumes of organic solvents, such as ethyl acetate, ketones, or halogenated hydrocarbons, with consumption rates reported to be as high as 5 tons of solvent per ton of product. This heavy reliance on solvent extraction not only inflates operational expenditures due to solvent purchase and recovery costs but also introduces significant safety hazards related to volatile organic compound (VOC) emissions and fire risks. The cumulative effect of these factors results in a process with a large environmental footprint and compromised economic viability in an increasingly regulated market.
The Novel Approach
In stark contrast to the legacy hydrochloric acid method, the process disclosed in CN114773295A utilizes concentrated sulfuric acid for the hydrolysis and esterification steps, fundamentally altering the phase behavior of the reaction mixture. This strategic substitution allows the reaction system to undergo a clean phase separation upon neutralization with ammonia or alkali, effectively partitioning the DL-pantolactone into an organic layer without the need for external extraction solvents. The aqueous phase, rich in ammonium sulfate, can be further processed to recover crystalline ammonium sulfate, turning a potential waste stream into a value-added co-product. This approach not only simplifies the unit operations by removing the extraction and solvent recovery columns but also drastically reduces the volume of effluent requiring treatment. The result is a streamlined workflow that enhances overall atom economy and aligns with modern principles of sustainable manufacturing, offering a compelling alternative for producers seeking to optimize their pantolactone supply chains.
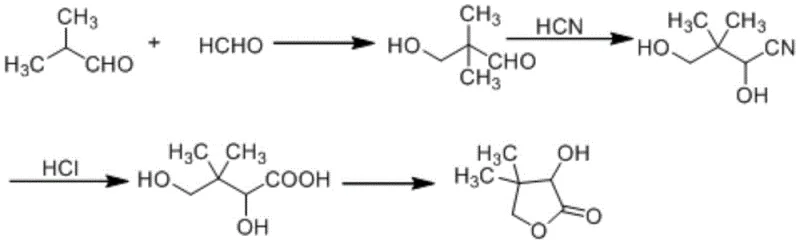
Mechanistic Insights into Sulfuric Acid Catalyzed Hydrolysis and Cyclization
The core chemical innovation lies in the precise control of the hydrolysis and subsequent lactonization steps using sulfuric acid as the catalytic and dehydrating agent. In the initial stages, formaldehyde and isobutyraldehyde undergo a base-catalyzed aldol condensation to form hydroxypivalaldehyde, which is then converted to 2,4-dihydroxy-3,3-dimethylbutyronitrile via reaction with hydrocyanic acid. The critical divergence from prior art occurs when sulfuric acid is introduced to this nitrile intermediate; the strong acid facilitates the hydrolysis of the cyano group to a carboxylic acid while simultaneously promoting intramolecular esterification to form the five-membered lactone ring. Unlike hydrochloric acid, which forms highly soluble ammonium chloride salts that remain in the aqueous phase and complicate product isolation, the sulfate salts formed exhibit different solubility characteristics that favor phase separation. This physicochemical property is exploited to allow the DL-pantolactone to naturally separate into an upper organic phase upon neutralization, enabling direct rectification of the crude product.
Furthermore, the impurity profile of the final product is significantly improved due to the absence of solvent-induced side reactions and the efficient removal of inorganic salts during the phase separation. In traditional solvent extraction processes, trace amounts of esters or ketones can often persist in the final product, requiring rigorous distillation to meet pharmaceutical grade specifications. The solvent-free nature of this new method minimizes the introduction of extraneous organic impurities, thereby reducing the burden on the final purification steps. Additionally, the recovery of ammonium sulfate from the brine phase serves as an effective purification mechanism for the aqueous stream, ensuring that the waste discharge meets stringent environmental standards. This dual benefit of high product purity and effective byproduct management underscores the robustness of the sulfuric acid route for large-scale commercial application.
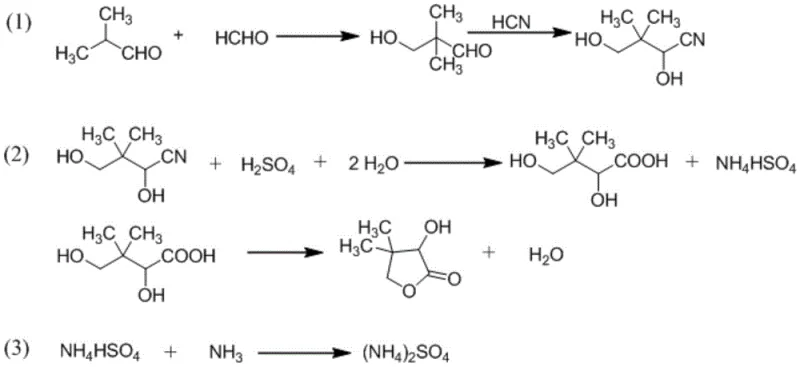
How to Synthesize DL-Pantolactone Efficiently
The synthesis of DL-pantolactone via this optimized route involves a sequence of well-defined chemical transformations that prioritize operational simplicity and yield maximization. The process begins with the condensation of readily available aldehydes, followed by cyanation and a unified hydrolysis-lactonization step that eliminates the need for intermediate isolation. Detailed standard operating procedures regarding temperature controls, molar ratios, and safety protocols for handling hydrocyanic acid are essential for successful implementation. For a comprehensive guide on the specific reaction conditions and workup procedures described in the patent literature, please refer to the technical breakdown below.
- Perform aldol condensation between formaldehyde and isobutyraldehyde using a base catalyst to form hydroxypivalaldehyde.
- React the hydroxypivalaldehyde with hydrocyanic acid to generate 2,4-dihydroxy-3,3-dimethylbutyronitrile.
- Add sulfuric acid for hydrolysis and esterification, followed by ammonia neutralization to separate the organic phase containing DL-pantolactone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN114773295A offers substantial strategic advantages centered around cost structure optimization and supply continuity. The elimination of organic solvent extraction represents a direct reduction in variable costs, as it removes the expense of purchasing tons of extraction solvents and the energy costs associated with their distillation and recovery. This simplification of the process flow also reduces the maintenance burden on equipment, as fewer columns and pumps are required, leading to lower capital expenditure for new facilities or retrofitting existing ones. Moreover, the ability to sell ammonium sulfate as a byproduct creates a new revenue stream that offsets raw material costs, further enhancing the overall margin profile of pantolactone production. These factors combine to create a more resilient supply chain that is less vulnerable to fluctuations in solvent prices and waste disposal fees.
- Cost Reduction in Manufacturing: The most significant financial impact stems from the complete removal of the solvent extraction unit operation, which traditionally accounts for a major portion of processing costs in pantolactone synthesis. By avoiding the use of approximately 5 tons of organic solvent per ton of product, manufacturers can achieve drastic savings in both raw material procurement and utility consumption for solvent recovery. Additionally, the high yield of 90.6% reported in the patent examples indicates superior raw material utilization, meaning less feedstock is wasted as byproducts. This efficiency translates directly into a lower cost of goods sold (COGS), allowing suppliers to offer more competitive pricing in the global vitamin market while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: Relying on a process that generates less hazardous waste and requires fewer complex separation steps inherently reduces the risk of production stoppages due to environmental compliance issues or equipment failure. The simplified workflow minimizes the number of potential failure points in the manufacturing line, ensuring a more consistent output of high-purity DL-pantolactone. Furthermore, the use of common industrial chemicals like sulfuric acid and ammonia, rather than specialized extraction solvents, ensures that raw material availability remains stable even during supply chain disruptions. This reliability is crucial for downstream customers who require uninterrupted supplies of Vitamin B5 precursors for their own continuous manufacturing processes.
- Scalability and Environmental Compliance: As regulatory pressures regarding industrial wastewater discharge intensify globally, the ability to significantly reduce wastewater volume provides a distinct competitive advantage. The conversion of waste salts into recoverable ammonium sulfate aligns with circular economy principles, making the facility more attractive to investors and regulators alike. This environmental stewardship facilitates easier permitting for capacity expansions and reduces the long-term liability associated with waste treatment. Consequently, manufacturers utilizing this technology are better positioned to scale up production to meet growing market demand without facing the prohibitive costs of expanding wastewater treatment infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel pantolactone synthesis method. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity on how this technology compares to established industry standards. Understanding these nuances is essential for stakeholders evaluating the feasibility of adopting this greener manufacturing route.
Q: How does the sulfuric acid method reduce wastewater compared to traditional HCl hydrolysis?
A: Traditional methods using hydrochloric acid generate large volumes of saline wastewater that are difficult to treat. The patented sulfuric acid route allows for the recovery of ammonium sulfate as a valuable byproduct through crystallization, significantly reducing liquid waste discharge.
Q: Is organic solvent extraction required in this new pantolactone synthesis process?
A: No. A major advantage of this process is the elimination of organic solvent extraction steps (such as ethyl acetate) typically used to isolate the product. The product separates directly into the organic phase after neutralization, drastically lowering energy consumption and solvent loss.
Q: What is the expected purity and yield of DL-pantolactone using this method?
A: According to the experimental data in patent CN114773295A, the process achieves a yield of approximately 90.6% with a product purity reaching 99.0%, demonstrating high efficiency suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable DL-Pantolactone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthesis routes like the one described in CN114773295A requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO specializing in complex pharmaceutical intermediates, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific safety requirements of cyanation chemistry while maintaining stringent purity specifications through our rigorous QC labs. We are committed to delivering high-quality DL-pantolactone that meets the exacting standards of the global vitamin and nutraceutical industries, ensuring that your supply chain remains secure and compliant.
We invite you to engage with our technical team to explore how this innovative synthesis method can be tailored to your specific production needs. By leveraging our process development capabilities, we can provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this solvent-free route for your operations. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you optimize your Vitamin B5 supply chain for the future.
