Advanced Synthesis of Soluble Tetraalkylphthalocyanines for High-Performance Optoelectronic Manufacturing
The landscape of organic electronics is undergoing a paradigm shift, driven by the urgent need to transition from costly vacuum deposition methods to more economical solution-processing techniques. Patent CN101255163A introduces a groundbreaking class of soluble tetraalkylphthalocyanine compounds that address the critical solubility limitations inherent in traditional phthalocyanine dyes. By strategically introducing alkyl substituents at specific positions on the phthalocyanine ring, this technology enables the fabrication of high-performance organic semiconductor layers using standard coating methods. For R&D directors and procurement specialists in the optoelectronic sector, this represents a significant opportunity to reduce manufacturing complexity while maintaining the exceptional stability and charge transport properties characteristic of the phthalocyanine core. The ability to process these materials in common organic solvents such as dichloromethane and tetrahydrofuran opens new avenues for the mass production of organic thin-film transistors (OTFTs) and photovoltaic devices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the deployment of phthalocyanine compounds in electronic devices has been severely constrained by their poor solubility in most organic media. Traditional unsubstituted metal phthalocyanines, such as copper phthalocyanine (CuPc) or zinc phthalocyanine (ZnPc), exhibit strong intermolecular pi-pi stacking interactions that lead to aggregation and precipitation. Consequently, the industry has relied heavily on vacuum thermal evaporation to deposit thin films, a process that requires sophisticated, capital-intensive equipment and operates under high vacuum conditions. This method not only incurs substantial energy costs but also limits the throughput and scalability required for large-area electronics like flexible displays or solar panels. Furthermore, the inability to use solution-based patterning techniques restricts the design flexibility and increases the overall cost of ownership for manufacturing facilities seeking to optimize their production lines for next-generation organic semiconductors.
The Novel Approach
The innovation detailed in CN101255163A circumvents these obstacles through a clever molecular engineering strategy that modifies the periphery of the phthalocyanine macrocycle. By synthesizing 4-alkylphthalonitrile precursors and subsequently cyclizing them, the resulting tetraalkylphthalocyanine compounds possess long alkyl chains (ranging from C4 to C18) that sterically hinder close packing of the aromatic cores. This structural modification dramatically improves solubility without compromising the intrinsic photoelectric properties of the molecule. The new approach allows for the preparation of uniform semiconductor layers via spin-coating, inkjet printing, or roll-to-roll processing, which are far more cost-effective and scalable than vacuum deposition. This shift not only simplifies the device fabrication workflow but also broadens the applicability of phthalocyanine materials in emerging fields such as printable electronics and low-cost sensors, offering a robust pathway for commercial scale-up of complex optoelectronic materials.
Mechanistic Insights into Alkyl-Substituted Phthalocyanine Synthesis
The synthesis mechanism relies on a multi-step organic transformation that begins with the functionalization of the phthalonitrile core. The process initiates with the conversion of commercially available 4-aminophthalonitrile into 4-iodophthalonitrile via a diazotization reaction followed by treatment with potassium iodide. This iodinated intermediate serves as a crucial coupling partner in a Sonogashira cross-coupling reaction, where it reacts with terminal alkyl alkynes in the presence of a palladium catalyst, specifically tetrakis(triphenylphosphine)palladium(0), and a copper co-catalyst. This step effectively installs the carbon chain precursor onto the aromatic ring. Subsequent catalytic hydrogenation using palladium on carbon (Pd/C) reduces the triple bond to a single bond, yielding the key 4-alkylphthalonitrile building block. The final cyclotetramerization involves heating four equivalents of this nitrile with a metal source (such as zinc acetate or tin chloride) and an organic base like DBU in a high-boiling solvent like n-hexanol. This condensation reaction forms the stable, conjugated macrocyclic structure essential for electronic applications.
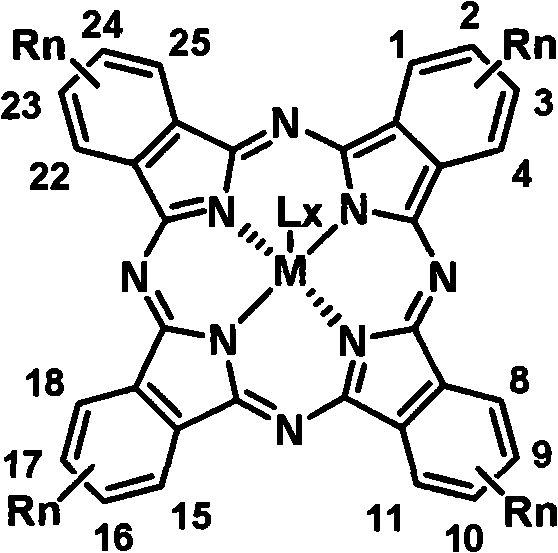
Controlling impurities during this synthesis is paramount for ensuring high charge carrier mobility in the final device. The use of column chromatography with specific eluent systems, such as petroleum ether mixed with ethyl acetate or chloroform, allows for the precise separation of the target tetraalkylphthalocyanine from unreacted precursors and oligomeric byproducts. The patent highlights that the position of the alkyl substitution—specifically at the 2, 9, 16, 23 or 3, 10, 17, 24 positions—is critical for maintaining symmetry and preventing the formation of regioisomers that could disrupt crystal packing in the solid state. Furthermore, the choice of the central metal ion (M) and optional axial ligands (L) provides an additional layer of tunability, enabling chemists to fine-tune the HOMO-LUMO energy levels to match specific device requirements. This rigorous control over molecular architecture ensures that the resulting high-purity optoelectronic material meets the stringent specifications demanded by advanced semiconductor applications.
How to Synthesize Soluble Tetraalkylphthalocyanine Efficiently
The preparation of these advanced materials follows a logical sequence of reactions designed to maximize yield and purity while utilizing standard laboratory equipment. The process leverages well-established coupling and cyclization chemistries that are familiar to process chemists, facilitating a smoother transition from bench-scale optimization to pilot plant operations. Detailed below is the standardized workflow derived from the patent examples, which outlines the critical parameters for successful synthesis.
- Prepare 4-iodophthalonitrile via diazotization of 4-aminophthalonitrile followed by reaction with potassium iodide.
- Perform Sonogashira coupling between 4-iodophthalonitrile and alkyl alkynes using Pd(PPh3)4 and CuI catalysts to form 4-(1-alkylalkynyl)phthalonitrile.
- Hydrogenate the alkyne intermediate using Pd/C catalyst to obtain 4-alkylphthalonitrile, then cyclize with metal salts and DBU base.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this soluble phthalocyanine technology offers compelling economic and logistical benefits that extend beyond simple material performance. The ability to process these semiconductors from solution eliminates the need for expensive vacuum evaporation infrastructure, representing a drastic reduction in capital expenditure for manufacturing facilities. Moreover, the synthetic route utilizes readily available starting materials and common reagents, mitigating supply chain risks associated with exotic or highly specialized precursors. This accessibility ensures a more stable and reliable optoelectronic material supplier network, reducing the likelihood of production delays caused by raw material shortages. The simplified processing conditions also translate to lower energy consumption and reduced operational overhead, contributing to a more sustainable and cost-efficient production model.
- Cost Reduction in Manufacturing: The transition from vacuum deposition to solution processing fundamentally alters the cost structure of organic electronic device fabrication. By removing the requirement for high-vacuum chambers and thermal evaporators, manufacturers can significantly lower both equipment maintenance costs and energy usage. Additionally, the high conversion rates observed in the hydrogenation and cyclization steps minimize waste generation, further enhancing the economic viability of the process. The elimination of complex purification steps often required for insoluble pigments also streamlines the downstream processing, leading to substantial cost savings in the overall manufacturing workflow without compromising on the quality of the final electronic grade material.
- Enhanced Supply Chain Reliability: The synthetic pathway described relies on robust chemical transformations that are less sensitive to minor fluctuations in reaction conditions compared to delicate vacuum processes. This robustness ensures consistent batch-to-batch quality, which is critical for maintaining long-term supply contracts with major electronics manufacturers. The use of common organic solvents and catalysts means that sourcing is not restricted to niche vendors, thereby diversifying the supply base and reducing dependency on single-source suppliers. This flexibility allows for better inventory management and faster response times to market demands, ensuring that the delivery of high-purity phthalocyanine derivatives remains uninterrupted even during periods of global supply chain volatility.
- Scalability and Environmental Compliance: The methodology is inherently scalable, as the reaction conditions (reflux temperatures, atmospheric pressure hydrogenation) are easily replicated in larger reactors without requiring specialized high-pressure or ultra-high vacuum equipment. This ease of scale-up facilitates rapid capacity expansion to meet growing market demand for organic semiconductors. From an environmental perspective, the process avoids the use of harsh halogenated solvents in the final purification stages where possible, and the efficient use of catalysts reduces heavy metal waste. These factors contribute to a greener manufacturing profile, aligning with increasingly strict global environmental regulations and corporate sustainability goals while maintaining high production efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this soluble phthalocyanine technology. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, offering clarity on performance metrics and application suitability.
Q: What is the primary advantage of tetraalkylphthalocyanines over traditional phthalocyanines?
A: The introduction of alkyl chains (C4-C18) significantly enhances solubility in common organic solvents like chloroform and THF, enabling solution-processing techniques such as spin-coating instead of expensive vacuum evaporation.
Q: Which metals can be coordinated in the center of this phthalocyanine structure?
A: The patent specifies a wide range of central metals including Mg, Fe, Co, Ni, Cu, Zn, Sn, Al, Si, Ti, V, Mn, Ga, and In, allowing for tunable electronic and optical properties.
Q: Is the synthesis scalable for industrial production?
A: Yes, the process utilizes standard organic synthesis techniques like column chromatography and refluxing in common solvents, which are readily adaptable from laboratory to pilot and commercial scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetraalkylphthalocyanine Supplier
As the demand for solution-processable organic semiconductors continues to surge, partnering with an experienced chemical manufacturer is essential for securing a competitive edge. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of tetraalkylphthalocyanine delivered meets the exacting standards required for high-performance optoelectronic devices. We understand the critical nature of material consistency in semiconductor manufacturing and are committed to providing a stable, high-quality supply chain.
We invite you to collaborate with our technical team to explore how this innovative synthesis route can be integrated into your specific product lines. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your production volume. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and procurement decision-making processes, ensuring a seamless transition to next-generation organic electronic materials.
