Advanced Synthesis of Tetrabromoperylene Derivatives for High-Performance Organic Electronics Manufacturing
Introduction to Novel Perylene-Based Semiconductor Intermediates
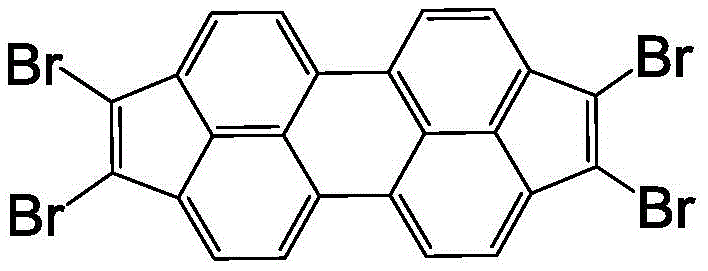
The rapid evolution of the organic electronics sector demands increasingly sophisticated molecular architectures capable of delivering superior charge carrier mobility and thermal stability. Patent CN110937977A introduces a significant breakthrough in this domain by disclosing a novel preparation method for 1,2,7,8-tetrabromobicyclopenteno[cd,lm]perylene, a critical structural unit for next-generation organic photoelectric and thermoelectric materials. This compound serves as a pivotal building block for constructing high-performance organic semiconductors used in field-effect transistors (OFETs), organic light-emitting diodes (OLEDs), and organic photovoltaics (OPV). The strategic incorporation of bromine atoms onto the dicyclopenteno-perylene core not only enhances the electron-withdrawing character of the molecule but also provides versatile reactive sites for further functionalization via cross-coupling reactions. As the industry shifts towards more efficient and scalable manufacturing processes, this patented technology offers a compelling solution for producing high-purity intermediates with exceptional consistency.
![Chemical structure of 1,2,7,8-tetrabromobicyclopenteno[cd,lm]perylene showing the extended conjugated system](/insights/img/tetrabromoperylene-synthesis-oled-supplier-20260305113957-01.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of polycyclic aromatic hydrocarbons like perylene has been fraught with challenges related to regioselectivity and reaction severity. Traditional halogenation protocols often rely on aggressive Lewis acid catalysts or elemental halogens, which can lead to uncontrolled poly-substitution, degradation of the sensitive aromatic core, and the generation of complex impurity profiles that are difficult to separate. Furthermore, many existing routes to functionalized dicyclopenteno-perylene derivatives require multi-step synthetic sequences involving protection and de-protection strategies, which drastically reduce overall atom economy and increase production costs. These conventional approaches frequently suffer from low yields and poor reproducibility when scaled beyond the laboratory bench, creating significant bottlenecks for supply chain managers who require reliable volumes of electronic-grade chemicals. The reliance on hazardous reagents also imposes stringent environmental compliance burdens, complicating waste disposal and increasing the total cost of ownership for manufacturing facilities.
The Novel Approach
In stark contrast, the methodology outlined in patent CN110937977A presents a streamlined, one-step radical bromination strategy that elegantly bypasses these historical inefficiencies. By utilizing 1,2,7,8-tetrahydrodicyclopentano[cd,lm]perylene as the starting material and employing peroxides as radical initiators, the process achieves direct conversion to the tetrabromo product with remarkable efficiency. This approach leverages the inherent reactivity of the allylic-like positions within the fused five-membered rings, facilitating simultaneous dehydrogenation and bromination to establish the fully conjugated system in a single operation. The versatility of this method is underscored by its compatibility with various brominating agents such as N-bromosuccinimide (NBS) and dibromohydantoin (DBDMH), allowing process chemists to optimize reagent costs based on market availability. The result is a robust synthetic route that delivers yields exceeding 80%, significantly outperforming traditional multi-step alternatives while simplifying the downstream purification workflow.
Mechanistic Insights into Peroxide-Catalyzed Radical Bromination
The core innovation of this synthesis lies in the mechanistic pathway driven by peroxide-catalyzed radical generation, which ensures high selectivity for the target tetrabromo species. Upon heating the reaction mixture to temperatures between 60°C and 90°C, the peroxide catalyst, such as dibenzoyl peroxide (BPO) or cyclohexanone peroxide, undergoes homolytic cleavage to generate free radical species. These radicals abstract hydrogen atoms from the saturated five-membered rings of the tetrahydro-perylene precursor, creating stable carbon-centered radicals at the bridgehead positions. Subsequently, these carbon radicals react with the brominating agent to introduce bromine atoms, followed by a second abstraction and bromination event that ultimately leads to the formation of the double bonds observed in the final product. This cascade of radical substitutions and eliminations effectively aromatizes the five-membered rings, extending the pi-conjugation across the entire molecular framework. The precise control over reaction temperature and stoichiometry prevents over-bromination of the central perylene core, ensuring that the substitution is confined strictly to the peripheral cyclopentene moieties.
From an impurity control perspective, this radical mechanism offers distinct advantages over electrophilic aromatic substitution, which is prone to generating isomeric byproducts. The thermodynamic stability of the resulting conjugated tetrabromo system drives the reaction equilibrium towards the desired product, minimizing the formation of partially brominated intermediates or regio-isomers. Moreover, the use of aprotic solvents like carbon tetrachloride, chloroform, THF, or DMF creates an inert environment that suppresses side reactions such as hydrolysis or oxidation of the sensitive aromatic system. The simplicity of the workup procedure, involving filtration and recrystallization, further attests to the cleanliness of the reaction profile, as the byproduct succinimide or hydantoin derivatives are easily removed during the aqueous washing steps. This high level of chemical fidelity is crucial for R&D directors who require intermediates with defined impurity spectra to ensure consistent device performance in final electronic applications.

How to Synthesize 1,2,7,8-Tetrabromobicyclopenteno[cd,lm]Perylene Efficiently
The practical implementation of this synthesis is designed for operational simplicity, making it highly attractive for both laboratory optimization and industrial scale-up. The general protocol involves charging a reaction vessel with the tetrahydro-perylene precursor, the chosen brominating agent, and the peroxide catalyst in an appropriate aprotic solvent. The mixture is then heated under reflux or stirred at controlled temperatures ranging from 60°C to 90°C overnight, allowing sufficient time for the radical chain reaction to reach completion. Following the reaction, the crude solid is isolated via filtration, and the solvent is removed under reduced pressure. A standard aqueous workup using sodium thiosulfate solution effectively quenches any residual oxidizing species, while subsequent washing with water and brine removes inorganic salts and polar byproducts. The final purification is achieved through recrystallization from ethanol, yielding the target compound as a high-purity yellow-orange solid suitable for immediate use in subsequent coupling reactions. For detailed standardized operating procedures and specific molar ratios optimized for different scales, please refer to the technical guide below.
- Combine 1,2,7,8-tetrahydrodicyclopentano[cd,lm]perylene with a brominating agent (NBS or DBDMH) and a peroxide catalyst in an aprotic solvent.
- Stir the reaction mixture overnight at temperatures between 60°C and 90°C to facilitate radical substitution and dehydrogenation.
- Filter solid impurities, remove solvent, wash with aqueous solutions, and recrystallize from ethanol to obtain the final yellow-orange product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis route translates into tangible strategic benefits regarding cost stability and supply continuity. The ability to achieve high yields in a single step drastically reduces the consumption of raw materials and solvents per kilogram of finished product, directly impacting the variable cost of goods sold. Unlike multi-step syntheses that accumulate yield losses at each stage, this consolidated approach maximizes throughput and minimizes the inventory of work-in-progress materials, thereby freeing up working capital. Furthermore, the use of commodity chemicals such as NBS, DBDMH, and common organic solvents ensures that the supply chain is not vulnerable to the volatility associated with exotic or proprietary reagents. This accessibility allows for diversified sourcing strategies, reducing the risk of supply disruptions caused by single-vendor dependencies or geopolitical constraints on specialized chemical feedstocks.
- Cost Reduction in Manufacturing: The elimination of complex multi-step sequences and the avoidance of expensive transition metal catalysts significantly lower the operational expenditure associated with production. By removing the need for costly purification columns or chromatography typically required to separate isomeric byproducts from traditional methods, the process reduces both labor hours and consumable costs. The high atom economy of the radical bromination ensures that a greater proportion of the starting mass is converted into valuable product rather than waste, optimizing the overall material efficiency. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to a lower carbon footprint and reduced utility expenses for the manufacturing facility.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method against minor variations in reaction parameters ensures consistent batch-to-bquality, which is critical for maintaining long-term supply agreements with electronics manufacturers. The flexibility to use different solvents (CCl4, CHCl3, THF, DMF) and brominating agents provides process engineers with the agility to adapt to regional regulatory changes or temporary shortages of specific chemicals without halting production. This adaptability mitigates the risk of production stoppages and ensures a steady flow of intermediates to downstream customers. Moreover, the simplicity of the isolation and purification steps shortens the overall cycle time from raw material intake to finished goods, enabling faster response times to urgent customer orders and fluctuating market demand.
- Scalability and Environmental Compliance: The one-pot nature of the reaction facilitates straightforward scale-up from kilogram to tonne quantities without the need for complex reactor modifications or specialized equipment. The absence of heavy metal catalysts simplifies the waste stream treatment, as there is no need for expensive heavy metal scavenging or disposal procedures, aligning with increasingly stringent global environmental regulations. The solid byproducts generated, such as succinimide, are non-toxic and can often be recovered or disposed of with minimal environmental impact. This compliance advantage reduces the administrative burden and liability costs associated with hazardous waste management, making the process sustainable for long-term commercial operation in regulated jurisdictions.
Frequently Asked Questions (FAQ)
To address common technical inquiries regarding the implementation and application of this technology, we have compiled the following insights based on the patent specifications. These answers clarify the operational parameters and the specific advantages of the radical bromination pathway compared to legacy methods. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this intermediate into their own device fabrication workflows. The data presented reflects the optimized conditions described in the intellectual property documentation.
Q: What are the primary advantages of this bromination method over traditional halogenation?
A: This method utilizes a one-step radical process with peroxide catalysis, achieving yields over 80% without requiring complex multi-step sequences or harsh Lewis acids often found in conventional electrophilic aromatic substitution.
Q: Which solvents are compatible with this synthesis for large-scale production?
A: The patent specifies several aprotic solvents including carbon tetrachloride, chloroform, THF, and DMF, offering flexibility for process engineers to select based on toxicity profiles and boiling point requirements for solvent recovery.
Q: How does this intermediate contribute to organic semiconductor performance?
A: The introduction of bromine atoms and the formation of double bonds in the five-membered rings extend the conjugated system, enhancing electron-withdrawing properties and carrier mobility essential for OFETs and OLED applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,7,8-Tetrabromobicyclopenteno[cd,lm]Perylene Supplier
As the demand for high-performance organic semiconductors continues to surge, partnering with a manufacturer that possesses deep technical expertise in complex aromatic synthesis is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver consistent quality. Our state-of-the-art facilities are equipped to handle the specific safety and processing requirements of radical bromination reactions, ensuring that every batch meets stringent purity specifications required for electronic applications. With rigorous QC labs employing advanced analytical techniques, we guarantee that our 1,2,7,8-tetrabromobicyclopenteno[cd,lm]perylene is free from detrimental impurities that could compromise device efficiency, providing our clients with a secure foundation for their R&D and manufacturing activities.
We invite forward-thinking organizations to collaborate with us to unlock the full potential of this advanced material platform. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments that demonstrate how our optimized supply chain can enhance your project's timeline and budget. Let us be your strategic partner in navigating the complexities of organic electronic material sourcing.
