Scalable Production of N-phenyl bis(trifluoromethanesulfonyl) imide via Gas-Phase Fluorination
Scalable Production of N-phenyl bis(trifluoromethanesulfonyl) imide via Gas-Phase Fluorination
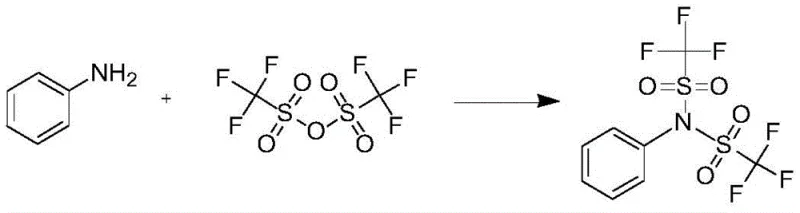
The pharmaceutical and fine chemical industries are constantly seeking more efficient routes for critical intermediates, and patent CN111269152A introduces a transformative method for synthesizing N-phenyl bis(trifluoromethanesulfonyl) imide. This compound serves as a highly efficient trifluoromethanesulfonylation reagent, essential for modifying enols, phenols, and amines in complex drug synthesis, including the production of the anti-prostate cancer agent Abiraterone. Traditional manufacturing relies heavily on expensive anhydrides or hazardous acid chlorides, creating bottlenecks in cost and safety. The disclosed innovation shifts the paradigm by utilizing trifluoromethanesulfonyl fluoride gas as the primary raw material, reacting it with aniline in a controlled, stepwise manner. This approach not only drastically lowers the entry cost for raw materials but also simplifies the purification process, yielding products with purity levels exceeding 99 percent through simple washing rather than complex chromatography.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-phenyl bis(trifluoromethanesulfonyl) imide has been plagued by economic and safety inefficiencies inherent to older chemical pathways. The most common traditional route, often referred to as Scheme 1, utilizes trifluoromethanesulfonic anhydride as the sulfonylating agent. As illustrated in the reaction diagram below, this method requires at least two equivalents of the anhydride, which is an exceptionally costly reagent, thereby inflating the overall production budget significantly. Furthermore, this reaction is highly exothermic in its early stages, necessitating rigorous low-temperature control and slow dropwise addition to prevent thermal runaway. Even under these strict conditions, which often include inert gas protection, the maximum yield rarely exceeds 86 percent unless extremely precise parameters are met, leading to substantial material loss and increased waste generation.
Alternative historical methods, such as Scheme 2, attempt to bypass the anhydride by generating trifluoromethanesulfonyl chloride in situ from trifluoromethanesulfonic acid and thionyl chloride. However, this pathway introduces severe safety hazards and environmental concerns. The use of thionyl chloride generates irritant acid mists that pose significant risks to operators and require specialized scrubbing equipment to manage emissions. Additionally, the reaction progress must be meticulously controlled via slow dripping to avoid dangerous heat accumulation, making the process difficult to scale safely. The resulting intermediate often requires concentration steps that further expose personnel to hazardous vapors, rendering this method less desirable for modern, green chemistry-compliant manufacturing facilities aiming for high throughput.
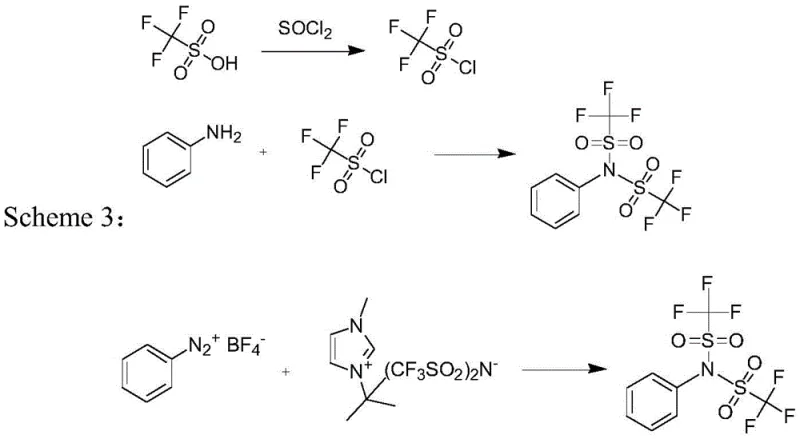
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent leverages trifluoromethanesulfonyl fluoride gas, a raw material that is not only more economically viable but also allows for a cleaner reaction profile. By reacting this gas directly with aniline in the presence of an organic base within a strong polar solvent, the process efficiently generates the mono-substituted intermediate, N-phenyl trifluoromethanesulfonamide. This step avoids the violent exotherms associated with anhydride additions and eliminates the need for corrosive thionyl chloride. The subsequent second step involves a catalytic trifluoromethanesulfonylation in a weak polar solvent, which drives the reaction to completion with high conversion rates. Crucially, the gaseous nature of the reagent allows for a closed-loop system where excess gas can be recovered and recycled, virtually eliminating atmospheric emissions and reducing raw material consumption per kilogram of product.
Mechanistic Insights into Stepwise Gas-Liquid Trifluoromethanesulfonylation
The core of this technological breakthrough lies in the mechanistic efficiency of the nucleophilic substitution reactions facilitated by the specific choice of solvents and catalysts. In the first stage, the organic base, such as triethylenediamine or DBU, acts as a potent acid-binding agent, scavenging the hydrogen fluoride generated during the attack of the aniline nitrogen on the sulfur atom of the trifluoromethanesulfonyl fluoride. This neutralization drives the equilibrium forward, ensuring high conversion to the mono-amide intermediate without the formation of significant byproducts. The use of strong polar solvents like acetonitrile or DMF in this initial phase enhances the solubility of the ionic intermediates and stabilizes the transition state, allowing the reaction to proceed smoothly at moderate temperatures ranging from 0 to 80 degrees Celsius without the need for cryogenic cooling.
The second stage of the mechanism involves the activation of the mono-amide for the second sulfonylation event, which is kinetically more challenging due to the reduced nucleophilicity of the nitrogen atom after the first substitution. Here, the addition of a nucleophilic catalyst, specifically 4-dimethylaminopyridine (DMAP) or 4-pyrrolylpyridine, plays a pivotal role in lowering the activation energy. The catalyst likely forms a reactive acyl pyridinium-like intermediate with the sulfonyl fluoride, making the sulfur center more electrophilic and susceptible to attack by the less reactive amide nitrogen. This catalytic cycle ensures that the reaction proceeds to the bis-substituted product with high selectivity, minimizing the risk of over-reaction or decomposition, which is a common issue in non-catalyzed high-temperature variations of this chemistry.
How to Synthesize N-phenyl bis(trifluoromethanesulfonyl) imide Efficiently
The operational protocol for this synthesis is designed for industrial robustness, prioritizing safety and ease of purification while maintaining high chemical fidelity. The process begins by charging a stainless steel reactor with aniline and an organic base dissolved in a polar solvent, followed by the introduction of trifluoromethanesulfonyl fluoride gas under slight pressure. After the first substitution is complete, the solvent is distilled off, and the residue is treated with a weak polar solvent and a catalytic amount of DMAP before introducing a second charge of the fluoride gas. The final isolation is remarkably simple, involving the removal of solvents and a wash with an alcohol such as ethanol or methanol, which precipitates the product as high-purity white crystals. For the detailed standardized operating procedures and specific parameter settings, please refer to the guide below.
- React aniline with organic base in a polar solvent under trifluoromethanesulfonyl fluoride gas to form N-phenyl trifluoromethanesulfonamide.
- Remove polar solvent, add weak polar solvent and catalyst, then react with excess trifluoromethanesulfonyl fluoride gas.
- Recover solvent and wash the crude product with an alcohol solvent to obtain high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this gas-phase fluorination methodology represents a strategic opportunity to optimize the cost structure of key pharmaceutical intermediates. By replacing expensive liquid reagents like trifluoromethanesulfonic anhydride with gaseous trifluoromethanesulfonyl fluoride, manufacturers can achieve substantial cost savings on raw material acquisition. The ability to recycle unreacted gas further amplifies these economic benefits, effectively reducing the net consumption of the fluorinating agent per batch. Moreover, the elimination of hazardous reagents like thionyl chloride removes the need for complex acid gas scrubbing systems and reduces the regulatory burden associated with handling corrosive liquids, leading to lower operational expenditures and insurance costs.
- Cost Reduction in Manufacturing: The replacement of high-cost anhydrides with more affordable fluoride gas, combined with the recycling of excess reagent, leads to a drastic reduction in variable production costs. The simplified purification process, which relies on crystallization and washing rather than column chromatography or extensive distillation, further decreases energy consumption and labor hours required per unit of output. This lean manufacturing approach ensures that the final product remains competitive in price-sensitive markets without compromising on quality standards.
- Enhanced Supply Chain Reliability: Utilizing a gaseous reagent that can be stored in bulk cylinders and fed on-demand improves inventory management and reduces the risks associated with the degradation of liquid reagents over time. The mild reaction conditions, which do not strictly require inert gas protection or extreme low temperatures, make the process more resilient to utility fluctuations and equipment limitations. This robustness ensures consistent production schedules and reliable delivery timelines for downstream customers relying on this critical intermediate for their own API synthesis.
- Scalability and Environmental Compliance: The closed-loop nature of the gas handling system aligns perfectly with modern environmental, health, and safety (EHS) standards by preventing the release of volatile fluorinated compounds into the atmosphere. The absence of heavy metal catalysts and corrosive acid byproducts simplifies waste treatment protocols, allowing for easier compliance with increasingly stringent global environmental regulations. This green chemistry profile facilitates smoother regulatory approvals and supports the sustainability goals of multinational pharmaceutical partners.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of adopting this new synthesis route, we have compiled answers to common inquiries regarding the process specifics and quality outcomes. These insights are derived directly from the experimental data and technical disclosures found within the patent literature, ensuring accuracy and relevance for industrial application. Understanding these nuances is critical for R&D directors planning pilot runs and for quality assurance teams establishing specification limits for incoming materials.
Q: What are the primary advantages of using trifluoromethanesulfonyl fluoride over trifluoromethanesulfonic anhydride?
A: Trifluoromethanesulfonyl fluoride is significantly cheaper than trifluoromethanesulfonic anhydride and allows for a closed-loop recycling system, reducing both raw material costs and environmental waste.
Q: Does this new method require inert gas protection?
A: Unlike conventional methods requiring strict nitrogen protection and low temperatures, this novel approach operates under mild conditions without the absolute necessity for inert gas shielding, simplifying operational complexity.
Q: What purity levels can be achieved with this synthesis route?
A: The process yields a crude product of high quality that, after simple alcohol washing, achieves a purity exceeding 99%, suitable for sensitive pharmaceutical applications like Abiraterone synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-phenyl bis(trifluoromethanesulfonyl) imide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to full-scale manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of N-phenyl bis(trifluoromethanesulfonyl) imide meets the exacting standards required for pharmaceutical synthesis, providing our partners with the confidence they need to move forward with their projects.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the potential economic impact of switching to this method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your production needs, ensuring a partnership built on transparency, technical excellence, and mutual growth.
