Scalable Synthesis of Latamoxef Sodium 7-Position Side Chain for Commercial API Production
Scalable Synthesis of Latamoxef Sodium 7-Position Side Chain for Commercial API Production
The pharmaceutical industry continuously seeks robust and scalable pathways for critical antibiotic intermediates, particularly for semi-synthetic cephalosporins like Latamoxef sodium. Patent CN102718656A introduces a groundbreaking preparation method for the 7-position side chain of Latamoxef sodium, specifically 2-(4-hydroxyphenyl)-propanedioic acid 1-[(4-methoxyphenyl)methyl] ester. This innovation addresses long-standing challenges in beta-lactam antibiotic manufacturing by replacing complex, hazardous multi-step sequences with a concise three-step protocol. The strategic value of this technology lies in its ability to deliver high-purity intermediates while drastically simplifying the supply chain logistics for API producers. By utilizing readily available starting materials such as p-hydroxyphenylacetic acid and p-methoxybenzyl chloride, the process mitigates the reliance on exotic reagents that often bottleneck production schedules. 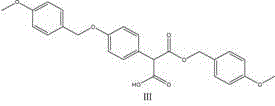
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this critical side chain has been plagued by operational inefficiencies and safety hazards that deter large-scale adoption. Traditional routes often commence with phenylacetonitrile, requiring more than ten synthetic steps and involving nearly thirty different reagents, which exponentially increases the cost of goods sold (COGS) and waste generation. A significant pain point in these legacy methods is the reliance on n-butyllithium to generate lithium diisopropylamide (LDA), a reagent that demands strictly anhydrous conditions and cryogenic temperatures, posing severe safety risks and requiring specialized, expensive reactor infrastructure. Furthermore, alternative approaches utilizing methyl p-hydroxyphenylacetate often employ N,N-dimethyl sulfoxide (DMSO) as a solvent, which has a high boiling point and is notoriously difficult to remove completely, leading to solvent retention issues in the final API and complicating recycling protocols. These factors collectively render conventional methods economically unviable for modern, cost-sensitive generic drug manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in CN102718656A streamlines the entire production workflow into just three distinct chemical transformations, fundamentally altering the economic landscape for this intermediate. The process initiates with a straightforward alkylation or esterification step using inexpensive carbonate bases like potassium carbonate or sodium carbonate in low-boiling aprotic solvents, which facilitates easy solvent recovery and energy-efficient distillation. 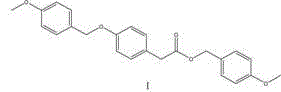
This initial step yields Intermediate I with high efficiency, setting the stage for a subsequent Claisen-type condensation that constructs the malonic acid backbone without the need for hazardous organolithium reagents. The final oxidation step utilizes benign oxidants such as hydrogen peroxide or sodium hypochlorite, eliminating the environmental burden associated with heavy metal oxidants. This holistic redesign not only reduces the number of unit operations but also ensures that the reaction conditions remain within a mild temperature range of 60°C to 100°C, making the process inherently safer and compatible with standard glass-lined or stainless-steel reactors found in most multipurpose chemical plants.
Mechanistic Insights into the Three-Step Cascade Synthesis
The core of this technological breakthrough lies in the precise orchestration of functional group transformations that build complexity while maintaining operational simplicity. The first stage involves the activation of p-hydroxyphenylacetic acid, where the carboxylic acid moiety reacts with p-methoxybenzyl chloride in the presence of a carbonate base. This reaction effectively installs the protecting group or ester linkage required for downstream processing, leveraging the nucleophilicity of the carboxylate anion generated in situ. The choice of aprotic solvents such as acetonitrile or DMF is critical here, as they solubilize the ionic intermediates while remaining inert to the reaction conditions, ensuring high conversion rates without side reactions. Following isolation and recrystallization, Intermediate I is obtained with exceptional purity, providing a clean substrate for the subsequent carbon-carbon bond-forming event.
The second mechanistic phase employs a classic condensation strategy where Intermediate I is treated with sodium alkoxide and a formate ester, such as ethyl formate or methyl formate, in a toluene medium. This step introduces the second carbonyl group necessary for the propanedioic acid structure, effectively converting the acetate derivative into a malonate derivative through a nucleophilic acyl substitution mechanism. 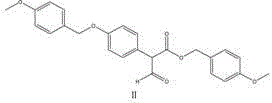
The use of toluene allows for the azeotropic removal of water or alcohol byproducts, driving the equilibrium towards the desired Intermediate II. Finally, the third step involves a controlled oxidation where Intermediate II is subjected to hydrogen peroxide or sodium hypochlorite in methanol. This oxidation likely targets specific susceptible positions to finalize the oxidation state of the side chain, followed by a rigorous workup involving acidification and crystallization. The entire sequence is designed to minimize impurity carryover, with each crystallization step acting as a purge for potential byproducts, ensuring the final material meets the stringent specifications required for antibiotic synthesis.
How to Synthesize Latamoxef Sodium 7-Position Side Chain Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and temperature control to maximize yield and purity. The process begins by dissolving p-hydroxyphenylacetic acid and a carbonate base in a non-protonic solvent, followed by the addition of p-methoxybenzyl chloride at elevated temperatures to drive the formation of Intermediate I. Once isolated, Intermediate I is reacted with sodium alkoxide and a formate ester in toluene under controlled cooling to generate Intermediate II, which is then extracted and purified via acidification. The final transformation involves dissolving Intermediate II in methanol and slowly adding an oxidant under reflux conditions, followed by concentration and crystallization to yield the target side chain. For detailed standard operating procedures and specific parameter optimization, please refer to the technical guide below.
- React p-hydroxyphenylacetic acid with p-methoxybenzyl chloride in an aprotic solvent using carbonate bases to form Intermediate I.
- Perform Claisen condensation on Intermediate I using sodium alkoxide and formate esters in toluene to generate Intermediate II.
- Oxidize Intermediate II in methanol using hydrogen peroxide or sodium hypochlorite, followed by crystallization to obtain the final side chain.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers transformative benefits that extend far beyond simple chemical yield improvements. By eliminating the need for cryogenic reagents like n-butyllithium and high-boiling solvents like DMSO, the process significantly reduces the dependency on specialized raw materials that are often subject to volatile market pricing and supply disruptions. The shift to commodity chemicals such as carbonates, toluene, and hydrogen peroxide creates a more resilient supply chain, as these materials are globally sourced and available in bulk quantities from multiple vendors. Furthermore, the reduction in reaction steps from over ten to merely three drastically cuts down on labor costs, reactor occupancy time, and utility consumption, leading to substantial overall cost reductions in pharmaceutical intermediate manufacturing.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the replacement of expensive, hazardous reagents with inexpensive, safe alternatives. The elimination of n-butyllithium removes the need for costly inert atmosphere handling and cryogenic cooling systems, which are capital-intensive to install and maintain. Additionally, the use of low-boiling aprotic solvents allows for energy-efficient distillation and solvent recovery, further lowering the operational expenditure (OPEX) associated with solvent purchase and waste disposal. The simplified three-step sequence also minimizes material loss during transfer and purification, enhancing the overall mass balance and reducing the cost per kilogram of the final active ingredient.
- Enhanced Supply Chain Reliability: From a logistics perspective, the reliance on stable, shelf-stable reagents like potassium carbonate and p-methoxybenzyl chloride ensures consistent production scheduling without the risk of reagent degradation or delivery delays common with sensitive organometallics. The robustness of the reaction conditions, which tolerate a broader range of temperatures and moisture levels compared to traditional methods, reduces the likelihood of batch failures and production downtime. This reliability is crucial for maintaining continuous supply to API manufacturers, preventing stockouts that could disrupt the production of life-saving antibiotics and ensuring compliance with just-in-time delivery models.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing reaction conditions that are easily replicated in large-scale industrial reactors without the engineering challenges posed by exothermic organolithium reactions. The use of benign oxidants like hydrogen peroxide, which decomposes into water and oxygen, aligns with green chemistry principles and simplifies wastewater treatment protocols. This environmental compatibility reduces the regulatory burden and costs associated with hazardous waste disposal, making the facility more sustainable and compliant with increasingly strict environmental regulations governing pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the key advantages of this synthesis route over conventional methods?
A: This method eliminates the need for hazardous cryogenic reagents like n-butyllithium and avoids high-boiling solvents like DMSO, significantly reducing energy consumption and safety risks while simplifying purification.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the reaction conditions are mild (60-100°C) and utilize common industrial reagents like carbonates and hydrogen peroxide, making it highly adaptable for multi-ton production without specialized equipment.
Q: What purity levels can be achieved with this patented method?
A: The process includes efficient crystallization steps that consistently yield intermediates and final products with HPLC purity exceeding 99%, meeting stringent pharmaceutical standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Latamoxef Sodium 7-Position Side Chain Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of antibiotics. Our team of expert chemists has extensively evaluated the pathway described in CN102718656A and possesses the technical capability to execute this synthesis with precision and consistency. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering reliability. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Latamoxef sodium 7-position side chain meets the highest industry standards for pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can enhance your production efficiency and product quality.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
