Advanced Green Synthesis of DL-Pantoic Acid Lactone for Commercial Vitamin B5 Production
Advanced Green Synthesis of DL-Pantoic Acid Lactone for Commercial Vitamin B5 Production
The global demand for Vitamin B5 derivatives, specifically D-calcium pantothenate and D-panthenol, continues to drive innovation in the upstream synthesis of key intermediates. Patent CN113024491A introduces a groundbreaking preparation method for DL-alpha-hydroxy-beta, beta-dimethyl-gamma-butyrolactone, also known as DL-pantoic acid lactone, which serves as the critical precursor in this value chain. This technology addresses long-standing bottlenecks in the industry, such as low yield stability and high environmental burden, by optimizing the classic isobutyraldehyde-formaldehyde-hydrocyanic acid route. The disclosed method achieves high chemical purity and yield through precise control of reaction parameters, making it a highly attractive solution for manufacturers seeking to upgrade their production capabilities.
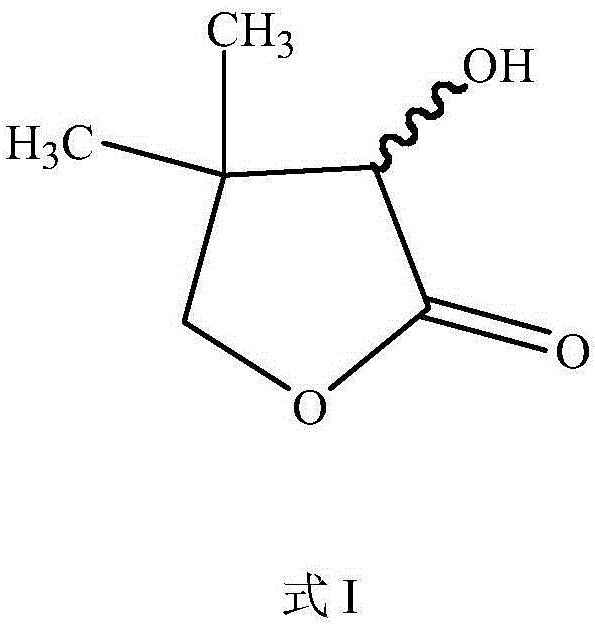
DL-alpha-hydroxy-beta, beta-dimethyl-gamma-butyrolactone is not merely a commodity chemical but a vital building block for coenzyme A synthesis, playing an indispensable role in fatty acid metabolism and antibody synthesis within biological systems. As the market for pharmaceuticals, food additives, and feed supplements expands, the pressure on suppliers to provide consistent, high-quality intermediates intensifies. The traditional technologies employed in China and globally have often suffered from high costs and variable quality, creating a supply chain vulnerability for downstream vitamin producers. This new patent offers a robust alternative that aligns with modern green chemistry principles, ensuring that the production of this essential intermediate is both economically viable and environmentally sustainable for the long term.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of DL-pantoic acid lactone has relied heavily on two primary routes: the isobutyraldehyde-formaldehyde-sodium cyanide method and the isobutyraldehyde-formaldehyde-hydrocyanic acid method. While the sodium cyanide route is mature, it is plagued by significant environmental drawbacks, primarily the generation of massive quantities of cyanide-containing waste salts and wastewater. This creates a severe post-treatment burden, requiring complex neutralization and disposal procedures that escalate operational costs and regulatory risks. Furthermore, the reaction efficiency in these older processes is often suboptimal, leading to inconsistent yields that fluctuate based on minor variations in raw material quality or operator handling.
Even the hydrocyanic acid route, while theoretically cleaner, has faced challenges regarding safety and specificity. Hydrocyanic acid is highly toxic and difficult to transport and store, necessitating rigorous safety measures that many older facilities struggle to maintain effectively. Without precise control over reaction conditions, particularly temperature and pH during the cyanohydrin formation step, hydrocyanic acid is prone to polymerization. This side reaction not only consumes valuable raw materials but also introduces impurities that complicate downstream purification, ultimately resulting in a final product that fails to meet the stringent purity specifications required for pharmaceutical-grade vitamin synthesis.
The Novel Approach
The methodology outlined in Patent CN113024491A represents a significant evolution of the hydrocyanic acid route, introducing critical optimizations that mitigate the risks associated with conventional practices. The core innovation lies in the refined control of the condensation and cyanohydrin reaction steps. By utilizing specific organic base catalysts such as diethylamine or triethylamine and strictly regulating the dropwise addition of isobutyraldehyde at temperatures between 10-30°C, the process ensures a high conversion rate to hydroxyl pivalaldehyde. This sets a solid foundation for the subsequent steps, minimizing the formation of early-stage byproducts.
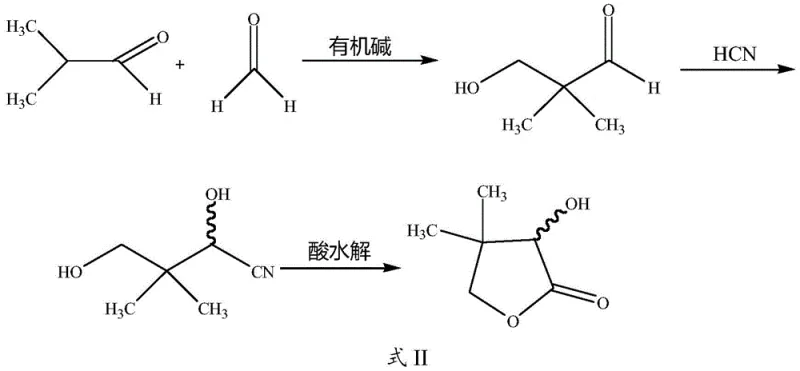
Furthermore, the novel approach implements a sophisticated protocol for the cyanohydrin reaction, where hydrocyanic acid and the aldehyde intermediate are added simultaneously via peristaltic pump. This allows for real-time adjustment of the pH value to maintain a narrow window of 8-9, while keeping the reaction temperature strictly between 0-20°C. Such precision prevents the thermal polymerization of HCN that plagues older methods. Following this, the acidic hydrolysis and lactonization are conducted under controlled conditions that facilitate the recovery of ammonium salt as a valuable byproduct. This holistic optimization results in a process that is not only simpler and more feasible for industrial scaling but also delivers superior yield and purity compared to legacy technologies.
Mechanistic Insights into Organic Base-Catalyzed Condensation and Cyclization
The success of this synthesis route hinges on the mechanistic efficiency of the initial aldol-like condensation between formaldehyde and isobutyraldehyde. In the presence of an organic base catalyst, the alpha-hydrogen of the isobutyraldehyde is activated, facilitating a nucleophilic attack on the carbonyl carbon of the formaldehyde. The choice of catalyst, such as ethylenediamine or diethylamine, is critical as it provides the necessary basicity to drive the reaction forward without promoting excessive side reactions like Cannizzaro disproportionation. The patent data indicates that maintaining the mass ratio of the catalyst to isobutyraldehyde between 6-8:100 creates an optimal kinetic environment, ensuring that the hydroxymethylation proceeds rapidly to form hydroxyl pivalaldehyde with high selectivity.
Following the condensation, the transition to the lactone structure involves a delicate balance of nucleophilic addition and intramolecular cyclization. During the cyanohydrin step, the cyanide ion attacks the carbonyl group of the hydroxyl pivalaldehyde. The subsequent acid hydrolysis converts the nitrile group into a carboxylic acid, which then spontaneously undergoes lactonization due to the proximity of the gamma-hydroxyl group. The patent emphasizes the importance of the acid concentration (30-60%) and temperature (60-100°C) during this phase. These parameters are tuned to ensure complete hydrolysis while promoting the thermodynamic favorability of the five-membered lactone ring closure. The final purification via high vacuum rectification at 20-100Pa effectively separates the target lactone from any remaining oligomers or unreacted precursors, guaranteeing a final purity exceeding 98%.
How to Synthesize DL-alpha-hydroxy-beta, beta-dimethyl-gamma-butyrolactone Efficiently
The synthesis of this high-value intermediate requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and safety. The process is divided into three distinct operational phases: the catalytic condensation of aldehydes, the controlled cyanohydrin formation followed by hydrolysis, and the final vacuum purification. Each stage demands precise monitoring of temperature, pH, and addition rates to maximize yield and minimize waste. For R&D teams looking to implement this technology, understanding the interplay between the organic base catalyst and the hydrocyanic acid addition rate is paramount. Detailed standardized operating procedures for these steps are essential for successful technology transfer.
- Mix formaldehyde with an organic base catalyst and dropwise add isobutyraldehyde at controlled temperatures (10-30°C) to generate hydroxyl pivalaldehyde.
- Perform a cyanohydrin reaction by adding hydrocyanic acid under strict pH (8-9) and temperature control, followed by acidic hydrolysis and lactonization to form the crude lactone.
- Purify the crude product via high vacuum rectification at 20-100Pa system pressure to obtain high-purity DL-alpha-hydroxy-beta, beta-dimethyl-gamma-butyrolactone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers compelling strategic advantages beyond mere technical performance. The shift towards a greener, more efficient process directly translates into risk mitigation and cost optimization across the entire value chain. By eliminating the heavy reliance on sodium cyanide and its associated waste streams, manufacturers can significantly reduce their environmental compliance costs and the logistical complexities of hazardous waste disposal. This creates a more resilient supply base that is less susceptible to regulatory shutdowns or sudden increases in waste treatment fees, ensuring a steadier flow of materials to downstream vitamin producers.
- Cost Reduction in Manufacturing: The optimized process achieves substantial cost savings through improved atom economy and the valorization of byproducts. Unlike traditional methods where waste salts represent a pure cost center, this method recovers ammonium salt, which can be sold or reused, thereby offsetting a portion of the raw material expenses. Additionally, the higher reaction yield means that less raw material is required to produce the same amount of finished product, drastically lowering the variable cost per kilogram. The elimination of expensive transition metal catalysts or complex purification resins further simplifies the cost structure, making the final intermediate more price-competitive in the global market.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the volatility of raw material availability and the complexity of synthesis. This method utilizes readily available bulk chemicals like formaldehyde and isobutyraldehyde, reducing dependency on niche or imported reagents. The robustness of the reaction conditions, particularly the tolerance achieved through precise pH control, ensures that production batches are consistent and reliable. This predictability allows supply chain planners to maintain leaner inventories with confidence, knowing that the lead time for high-purity pharmaceutical intermediates can be consistently met without unexpected delays caused by failed batches or quality deviations.
- Scalability and Environmental Compliance: As demand for Vitamin B5 grows, the ability to scale production without proportionally increasing environmental impact is crucial. This process is inherently designed for large-scale industrial production, with parameters that are easily manageable in standard stainless steel reactors. The reduction in hazardous waste generation aligns with increasingly strict global environmental regulations, future-proofing the manufacturing facility against tighter emission standards. This scalability ensures that suppliers can rapidly ramp up capacity to meet surges in market demand, providing a secure and sustainable source of critical intermediates for the pharmaceutical and nutrition industries.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific improvements and data points highlighted in the patent documentation, focusing on yield optimization, safety protocols, and economic benefits. Understanding these aspects is crucial for stakeholders evaluating the feasibility of adopting this new method for commercial production.
Q: How does this new preparation method improve upon traditional sodium cyanide routes?
A: Unlike the traditional isobutyraldehyde-formaldehyde-sodium cyanide route which generates large amounts of cyanide-containing waste salt and wastewater, this optimized method utilizes hydrocyanic acid with precise parameter control. This shift significantly reduces environmental protection pressure and post-treatment difficulties while improving overall atom economy and yield stability.
Q: What specific process controls prevent the polymerization of hydrocyanic acid during synthesis?
A: The patent specifies strict temperature management during the cyanohydrin reaction step. By controlling the dropping temperature between 0-20°C and maintaining the reaction temperature between -10°C to 10°C after addition, the process effectively suppresses the polymerization of hydrocyanic acid, which is a common cause of yield reduction in conventional methods.
Q: Does this process generate any valuable byproducts that can offset production costs?
A: Yes, the process includes a step where the water phase is concentrated after removing the crude product to recover ammonium salt. This byproduct possesses certain economic value, contributing to the overall cost-effectiveness and industrial viability of the manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable DL-alpha-hydroxy-beta, beta-dimethyl-gamma-butyrolactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of essential vitamins and pharmaceuticals. Our team of expert chemists has extensively analyzed advanced synthesis routes like the one described in Patent CN113024491A to ensure our production capabilities remain at the forefront of the industry. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you require pilot quantities or bulk supply, our output meets stringent purity specifications. Our rigorous QC labs employ state-of-the-art analytical techniques to verify every batch, guaranteeing that the DL-alpha-hydroxy-beta, beta-dimethyl-gamma-butyrolactone we deliver is free from harmful impurities and ready for immediate downstream processing.
We invite potential partners to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency. By collaborating with NINGBO INNO PHARMCHEM, you gain access to a reliable partner committed to delivering excellence in fine chemical intermediates, ensuring your production lines run smoothly and cost-effectively.
