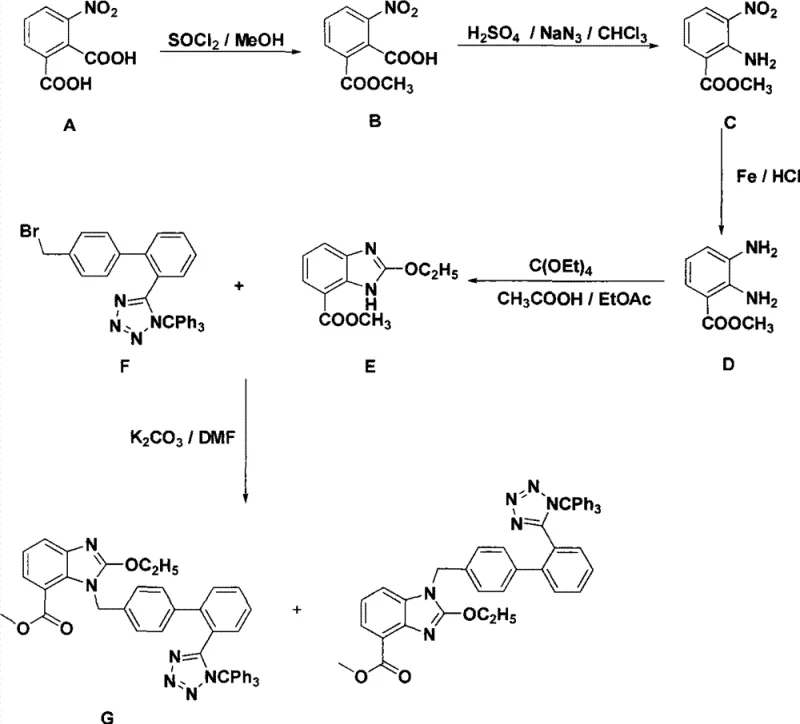
Advanced Modular Synthesis of Trityl Candesartan Intermediate for Commercial Scale-up
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for Angiotensin II receptor blockers, particularly for high-volume drugs like Candesartan Cilexetil. Patent CN101323610B discloses a novel technology for synthesizing a critical trityl-protected intermediate of Candesartan, addressing significant bottlenecks in existing manufacturing processes. This invention fundamentally restructures the synthesis into two distinct modules: the benzimidazole mother nucleus and the biphenyl tetrazole component. This strategic decoupling allows for the optimization of each fragment independently before the final coupling step, offering a streamlined pathway that bypasses the cumbersome protection and deprotection sequences typical of earlier methodologies. By starting from readily available 3-nitrophthalic acid, the process establishes a cost-effective foundation for large-scale production.
The implementation of this technology represents a significant leap forward in process chemistry, specifically designed to enhance the reliability of the pharmaceutical intermediates supplier network. The route demonstrates exceptional versatility, enabling the shared use of the biphenyl tetrazole module across multiple Sartan class drugs, including Losartan and Irbesartan. This modularity not only simplifies inventory management but also drastically reduces the capital investment required for setting up dedicated production lines for each specific drug substance. For R&D teams focused on process optimization, the detailed examples provided in the patent offer a comprehensive toolkit of reduction methods, ranging from tin chloride to catalytic hydrogenation, allowing for fine-tuning based on specific facility capabilities and environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Candesartan intermediates has been plagued by operational complexity and reliance on scarce starting materials. Traditional routes often commence with 2,3-diaminobenzoic acid methyl ester or require the synthesis of 3-nitro-2-tert-butoxyaminobenzoic acid methyl ester as a key precursor. The latter compound is notoriously difficult to procure commercially, forcing manufacturers to dedicate significant resources to its preparation, which inherently involves hazardous azidation and careful temperature control. Furthermore, early introduction of the highly reactive tetrazole ring in conventional pathways necessitates rigorous nitrogen protection strategies, typically involving tert-butoxycarbonyl (Boc) groups. These additional protection and deprotection steps not only extend the overall reaction time but also introduce multiple opportunities for impurity generation, complicating downstream purification and reducing overall yield efficiency.
The Novel Approach
In stark contrast, the novel approach detailed in CN101323610B elegantly circumvents these hurdles by delaying the introduction of complex functionalities until the final stages. The process initiates with a highly efficient selective mono-esterification of 3-nitrophthalic acid, achieving yields as high as 99% under optimized conditions using thionyl chloride and methanol. This is followed by a Schmidt reaction to convert the remaining carboxylic acid directly into an amine, bypassing the need for pre-functionalized amino-acid derivatives. By deferring the construction of the tetrazole ring and utilizing a modular coupling strategy, the new method eliminates the need for auxiliary protecting groups on the nitrogen atoms during the early synthetic stages. This results in a drastically simplified operational sequence that is far more amenable to commercial scale-up of complex pharmaceutical intermediates, reducing both the physical footprint of the reaction and the chemical waste generated per kilogram of product.
Mechanistic Insights into Selective Esterification and Benzimidazole Cyclization
The cornerstone of this synthetic strategy lies in the precise control of chemoselectivity during the initial functionalization of the phthalic acid derivative. The reaction of 3-nitrophthalic acid with thionyl chloride in methanol exploits the steric and electronic differences between the two carboxylic acid groups adjacent to the nitro substituent. Under controlled low-temperature conditions followed by reflux, the system favors the formation of the mono-methyl ester while leaving the second carboxylic acid intact for subsequent transformation. This selectivity is critical, as di-ester formation would block the pathway to the desired benzimidazole core. Following esterification, the conversion of the free carboxylic acid to an amine via the Schmidt reaction mechanism involves the generation of an acyl azide intermediate in situ, which undergoes rearrangement to an isocyanate and subsequent hydrolysis to the amine. This one-pot transformation is a powerful tool for introducing nitrogen functionality without requiring harsh reducing agents at this early stage.
Subsequent steps focus on the construction of the benzimidazole ring, a process that demands rigorous impurity control to ensure the quality of the final API. The reduction of the nitro group to an amine can be achieved through various mechanistic pathways, including metal-acid reductions using tin(II) chloride, iron powder, or zinc powder, as well as catalytic hydrogenation with Raney nickel. Each method offers distinct advantages regarding workup procedures and metal residue profiles. Once the diamine intermediate is formed, cyclization with tetraethyl orthoformate proceeds via nucleophilic attack of the amines on the orthoester carbon, followed by elimination of ethanol to close the imidazole ring. The choice of reduction method significantly influences the purity of the diamine precursor, thereby affecting the efficiency of the cyclization step. By providing multiple validated reduction protocols, the patent empowers manufacturers to select the method that best aligns with their specific high-purity pharmaceutical intermediates quality standards and waste treatment capabilities.
How to Synthesize Trityl Candesartan Intermediate Efficiently
The synthesis of the target trityl candesartan intermediate is achieved through a convergent strategy that maximizes yield and minimizes processing time. The protocol begins with the preparation of the benzimidazole core, followed by a nucleophilic substitution reaction with a bromotrityl tetrazole derivative. This final coupling step is facilitated by a base such as potassium carbonate in a polar aprotic solvent like DMF, ensuring complete conversion of the benzimidazole nitrogen. The detailed standardized synthetic steps, including specific molar ratios, temperature profiles, and workup procedures for each stage from esterification to final purification, are outlined below to guide process engineers in replicating this high-efficiency route.
- Perform selective mono-esterification of 3-nitrophthalic acid using thionyl chloride and methanol to obtain 2-methyl formate-6-nitro-benzoic acid.
- Convert the remaining carboxylic acid group to an amine via Schmidt reaction conditions using sodium azide and sulfuric acid in chloroform.
- Reduce the nitro group to an amine using metal reduction (Fe, Zn, or SnCl2) or catalytic hydrogenation, followed by cyclization with tetraethyl orthoformate.
- Couple the resulting benzimidazole intermediate with a bromotrityl tetrazole derivative using potassium carbonate in DMF to yield the final trityl protected intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers profound strategic benefits beyond mere technical feasibility. The primary advantage stems from the modular architecture of the synthesis, which decouples the production of the benzimidazole nucleus from the biphenyl tetrazole moiety. This separation allows for the independent sourcing or manufacturing of these two key building blocks, significantly mitigating the risk of supply chain disruptions. If a bottleneck occurs in the supply of one module, production of the other can continue uninterrupted, maintaining inventory buffers and ensuring continuity of supply for the final API. Furthermore, the ability to utilize the same biphenyl tetrazole intermediate for multiple Sartan drugs creates economies of scale, allowing manufacturers to purchase raw materials in larger volumes and negotiate better pricing terms with upstream suppliers.
- Cost Reduction in Manufacturing: The elimination of complex protection and deprotection steps, particularly those involving tert-butoxycarbonyl groups, translates directly into substantial cost savings. By removing these auxiliary operations, manufacturers reduce the consumption of expensive protecting group reagents and the solvents required for their removal. Additionally, the high yield observed in the initial esterification step (up to 99%) ensures that raw material utilization is maximized, minimizing waste disposal costs. The use of commodity chemicals like iron powder or zinc for reduction, rather than precious metal catalysts in every step, further lowers the direct material cost profile, making the process economically viable for generic drug production where margin pressure is intense.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as 3-nitrophthalic acid and methanol ensures a stable supply base that is not subject to the volatility of specialized chemical markets. Unlike routes dependent on custom-synthesized intermediates like 3-nitro-2-tert-butoxyaminobenzoic acid methyl ester, this method utilizes bulk chemicals that can be sourced from multiple global vendors. This diversification of the supply base reduces the lead time for high-purity pharmaceutical intermediates and protects against single-source failures. The robustness of the reaction conditions, which tolerate a range of temperatures and reagent grades, also contributes to consistent batch-to-batch reliability, a critical factor for maintaining regulatory compliance and customer trust.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the process offers significant advantages for large-scale operations. The avoidance of hazardous protecting group chemistry reduces the generation of toxic byproducts, simplifying effluent treatment and lowering the environmental compliance burden. While the use of sodium azide requires careful safety management, the overall reduction in the number of synthetic steps decreases the total energy consumption and solvent usage per kilogram of product. The flexibility to choose between different reduction methods (e.g., iron powder vs. catalytic hydrogenation) allows facilities to optimize their waste streams based on local regulatory frameworks, ensuring that the cost reduction in pharmaceutical intermediates manufacturing does not come at the expense of environmental stewardship.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, scalability potential, and quality control measures. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: What are the key advantages of this modular synthesis route compared to traditional methods?
A: This route avoids the use of difficult-to-obtain starting materials like 3-nitro-2-tert-butoxyaminobenzoic acid methyl ester and eliminates complex protection and deprotection steps associated with tert-butoxycarbonyl groups, significantly simplifying the operational workflow.
Q: How does this process impact impurity profiles and product purity?
A: By utilizing selective mono-esterification and robust reduction methods (such as iron or zinc powder reduction), the process minimizes side reactions. The final purification via recrystallization from ethyl acetate and ether ensures high purity specifications suitable for API manufacturing.
Q: Is this synthetic route scalable for industrial production?
A: Yes, the route employs common industrial reagents like thionyl chloride, sodium azide, and iron powder, and avoids exotic catalysts. The modular design allows for the parallel production of the biphenyl tetrazole module, facilitating large-scale manufacturing from 100 kgs to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trityl Candesartan Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the competitive landscape of antihypertensive drug manufacturing. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and robust. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the absence of genotoxic impurities and residual solvents. Our capability to implement the modular synthesis described in CN101323610B allows us to offer a reliable supply of high-quality intermediates that support your long-term production goals.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume requirements and cost targets. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this streamlined methodology. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can drive value for your supply chain.
