Optimized Synthesis of Metoprolol Related Substance E for Commercial API Production
The pharmaceutical industry's relentless pursuit of cardiovascular therapeutic efficacy demands rigorous control over impurity profiles, particularly for beta-blockers like Metoprolol. Patent CN102964259A introduces a groundbreaking preparation method for Metoprolol Related Substance E, a critical reference standard and impurity marker. This novel synthetic route addresses the longstanding challenges of cost and availability associated with traditional methods. By leveraging a six-step sequence starting from the commercially abundant 2-Hydroxyphenyl Acetic Acid, this technology offers a robust pathway for producing high-purity intermediates. For R&D directors and procurement specialists, understanding this methodology is essential for securing reliable supply chains and ensuring the quality of final API batches. The following analysis details the technical merits and commercial viability of this process.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Metoprolol Related Substance E has been hindered by significant economic and operational bottlenecks. Prior art, such as the method reported in the Hebei Normal University Journal, relies on 2-(2-methoxyethyl) phenol as the starting raw material. This specific precursor is not only difficult to source on a global scale but also commands a prohibitively high market price, creating a fragile supply chain for manufacturers. Furthermore, the reaction yield for the coupling of this phenol with epichlorohydrin is reported to be merely 40%, which is commercially unsustainable for large-scale production. Alternative routes involving bromoacetonitrile require complex protection and deprotection steps, often necessitating expensive platinum oxide catalysts for hydrogenation. These factors cumulatively result in a low total recovery rate and a high environmental burden due to the generation of heavy metal waste.
The Novel Approach
In stark contrast, the method disclosed in CN102964259A revolutionizes the production landscape by utilizing 2-Hydroxyphenyl Acetic Acid, a commodity chemical with stable pricing and widespread availability. This strategic shift in starting materials eliminates the dependency on scarce phenolic derivatives. The new route employs a clever protection-deprotection strategy using benzyl groups, which are easily installed and removed under mild conditions. By avoiding the use of platinum catalysts and opting for palladium carbon, the process significantly reduces the cost of goods sold (COGS). The operational simplicity is further enhanced by the fact that intermediate products do not require rigorous purification between steps; simple filtration and extraction suffice. This streamlined workflow not only boosts overall yield but also drastically shortens the production cycle time, offering a compelling value proposition for industrial adoption.
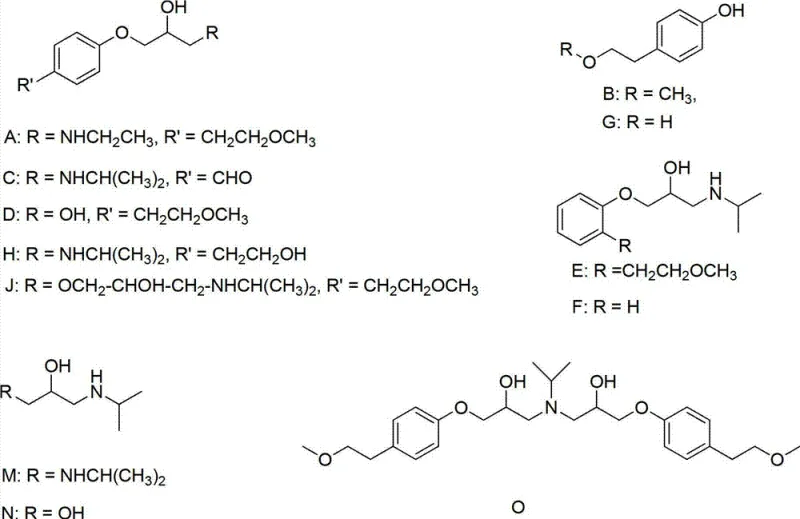
Mechanistic Insights into the Six-Step Synthetic Route
The core of this technology lies in its efficient manipulation of functional groups to construct the complex ether-amine backbone of the target molecule. The process initiates with the reduction of 2-Hydroxyphenyl Acetic Acid to 2-hydroxyphenylethyl alcohol. This is achieved by first activating the carboxylic acid with a chloroformate, such as methyl chloroformate, to form a mixed anhydride in situ. Subsequent reduction with sodium borohydride proceeds smoothly under alkaline conditions, typically using triethylamine as the base. This step is critical as it sets the stereochemical and structural foundation for the subsequent etherification. The choice of sodium borohydride over more aggressive reducing agents ensures high chemoselectivity, preserving the phenolic hydroxyl group for later functionalization while minimizing side reactions that could lead to difficult-to-remove impurities.
Following the initial reduction, the synthesis proceeds through a series of protection and coupling reactions designed to maximize yield and purity. The phenolic hydroxyl is protected as a benzyl ether using benzyl chloride, followed by methylation of the side-chain alcohol using methyl iodide and sodium hydride. A key innovation is the subsequent hydrogenolysis step using palladium carbon to remove the benzyl protecting group, regenerating the phenol without affecting the newly formed methoxy ether. The final stages involve a phase-transfer catalyzed reaction with epichlorohydrin to form the glycidyl ether, followed by ring-opening with isopropylamine. The use of tetra-n-butyl ammonium bromide as a phase transfer catalyst accelerates the reaction rate and significantly reduces impurity formation, ensuring a final product purity of 99.2% as confirmed by HPLC analysis.
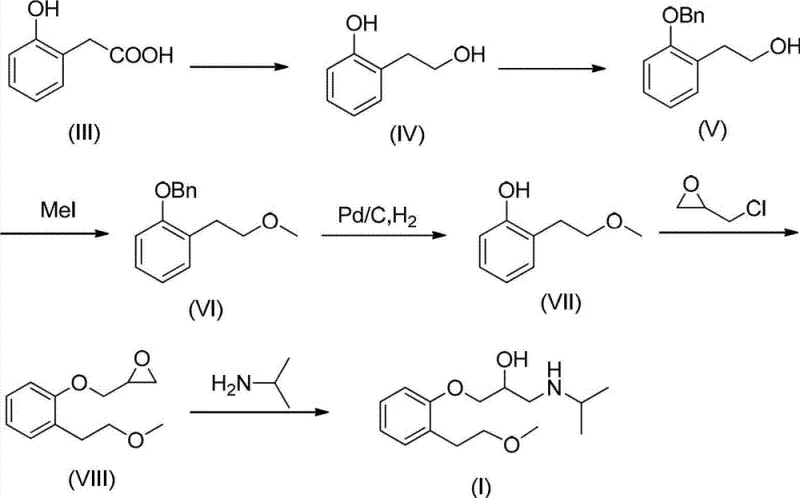
How to Synthesize Metoprolol Related Substance E Efficiently
Implementing this synthesis requires precise control over reaction parameters to replicate the high yields reported in the patent data. The process is designed to be telescoped where possible, minimizing the handling of intermediates and reducing solvent consumption. The initial reduction and protection steps are exothermic and require temperature control between 0°C and 10°C to prevent side reactions. The hydrogenolysis step is conducted at room temperature, offering energy savings compared to high-temperature alternatives. For the final amine opening, refluxing in isopropanol ensures complete conversion. Detailed standard operating procedures (SOPs) regarding reagent stoichiometry, addition rates, and workup protocols are essential for technology transfer. The following section outlines the structural framework for the standardized synthesis steps.
- Reduction of 2-Hydroxyphenyl Acetic Acid to 2-hydroxyphenylethyl alcohol using chloroformates and sodium borohydride.
- Protection of the phenolic hydroxyl group via benzylation followed by methylation of the side-chain alcohol.
- Deprotection via palladium-catalyzed hydrogenolysis, followed by phase-transfer catalyzed epoxidation and isopropylamine ring opening.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates into tangible strategic benefits beyond mere technical feasibility. The shift from exotic starting materials to commodity chemicals like 2-Hydroxyphenyl Acetic Acid mitigates the risk of supply disruptions and price volatility. Traditional routes relying on specialized phenols are vulnerable to single-source supplier issues, whereas the new method leverages a robust global market for basic organic acids. Furthermore, the elimination of expensive platinum catalysts and the reduction in purification steps lead to a substantial decrease in manufacturing costs. This cost efficiency allows for more competitive pricing in the tendering process for API intermediates. The simplified workup procedures also mean faster batch turnover, enhancing the overall agility of the supply chain to respond to market demands.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the replacement of high-cost reagents with affordable alternatives. By avoiding the use of platinum oxide and expensive starting phenols, the direct material cost is significantly lowered. Additionally, the ability to use crude intermediates directly in subsequent steps reduces solvent usage and labor hours associated with chromatography or recrystallization. This lean manufacturing approach results in a lower cost per kilogram, providing a competitive edge in the pricing of high-purity pharmaceutical intermediates without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available reagents such as sodium borohydride, benzyl chloride, and epichlorohydrin. These chemicals are produced by multiple manufacturers globally, reducing the risk of bottlenecks. The robustness of the reaction conditions, which tolerate standard industrial equipment and do not require cryogenic temperatures or ultra-high vacuum, further ensures consistent production schedules. This reliability is crucial for maintaining the continuity of API production lines, preventing costly downtime, and ensuring that inventory levels remain optimized to meet regulatory and commercial deadlines.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route offers distinct advantages. The avoidance of heavy metal catalysts like platinum simplifies waste treatment and reduces the environmental footprint of the manufacturing process. The high atom economy of the reduction and coupling steps minimizes waste generation. Furthermore, the process has been demonstrated to scale effectively, with yields remaining consistent from gram to kilogram scales. This scalability ensures that the method is viable for commercial production volumes ranging from 100 kgs to 100 MT annually, meeting the rigorous demands of the global pharmaceutical market while adhering to increasingly strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits for potential partners. Understanding these details is vital for assessing the feasibility of integrating this route into existing manufacturing portfolios. The answers reflect the specific advantages in terms of purity, cost, and operational simplicity that distinguish this technology from legacy methods.
Q: What is the primary advantage of this synthesis route over prior art?
A: The primary advantage is the use of readily available 2-Hydroxyphenyl Acetic Acid as a starting material, avoiding the expensive and hard-to-source 2-(2-methoxyethyl) phenol used in previous methods, significantly reducing raw material costs.
Q: How is high purity achieved in the final product?
A: High purity (99.2% by HPLC) is achieved through a streamlined process where intermediates do not require complex purification, relying instead on efficient extraction and crystallization steps that minimize impurity carryover.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes common reagents like sodium borohydride and palladium carbon, and avoids expensive platinum catalysts, making it highly scalable and environmentally friendly for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Metoprolol Related Substance E Supplier
The technical potential of the CN102964259A synthesis route is immense, offering a pathway to high-quality intermediates that meet the stringent requirements of modern cardiovascular drug manufacturing. NINGBO INNO PHARMCHEM stands ready to leverage this technology, bringing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, ensuring that every batch of Metoprolol Related Substance E delivered meets the highest international standards. We understand the critical nature of reference standards and impurities in the regulatory approval process and are committed to providing materials that facilitate your compliance and quality assurance goals.
We invite you to explore how this optimized synthesis can enhance your supply chain efficiency and reduce overall manufacturing costs. Our technical procurement team is available to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a reliable source of complex pharmaceutical intermediates, backed by deep technical expertise and a commitment to long-term supply stability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
