Scalable Synthesis of Chiral 2-Amino-1-phenylethylalcohol for Global Beta-Agonist Production
Scalable Synthesis of Chiral 2-Amino-1-phenylethylalcohol for Global Beta-Agonist Production
The pharmaceutical industry's relentless pursuit of high-purity chiral intermediates has led to significant advancements in the synthesis of phenylethanolamine derivatives, which serve as the backbone for critical beta-agonist medications treating asthma and heart failure. Patent CN101575298B introduces a robust and economically viable methodology for preparing chiral 2-amino-1-phenylethylalcohol, a pivotal building block for drugs such as Albuterol, Formoterol, and Salmeterol. Unlike traditional resolution methods that suffer from theoretical yield limitations of 50%, or asymmetric syntheses requiring costly transition metal catalysts, this invention leverages a chiral pool strategy starting from readily available chiral 1,2-styrolylalcohol. The process ensures the transfer of chirality with high fidelity, delivering both R and S enantiomers with exceptional optical purity suitable for stringent regulatory requirements. By utilizing a sequence of selective tosylation, nucleophilic substitution, and hydrazinolysis, the technology provides a reliable pathway for producing these vital intermediates without the burden of expensive chiral auxiliaries.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of optically pure 2-amino-1-phenylethyl alcohol has been fraught with challenges that hinder efficient large-scale manufacturing. Traditional resolution techniques, such as those employing di-p-toluoyl-L-tartaric acid, are inherently inefficient because they discard half of the racemic material, leading to substantial waste and increased raw material costs. Furthermore, enzymatic resolution methods, while specific, often struggle with substrate loading limits and require precise control of biological conditions that are difficult to maintain in massive industrial reactors. On the other hand, direct asymmetric synthesis routes, such as the asymmetric reduction of alpha-halo acetophenones using rhodium-BINAP complexes or CBS borane reductions, involve prohibitively expensive chiral ligands and catalysts. These metal-based systems not only drive up the cost of goods significantly but also introduce the risk of heavy metal contamination, necessitating complex and costly purification steps to meet ppm-level impurity specifications required for active pharmaceutical ingredients (APIs).
The Novel Approach
The methodology outlined in the patent data presents a paradigm shift by adopting a chiral pool synthesis that bypasses the need for external chiral induction during the reaction sequence. By starting with commercially sourced chiral 1,2-phenylethyl alcohol, which can be produced via established Sharpless asymmetric oxidation on a multi-ton scale, the process anchors the stereochemistry at the very beginning. The innovation lies in the chemoselective protection of the primary hydroxyl group via tosylation, leaving the chiral benzylic center undisturbed. This is followed by a Gabriel synthesis-type substitution using potassium phthalimide, a reagent known for its reliability and ease of handling. Finally, the removal of the phthalimide protecting group via hydrazinolysis yields the free amine. This route is characterized by mild reaction conditions, the absence of precious metals, and the use of commodity chemicals, making it uniquely suited for cost-sensitive, high-volume production environments where supply chain stability is paramount.
Mechanistic Insights into Chemo-selective Tosylation and Substitution
The core chemical elegance of this process resides in the precise differentiation between the two hydroxyl groups present in the starting 1,2-diol. In the first step, the reaction employs Tosyl chloride in the presence of triethylamine and a catalytic amount of dibutyltin oxide. The tin catalyst plays a crucial role in activating the primary hydroxyl group over the secondary benzylic hydroxyl group, ensuring that the tosylate forms exclusively at the terminal carbon. This selectivity is vital because any tosylation at the benzylic position could lead to racemization or elimination side reactions under subsequent basic conditions. The reaction is typically conducted at controlled temperatures between 25°C and 30°C, preventing thermal degradation while ensuring complete conversion. The resulting chiral 2-tosic acid-1-phenyl-1,2-glycol serves as an activated electrophile, primed for nucleophilic attack while preserving the stereochemical integrity of the adjacent chiral center.
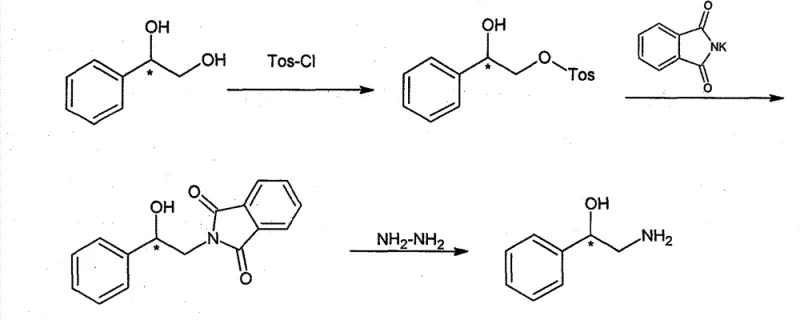
Following the activation step, the intermediate undergoes a nucleophilic substitution with potassium phthalimide in dimethylformamide (DMF) at elevated temperatures around 120°C. This step effectively installs the nitrogen atom required for the final amine functionality. Since the reaction occurs at the primary carbon (the site of the tosylate), the chiral benzylic carbon is not involved in the bond-breaking or bond-forming events of this step, thereby guaranteeing the retention of configuration. The final deprotection utilizes hydrazine hydrate in ethanol under reflux, a classic method for cleaving phthalimides. Hydrazine attacks the carbonyl carbons of the phthalimide ring, releasing the free amine and forming phthalhydrazide as a byproduct, which is easily removed by filtration. This mechanistic pathway ensures that the optical purity of the starting material is quantitatively transferred to the final product, yielding 2-amino-1-phenylethylalcohol with high enantiomeric excess.
How to Synthesize 2-Amino-1-phenylethylalcohol Efficiently
The synthesis of this critical chiral intermediate is streamlined into three distinct operational stages that balance reaction efficiency with ease of purification. The process begins with the activation of the chiral diol, followed by the introduction of the nitrogen source, and concludes with the liberation of the amine. Each step has been optimized in the patent examples to demonstrate scalability, with embodiment data showing successful translation from gram-scale laboratory experiments to kilogram-scale pilot runs. The detailed standardized synthetic steps, including specific molar ratios, solvent choices, and temperature profiles necessary for reproducible high-yield production, are outlined in the technical guide below.
- Perform selective mono-tosylation of chiral 1,2-phenylethyl alcohol using Tosyl chloride and triethylamine, optionally catalyzed by dibutyltin oxide, to protect the primary hydroxyl group.
- Conduct a nucleophilic substitution reaction between the resulting tosylate and potassium phthalimide in DMF at elevated temperatures (approx. 120°C) to introduce the nitrogen functionality.
- Execute a hydrazinolysis reaction using hydrazine hydrate in ethanol under reflux conditions to cleave the phthalimide protecting group and yield the final free amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers profound strategic benefits that extend beyond simple unit cost calculations. By eliminating the dependency on volatile markets for precious metal catalysts like rhodium or specialized chiral ligands, manufacturers can stabilize their raw material costs and reduce exposure to supply disruptions. The use of commodity reagents such as Tosyl chloride, potassium phthalimide, and hydrazine hydrate ensures that sourcing is straightforward and competitive, with multiple global suppliers available to mitigate risk. Furthermore, the process avoids the need for complex chromatographic separations often required in asymmetric catalysis, relying instead on crystallization and filtration, which significantly lowers capital expenditure on processing equipment and reduces solvent consumption.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the substitution of expensive catalytic systems with stoichiometric reagents that are inexpensive and widely available. By removing the requirement for transition metal catalysts, the downstream processing costs associated with metal scavenging and validation are completely eradicated. Additionally, the high atom economy of the Gabriel synthesis approach minimizes waste generation, leading to lower disposal costs and improved overall process mass intensity. The ability to utilize low-cost chiral starting materials derived from bulk petrochemical feedstocks further drives down the cost basis, making the final intermediate highly competitive in the global marketplace.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the reliance on mature, established chemical transformations rather than novel, unproven technologies. The reagents used in this pathway are standard inventory items for most fine chemical manufacturers, reducing lead times for raw material acquisition. Moreover, the robustness of the reaction conditions—operating at ambient or moderately elevated temperatures without the need for cryogenic cooling or inert atmosphere extremes—simplifies logistics and storage requirements. This operational simplicity translates to higher uptime and consistent output, ensuring that downstream API manufacturers receive their critical intermediates on schedule without interruption.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this route is exceptionally well-suited for industrial expansion. The patent data explicitly demonstrates successful scale-up to multi-kilogram batches, proving that heat transfer and mixing issues are manageable in large reactors. The avoidance of heavy metals aligns perfectly with increasingly stringent environmental regulations regarding wastewater discharge and residual impurities in pharmaceutical products. The solid byproducts generated, such as phthalhydrazide, are easy to isolate and dispose of, reducing the burden on effluent treatment plants and facilitating a greener manufacturing profile that appeals to environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of chiral 2-amino-1-phenylethylalcohol. These answers are derived directly from the technical specifications and comparative data provided in the patent literature, offering clarity on why this specific synthetic route is gaining traction among forward-thinking chemical manufacturers. Understanding these nuances is essential for making informed decisions about supplier qualification and process integration.
Q: Why is this chiral pool method preferred over asymmetric catalysis for 2-amino-1-phenylethylalcohol?
A: While asymmetric catalysis (e.g., CBS reduction) offers high enantioselectivity, it often relies on expensive chiral ligands and metal catalysts that are difficult to remove completely. The chiral pool method described in CN101575298B utilizes commercially available, low-cost chiral 1,2-phenylethyl alcohol (derived from Sharpless oxidation), eliminating the need for precious metal catalysts and simplifying the purification process for industrial scale-up.
Q: How does the process ensure the retention of optical configuration?
A: The synthetic route strategically targets the primary hydroxyl group of the 1,2-diol for functionalization while leaving the chiral benzylic center untouched. By performing selective tosylation at the primary position followed by substitution, the stereocenter at the benzylic carbon remains intact throughout the sequence, ensuring the final amine product retains the high optical purity of the starting material.
Q: What are the key advantages for large-scale manufacturing of this intermediate?
A: The process avoids cryogenic conditions typically required for some asymmetric syntheses, operating instead at manageable temperatures (25-30°C for tosylation, 120°C for substitution). Furthermore, the use of robust reagents like potassium phthalimide and hydrazine hydrate allows for straightforward workup procedures, such as filtration and recrystallization, which are highly favorable for ton-scale production compared to complex chromatographic separations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-1-phenylethylalcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of your final therapeutic product is inextricably linked to the purity and consistency of your starting materials. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering reliability. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to verify stringent purity specifications, guaranteeing that every batch of chiral intermediate meets the exacting standards required for global regulatory filings. We are committed to delivering not just a chemical product, but a partnership built on technical excellence and supply chain security.
We invite you to engage with our technical procurement team to discuss how our optimized manufacturing processes can support your project timelines and budgetary goals. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our efficient synthetic routes can reduce your overall cost of goods. We encourage potential partners to contact us directly to obtain specific COA data and comprehensive route feasibility assessments tailored to your unique development requirements.
