Scalable Visible Light Copper Catalysis for Commercial Production of Complex Isoquinolinones
Scalable Visible Light Copper Catalysis for Commercial Production of Complex Isoquinolinones
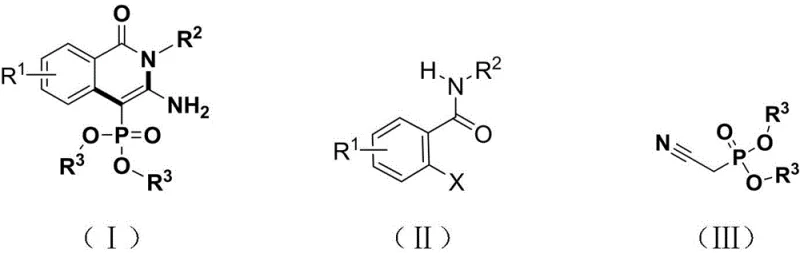
The pharmaceutical industry is constantly seeking efficient pathways to access structurally diverse heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. Patent CN112142796A introduces a groundbreaking synthesis method for C-4-site phosphate ester substituted 2-aminoisoquinolinone derivatives, utilizing a visible light-promoted copper catalysis strategy. This innovation addresses the longstanding challenges associated with constructing phosphorus-containing nitrogen heterocycles, which are increasingly recognized for their potent anticancer, antibacterial, and antiviral activities. By leveraging near-blue light irradiation at wavelengths between 420nm and 470nm, the process achieves high conversion rates under exceptionally mild conditions of normal temperature and pressure. This technological leap not only simplifies the synthetic route but also aligns perfectly with the modern demands for green chemistry and sustainable manufacturing practices in the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing isoquinolinone frameworks often rely on harsh thermal conditions, requiring elevated temperatures and prolonged reaction times that can degrade sensitive functional groups. These conventional methods frequently necessitate the use of expensive palladium or rhodium catalysts, which introduce significant cost burdens and complicate the removal of toxic heavy metal residues from the final active pharmaceutical ingredients. Furthermore, multi-step sequences involving protection and deprotection strategies are commonly required to achieve regioselectivity, leading to lower overall atom economy and increased waste generation. The reliance on high-energy inputs and complex purification protocols creates substantial bottlenecks for procurement teams aiming to reduce the cost of goods sold for key intermediates. Additionally, the limited substrate scope of many thermal coupling reactions restricts the ability of R&D departments to rapidly explore structure-activity relationships for new drug candidates.
The Novel Approach
In stark contrast, the novel approach detailed in the patent employs a visible light-promoted copper catalytic system that operates efficiently at room temperature, drastically reducing energy consumption and operational complexity. By utilizing earth-abundant copper salts combined with a specialized phenanthroline ligand, the method achieves high catalytic activity without the need for precious metals, thereby offering a compelling value proposition for cost reduction in pharmaceutical intermediate manufacturing. The one-pot nature of this transformation merges the coupling and cyclization steps, effectively shortening the synthetic timeline and minimizing the handling of hazardous intermediates. This streamlined process demonstrates exceptional compatibility with a wide range of substituents on both the benzamide and phosphate components, enabling the rapid generation of diverse chemical libraries. The mild conditions also preserve the integrity of labile groups, ensuring higher purity profiles and reducing the burden on downstream processing units.
Mechanistic Insights into Visible Light-Promoted Copper Catalysis
The core of this transformative synthesis lies in the intricate interplay between the copper catalyst and visible light energy, which drives the reaction through a unique photoredox cycle. Initially, the substituted cyanomethyl phosphate compound reacts with the copper catalyst and the 4,7-diphenyl-1,10-phenanthroline ligand in the presence of a base to form a monovalent copper intermediate. Upon irradiation with near-blue light, this ground-state complex absorbs photons to reach an excited state, significantly enhancing its reducing power and facilitating the subsequent oxidative addition step with the o-halogenated benzamide derivative. This photo-excitation is crucial as it lowers the activation energy barrier for the carbon-phosphorus bond formation, allowing the reaction to proceed smoothly without external heating. The resulting trivalent copper intermediate then undergoes reductive elimination to forge the new C-C bond, releasing the coupled product and regenerating the active copper species for the next catalytic turnover.
Following the initial coupling event, the reaction pathway diverges into an intramolecular cyclization sequence that constructs the characteristic isoquinolinone core. The intermediate formed after reductive elimination undergoes a copper-assisted nucleophilic addition, where the amide nitrogen attacks the nitrile carbon to close the ring. This step is followed by an isomerization process that stabilizes the final structure, yielding the thermodynamically favored C-4 phosphate substituted 2-aminoisoquinolinone derivative. The precise control over the oxidation states of copper, mediated by the ligand environment and light intensity, ensures high selectivity and minimizes the formation of homocoupling byproducts. Understanding this mechanistic nuance is vital for R&D directors aiming to optimize reaction parameters for specific substrates, as slight adjustments in light wavelength or ligand loading can profoundly impact the efficiency of the catalytic cycle.
How to Synthesize C-4 Phosphate Substituted 2-Aminoisoquinolinone Derivatives Efficiently
The practical implementation of this synthesis involves a straightforward protocol that can be easily adapted for both laboratory screening and pilot-scale production. Operators begin by charging a reaction vessel with the requisite o-halogenated benzamide derivative and the substituted cyanomethyl phosphate compound in a molar ratio optimized for maximum yield. The copper salt catalyst and the bidentate ligand are added along with a suitable organic polar solvent such as DMSO or DMF, followed by the introduction of a base like potassium carbonate or triethylamine. After ensuring an inert nitrogen atmosphere to prevent catalyst oxidation, the mixture is subjected to blue LED irradiation while stirring at ambient temperature. Detailed standardized synthesis steps follow below to guide technical teams through the precise execution of this methodology.
- Prepare the reaction mixture by combining o-halogenated benzamide derivatives, substituted cyanomethyl phosphate compounds, copper salt catalyst, and 4,7-diphenyl-1,10-phenanthroline ligand in an organic polar solvent.
- Add a carbonate or nitrogenous organic base to the mixture, purge the system with nitrogen gas three times to ensure an inert atmosphere, and position the reactor under 420nm-470nm blue LED irradiation.
- Stir the reaction at room temperature for 3 to 10 hours, then perform aqueous workup followed by silica gel column chromatography to isolate the high-purity target derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this visible light-mediated technology presents a strategic opportunity to enhance supply reliability while driving down manufacturing costs. The shift from precious metal catalysts to inexpensive copper salts eliminates the volatility associated with rhodium or palladium pricing, stabilizing the raw material budget for long-term production contracts. Moreover, the simplified one-pot procedure reduces the number of unit operations required, which directly translates to lower labor costs and decreased consumption of solvents and consumables. The ability to run reactions at room temperature also alleviates the strain on facility utilities, as there is no need for extensive heating or cooling infrastructure, further contributing to substantial cost savings in utility overheads.
- Cost Reduction in Manufacturing: The replacement of expensive noble metal catalysts with abundant copper salts fundamentally alters the cost structure of producing these complex intermediates. By eliminating the need for rigorous heavy metal scavenging steps typically required for palladium-catalyzed processes, manufacturers can significantly reduce the cost of purification materials and waste disposal. The high atom economy of the one-pot reaction minimizes raw material waste, ensuring that a greater proportion of input costs are converted into valuable product output. Additionally, the mild reaction conditions reduce energy expenditures associated with heating and cooling, leading to a leaner and more cost-effective production model overall.
- Enhanced Supply Chain Reliability: Utilizing readily available starting materials such as o-halogenated benzamides and cyanomethyl phosphates ensures a robust supply chain that is less susceptible to geopolitical disruptions or shortages of exotic reagents. The simplicity of the reaction setup allows for flexible manufacturing scheduling, as the process does not require specialized high-pressure reactors or complex thermal management systems. This operational flexibility enables suppliers to respond more rapidly to fluctuating market demands, reducing lead times for high-purity pharmaceutical intermediates. Furthermore, the stability of the copper catalyst system under ambient conditions simplifies storage and handling logistics, minimizing the risk of supply interruptions due to reagent degradation.
- Scalability and Environmental Compliance: The transition from batch thermal processes to continuous flow photochemistry is highly feasible with this methodology, offering a clear path for commercial scale-up of complex pharmaceutical intermediates. The reduction in hazardous waste generation, driven by higher selectivity and fewer purification steps, aligns with increasingly stringent environmental regulations and corporate sustainability goals. The absence of toxic heavy metals in the catalyst system simplifies effluent treatment processes, lowering the environmental compliance burden on manufacturing sites. This eco-friendly profile not only mitigates regulatory risks but also enhances the brand reputation of companies adopting green chemistry principles in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible light-promoted synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and benefits for potential partners. Understanding these aspects is crucial for making informed decisions about integrating this novel route into existing production portfolios.
Q: What are the primary advantages of using visible light promotion in this synthesis?
A: The use of visible light promotion allows the reaction to proceed at room temperature and normal pressure, eliminating the need for energy-intensive heating or high-pressure equipment. This significantly reduces operational costs and enhances safety profiles while maintaining high yields and excellent functional group tolerance.
Q: Is this copper-catalyzed method suitable for large-scale industrial production?
A: Yes, the method utilizes earth-abundant copper salts and simple LED light sources, making it highly scalable. The one-pot procedure minimizes intermediate isolation steps, which streamlines the workflow for commercial scale-up of complex pharmaceutical intermediates.
Q: How does this method improve the impurity profile compared to traditional thermal methods?
A: The mild reaction conditions prevent thermal degradation of sensitive functional groups and reduce the formation of side products often associated with high-temperature coupling reactions. This results in a cleaner crude product, simplifying downstream purification and ensuring stringent purity specifications for drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aminoisoquinolinone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of visible light-promoted copper catalysis in accelerating the development of novel therapeutic agents. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless and efficient. Our state-of-the-art facilities are equipped with advanced photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications for even the most complex heterocyclic structures. We are committed to delivering high-quality intermediates that empower your R&D teams to push the boundaries of medicinal chemistry without being constrained by supply limitations.
We invite you to collaborate with our technical procurement team to explore how this innovative synthesis method can optimize your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits tailored to your volume needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that demonstrate our capability to be your trusted partner in bringing life-saving medicines to market faster and more affordably.
