Advanced Ambient-Pressure Synthesis of N-Ethylaniline for High-Purity Pharmaceutical Intermediates
The chemical industry is currently witnessing a significant paradigm shift in the synthesis of key aromatic amines, driven by the urgent need for safer and more efficient manufacturing protocols. Patent CN103145562A introduces a groundbreaking preparation method for N-ethylaniline that fundamentally alters the reaction landscape by replacing hazardous high-pressure conditions with a mild, ambient-pressure protocol. This innovation utilizes aniline and acetaldehyde as primary starting materials to synthesize a Schiff base intermediate, which is subsequently reduced to the target amine. For R&D directors and process engineers, this represents a critical evolution away from the legacy reliance on ethanol and concentrated hydrochloric acid under extreme thermal stress. The transition to this reductive amination pathway not only enhances the safety profile of the operation but also dramatically improves the atom economy and final product quality. By operating at normal temperature and pressure, the technology effectively decouples production capacity from the limitations of specialized high-pressure autoclaves, offering a robust solution for the reliable pharmaceutical intermediates supplier market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the domestic and international production of N-ethylaniline has been dominated by a cumbersome alkylation process involving aniline and ethanol under aggressive acidic conditions. This traditional methodology necessitates the use of concentrated hydrochloric acid as a catalyst and requires reaction environments reaching temperatures as high as 300°C and pressures up to 9.84MPa. Such extreme parameters impose severe constraints on equipment integrity, leading to rapid corrosion of reactor vessels and necessitating frequent, costly maintenance cycles. Furthermore, the thermodynamic conditions favor uncontrolled poly-alkylation, resulting in a crude reaction mixture containing significant quantities of N,N-diethylaniline impurities alongside unreacted aniline. The separation of these closely boiling components is energy-intensive and often results in suboptimal overall yields, typically hovering between 65% and 70%. From a supply chain perspective, the reliance on high-pressure infrastructure creates bottlenecks in cost reduction in pharmaceutical intermediates manufacturing, as the capital expenditure for safety-compliant autoclaves is substantial.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages the higher reactivity of acetaldehyde compared to ethanol to facilitate nucleophilic attack under benign conditions. By shifting the reactant profile to aniline and acetaldehyde, the synthesis proceeds efficiently at normal temperature and pressure, completely eliminating the need for external heating sources or pressurized containment systems. This methodological pivot allows for the precise formation of a Schiff base intermediate, which serves as a distinct chemical handle for subsequent selective reduction. The avoidance of strong mineral acids like hydrochloric acid mitigates corrosion issues entirely, extending the operational lifespan of standard glass-lined or stainless steel reactors. Consequently, the process achieves a transformation efficiency that far exceeds prior art, with reported yields surpassing 96% and product purity levels exceeding 97%. This leap in performance underscores the viability of this route for the commercial scale-up of complex fine chemicals, providing a streamlined pathway that aligns with modern green chemistry principles.
Mechanistic Insights into Reductive Amination of Aniline
The core of this technological advancement lies in the two-stage mechanistic sequence involving nucleophilic addition followed by hydride reduction. Initially, the lone pair of electrons on the nitrogen atom of the aniline molecule performs a nucleophilic attack on the electrophilic carbonyl carbon of the acetaldehyde. This interaction leads to the elimination of a water molecule and the formation of an imine, commonly referred to as a Schiff base. This step is kinetically favorable at ambient temperatures due to the high electrophilicity of the aldehyde group compared to the hydroxyl group of ethanol. The stability of this intermediate is crucial, as it prevents the runaway alkylation seen in the ethanol-based route. Following the formation of the imine, the introduction of a reducing agent, specifically sodium borohydride (NaBH4) or similar hydride sources, facilitates the transfer of hydride ions to the carbon-nitrogen double bond. This reduction step saturates the bond, converting the imine into the secondary amine, N-ethylaniline, with high selectivity.
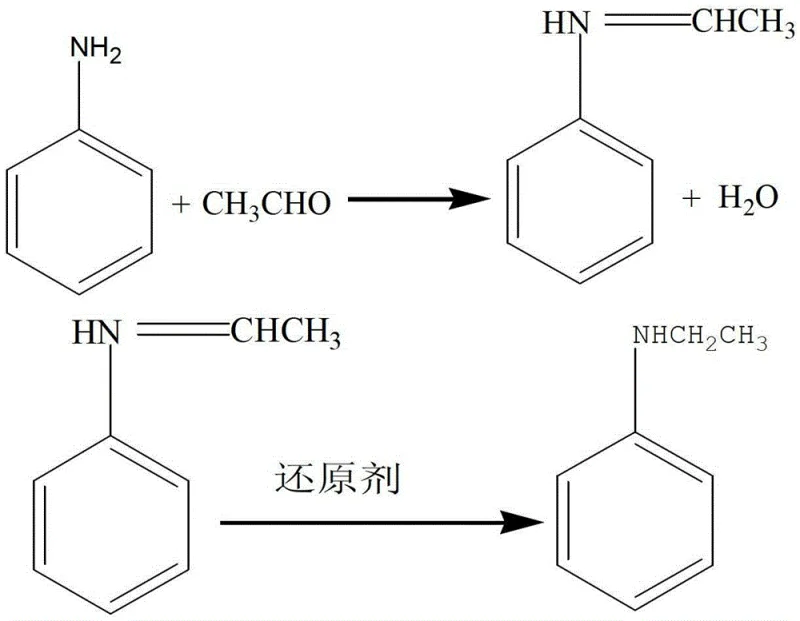
Controlling the stoichiometry of the reducing agent is paramount to minimizing impurity profiles and maximizing yield. The patent specifies a molar ratio of aniline to reducing agent between 1:0.25 and 1:0.5, ensuring complete conversion of the imine without introducing excess boron-containing byproducts that could complicate downstream purification. The use of alcoholic solvents, such as ethanol or methanol, further stabilizes the transition states and ensures homogeneous mixing of the reagents. By maintaining the reaction temperature between 10°C and 30°C, the process avoids the volatility issues associated with acetaldehyde while preventing thermal degradation of the product. This precise control over the reaction environment results in a crude product with minimal N,N-diethylaniline contamination, significantly reducing the burden on the distillation columns during the workup phase. For quality assurance teams, this mechanistic clarity translates to a predictable and reproducible impurity spectrum, facilitating easier regulatory compliance for high-purity OLED material or pharmaceutical applications.
How to Synthesize N-Ethylaniline Efficiently
The operational procedure for this synthesis is designed for simplicity and scalability, making it highly attractive for contract development and manufacturing organizations. The process begins with the preparation of an alcoholic solution of acetaldehyde, which is then added to a reactor containing aniline under constant stirring at ambient conditions. This initial nucleophilic phase is allowed to proceed for approximately 0.5 to 1.5 hours, sufficient time to ensure the complete consumption of the aniline and formation of the Schiff base. Subsequently, the reducing agent, dissolved in the same alcoholic solvent to maintain homogeneity, is introduced to the reaction mixture. The reduction phase typically requires 2 to 4 hours to reach completion, monitored via liquid chromatography to ensure the normalized content of N-ethylaniline exceeds 98%. The detailed standardized synthesis steps, including specific workup procedures like alkalization and acidification for phase separation, are outlined below to guide process implementation.
- Conduct a nucleophilic reaction between aniline and an alcoholic solution of acetaldehyde at normal temperature and pressure for 0.5 to 1.5 hours to form the Schiff base intermediate.
- Introduce a reducing agent, such as sodium borohydride dissolved in alcohol, to the reaction mixture and stir for 2 to 4 hours to reduce the imine bond to the amine.
- Perform post-treatment involving distillation, alkalization with NaOH to separate organic phases, acidification with sulfuric acid, and final redistillation to isolate high-purity N-ethylaniline.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ambient-pressure synthesis route offers profound strategic advantages that extend beyond simple unit cost calculations. The elimination of high-pressure and high-temperature requirements fundamentally changes the capital expenditure model for production facilities. Manufacturers no longer need to invest in expensive, specialized autoclaves rated for nearly 10MPa of pressure, nor do they need to allocate significant budgets for the maintenance and certification of such hazardous equipment. This reduction in infrastructure complexity directly translates to lower overhead costs and faster deployment of new production lines. Furthermore, the use of non-corrosive reagents eliminates the recurring cost of replacing reactor linings damaged by concentrated hydrochloric acid, thereby enhancing the long-term economic viability of the manufacturing process. These factors collectively contribute to substantial cost savings in fine chemical intermediates manufacturing, allowing suppliers to offer more competitive pricing without compromising on margin.
- Cost Reduction in Manufacturing: The shift to ambient pressure operations removes the energy intensity associated with heating reactors to 300°C and compressing gases to high pressures. This drastic reduction in energy consumption lowers the variable cost per kilogram of product significantly. Additionally, the higher selectivity of the reaction minimizes the loss of raw materials to side products like N,N-diethylaniline, improving the overall material utilization rate. By avoiding the use of corrosive acids, the facility saves on waste treatment costs associated with neutralizing acidic effluents and disposing of corroded metal parts. These cumulative efficiencies create a leaner production model that is resilient to fluctuations in energy prices.
- Enhanced Supply Chain Reliability: Operating under normal temperature and pressure conditions significantly reduces the risk of unplanned shutdowns due to equipment failure or safety incidents. High-pressure systems are inherently prone to leaks and mechanical fatigue, which can disrupt production schedules and delay deliveries to downstream clients. The simplified equipment requirements also mean that the process can be easily transferred between different manufacturing sites or scaled up using standard off-the-shelf reactors. This flexibility ensures a more stable supply of high-purity pharmaceutical intermediates, reducing lead times and enhancing the reliability of the supply chain for global partners who depend on just-in-time delivery models.
- Scalability and Environmental Compliance: The benign nature of the reagents and conditions simplifies the environmental permitting process for new or expanded facilities. Without the need for high-pressure safety valves and complex pressure relief systems, the regulatory burden is lighter, accelerating the time-to-market for commercial scale production. The process generates fewer hazardous byproducts, aligning with increasingly stringent global environmental regulations regarding industrial emissions and waste disposal. This environmental compatibility not only safeguards the company's reputation but also future-proofs the manufacturing asset against tightening ecological standards, ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel N-ethylaniline synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of the process benefits. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific supply chain needs.
Q: What are the safety advantages of the new N-ethylaniline synthesis method compared to traditional processes?
A: The novel method operates at normal temperature and pressure, eliminating the need for high-pressure autoclaves (previously requiring up to 9.84MPa) and corrosive concentrated hydrochloric acid, thereby significantly reducing equipment corrosion risks and operational hazards.
Q: How does the acetaldehyde route improve product purity over the ethanol route?
A: By utilizing acetaldehyde instead of ethanol, the process minimizes the formation of N,N-diethylaniline impurities. The controlled stoichiometry and mild conditions allow for yields exceeding 96% with purity levels above 97%, surpassing the 65-70% yields typical of older high-temperature methods.
Q: Can this synthesis method be scaled for commercial production without specialized high-pressure equipment?
A: Yes, the process is designed for scalability using standard reaction vessels capable of handling ambient pressure operations. This removes the capital expenditure barrier associated with maintaining high-pressure, high-temperature reactor systems required by prior art.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Ethylaniline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to meet the evolving demands of the global pharmaceutical and fine chemical industries. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products with stringent purity specifications, leveraging our rigorous QC labs to verify that every batch meets the highest standards of quality and consistency. Our capability to implement the ambient-pressure reductive amination process positions us as a strategic partner capable of supplying high-volume N-ethylaniline with unmatched reliability and cost-effectiveness.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis method can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your manufacturing operations.
