Advanced Solvent-Free Synthesis of Tris(3,6-dioxaheptyl)amine for Industrial Phase-Transfer Catalysis
The chemical industry is constantly seeking more efficient pathways for producing high-performance phase-transfer catalysts and surfactants, and a pivotal advancement in this domain is detailed in patent CN101948394A. This intellectual property introduces a robust method for synthesizing tris(3,6-dioxaheptyl)amine, a compound of significant value in fine chemical applications. Unlike traditional approaches that struggle with selectivity and yield, this novel process utilizes a skeleton Cu-Ni bimetallic catalyst to drive the reductive amination of diglycol monomethyl ether. The strategic shift away from conventional single-metal catalysts represents a major leap forward in process chemistry, offering a solvent-free environment that drastically simplifies the reaction matrix. By operating under normal pressure and moderate temperatures between 130°C and 150°C, the method not only enhances safety profiles but also ensures a high degree of conversion efficiency. For a reliable tris(3,6-dioxaheptyl)amine supplier, adopting such a methodology is crucial for meeting the stringent quality demands of global pharmaceutical and agrochemical markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tris(3,6-dioxaheptyl)amine has been plagued by significant technical hurdles that hinder cost reduction in phase-transfer catalyst manufacturing. Prior art, such as the methods described in US6403834 and US4408075, predominantly relied on Raney Nickel catalysts, which inherently suffer from poor selectivity profiles. When using Raney Nickel, the reaction conditions often necessitate elevated temperatures reaching up to 170°C to achieve acceptable conversion rates, which unfortunately promotes a myriad of side reactions. These side reactions lead to the formation of complex impurity profiles, including various secondary amines and incomplete alkylation products, resulting in a total yield that rarely exceeds 60%. Furthermore, the separation of the desired tertiary amine from these byproducts is notoriously difficult and energy-intensive, requiring extensive rectification processes that erode profit margins. The low selectivity also means that a substantial portion of the valuable diglycol monomethyl ether feedstock is wasted, creating an unsustainable economic model for large-scale production.
The Novel Approach
The innovative strategy outlined in the patent data fundamentally alters the reaction landscape by introducing a skeleton Cu-Ni bimetallic catalyst system. This dual-metal approach leverages the synergistic effects of copper and nickel to lower the activation energy required for the reductive amination, allowing the process to proceed efficiently at temperatures between 130°C and 150°C. A critical feature of this novel approach is the solvent-free condition, which eliminates the need for volatile organic compounds and reduces the overall volume of the reaction mixture, thereby enhancing the throughput of existing reactor infrastructure. By carefully controlling the feeding volume ratio of hydrogen to ammonia within the range of 1:(5-10), the process effectively steers the reaction pathway towards the desired tertiary amine while suppressing the formation of primary and secondary amine intermediates. This precise control results in a dramatic improvement in yield, with patent examples demonstrating conversion efficiencies exceeding 91%, effectively doubling the output compared to legacy Raney Nickel methods.
Mechanistic Insights into Cu-Ni Catalyzed Reductive Amination
Understanding the mechanistic nuances of this transformation is essential for R&D teams aiming to replicate or optimize the process for high-purity tris(3,6-dioxaheptyl)amine production. The reaction proceeds through a classic reductive amination pathway where the diglycol monomethyl ether is initially dehydrogenated to form an aldehyde intermediate under the influence of the Cu-Ni catalyst. This aldehyde subsequently undergoes nucleophilic addition with ammonia to form an imine or enamine species, which are then rapidly hydrogenated to yield the primary amine. However, the true complexity lies in the subsequent alkylation steps where the primary amine reacts further with the aldehyde to form secondary and finally tertiary amines. The presence of copper in the catalyst lattice is believed to modulate the electron density on the nickel active sites, reducing the propensity for ether bond cleavage and aminolysis reactions that typically generate stubborn impurities. This modulation is key to maintaining the integrity of the polyether chains throughout the harsh thermal conditions of the synthesis.
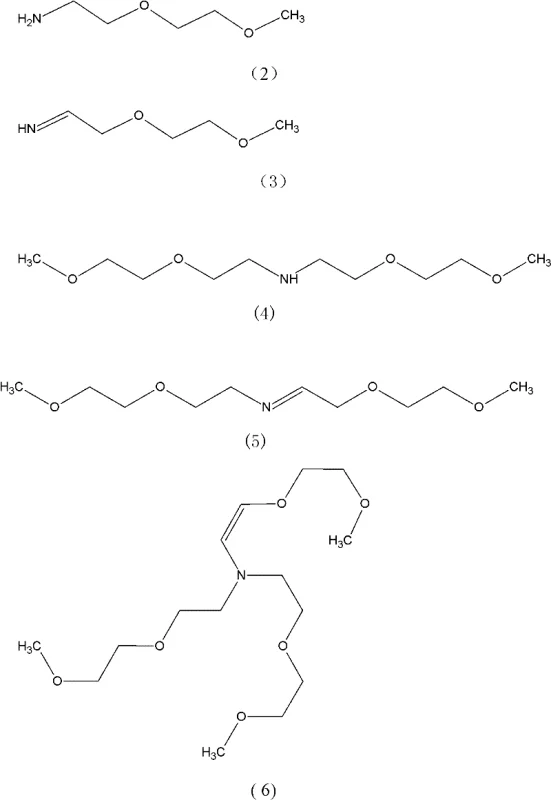
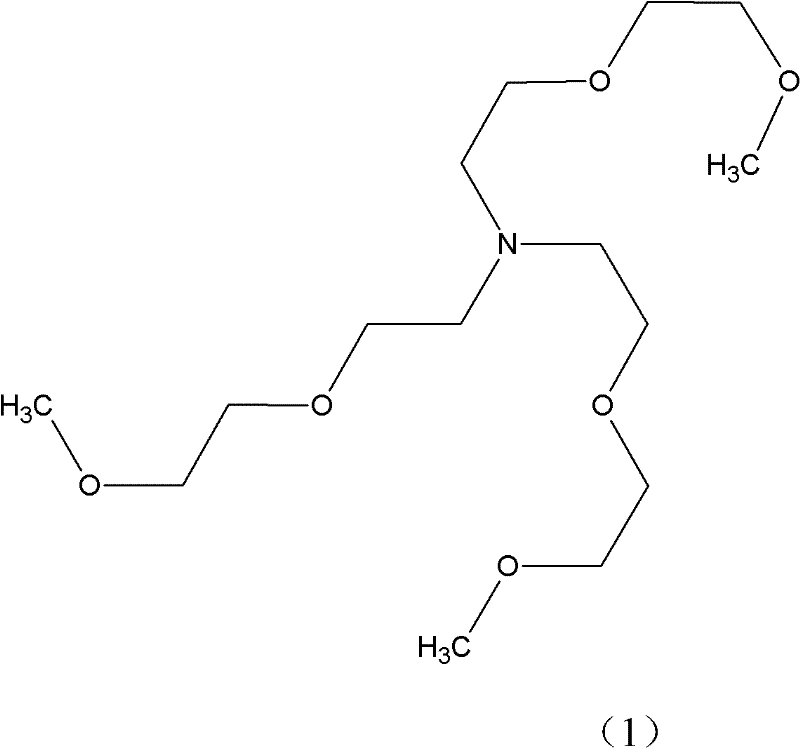
Impurity control is perhaps the most critical aspect of this mechanism, as the presence of secondary amines or incomplete alkylation products can severely impact the performance of the final surfactant. As illustrated in the reaction scheme, intermediates such as the primary amine (Formula 2) and various imine species (Formula 3, 5) are transient but necessary components of the pathway. However, if the reaction is not halted at the precise moment, or if the ammonia concentration drops too early, the system can stagnate at the secondary amine stage (Formula 4) or form mixed alkylated impurities (Formula 7). The patent specifies a sophisticated control strategy where ammonia introduction is stopped only when the mass ratio of the starting ether to the primary amine reaches a specific threshold of (1.5-2):1. This ensures that there is sufficient electrophile available to drive the reaction to completion without leaving excess ammonia that could complicate downstream separation. By continuing hydrogenation until the residual diglycol monomethyl ether is less than 2%, the process guarantees a clean profile suitable for sensitive applications in electronic materials or pharmaceutical synthesis.
How to Synthesize Tris(3,6-dioxaheptyl)amine Efficiently
Implementing this synthesis route requires careful attention to catalyst preparation and gas flow dynamics to ensure reproducibility and safety. The standardized protocol involves the activation of a Cu-Ni alloy powder with sodium hydroxide to create the skeletal catalyst, followed by a rigorous washing procedure to remove residual alkali. Once the catalyst is loaded into the reactor with the ether feedstock, the temperature is ramped up while simultaneously removing any moisture to prevent catalyst deactivation. The introduction of gases must be synchronized with the reaction exotherm to maintain the optimal temperature window, and the venting of excess ammonia must be managed through an absorption tower to comply with environmental regulations. For a detailed breakdown of the specific operational parameters and safety checks required for this transformation, please refer to the standardized synthesis guide below.
- Load diglycol monomethyl ether and activated skeleton Cu-Ni catalyst (20-30% Cu) into a reactor and heat to 130-150°C under stirring.
- Introduce hydrogen and ammonia gas at a volume ratio of 1: (5-10) while maintaining normal pressure and removing generated water.
- Stop ammonia flow when the mass ratio of ether to primary amine reaches 1.5: 1, then continue hydrogenation until ether content is below 2%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this Cu-Ni catalyzed process offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of solvents from the reaction mixture significantly reduces the volume of hazardous waste generated, leading to lower disposal costs and a smaller environmental footprint. This solvent-free approach also means that the reaction vessels can be charged with a higher density of reactants, effectively increasing the batch size capacity of existing infrastructure without the need for capital expenditure on larger tanks. Furthermore, the milder reaction conditions reduce the energy consumption associated with heating and cooling cycles, contributing to a more sustainable and cost-effective manufacturing operation. These factors combined create a robust supply chain capable of delivering consistent quality without the volatility associated with older, less efficient technologies.
- Cost Reduction in Manufacturing: The most immediate financial impact comes from the substantial increase in yield, which moves from roughly 59% in conventional methods to over 91% with the new catalyst. This near-doubling of efficiency means that nearly half the amount of raw material is required to produce the same quantity of finished goods, drastically lowering the cost of goods sold (COGS). Additionally, the high selectivity of the Cu-Ni catalyst minimizes the formation of difficult-to-remove impurities, which simplifies the purification process and reduces the consumption of utilities like steam and cooling water during distillation. By avoiding the use of expensive transition metals that require complex removal steps, the process further streamlines the downstream processing workflow, ensuring that the final product meets stringent purity specifications with minimal additional refinement.
- Enhanced Supply Chain Reliability: The reliance on diglycol monomethyl ether as a sole carbon source simplifies the raw material procurement strategy, as this commodity chemical is widely available from multiple global suppliers. The robustness of the skeleton Cu-Ni catalyst, which can be prepared from readily available alloy powders, ensures that the production process is not bottlenecked by the availability of exotic or proprietary catalytic materials. This stability allows for better forecasting and inventory management, reducing the risk of production stoppages due to supply shortages. Moreover, the ability to operate under normal pressure rather than high-pressure conditions reduces the maintenance requirements for reactor equipment, leading to higher uptime and more reliable delivery schedules for customers relying on just-in-time inventory models.
- Scalability and Environmental Compliance: Scaling up chemical processes often introduces new challenges regarding heat transfer and mixing, but the solvent-free nature of this reaction mitigates many of these risks by reducing the total fluid volume. The absence of volatile solvents also lowers the explosion hazard classification of the facility, potentially reducing insurance premiums and regulatory compliance burdens. From an environmental perspective, the process generates significantly less wastewater and organic waste, aligning with increasingly strict global regulations on industrial emissions. This green chemistry profile makes the manufacturing site more resilient to regulatory changes and enhances the brand reputation of the supplier as a responsible partner in the global chemical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized amine. These answers are derived directly from the technical specifications and experimental data provided in the patent literature to ensure accuracy and relevance for potential partners. Understanding these details is vital for evaluating the feasibility of integrating this material into your specific formulation or synthesis pipeline.
Q: What are the advantages of the Cu-Ni catalyst over traditional Raney Nickel?
A: The skeleton Cu-Ni bimetallic catalyst operates at lower temperatures (130-150°C vs 170°C) and significantly improves selectivity, reducing the formation of secondary amine impurities and increasing yield from approximately 59% to over 91%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the solvent-free nature of the reaction simplifies downstream processing and waste treatment, making it highly suitable for commercial scale-up of complex polyether amines without the need for extensive solvent recovery systems.
Q: How is the purity of the final tris(3,6-dioxaheptyl)amine controlled?
A: Purity is controlled by precisely managing the hydrogen-to-ammonia ratio and stopping ammonia introduction at a specific conversion point, which suppresses the formation of incomplete alkylation impurities like bis-substituted amines.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tris(3,6-dioxaheptyl)amine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced chemical intermediates requires more than just a laboratory recipe; it demands a partner with deep engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. We understand that for applications in pharmaceuticals and electronics, consistency is paramount, which is why our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications for every batch released. By leveraging the advanced Cu-Ni catalytic technology described in CN101948394A, we are positioned to offer a superior grade of tris(3,6-dioxaheptyl)amine that meets the exacting standards of modern industry.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs through the adoption of this superior synthetic route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. We encourage you to reach out to request specific COA data and route feasibility assessments to see firsthand how our capabilities can support your long-term strategic goals. Let us help you secure a stable, high-quality supply of this critical intermediate while driving down your operational expenses.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
