Optimizing Sulcardine Sulfate Production: A Novel Synthetic Route for High-Purity API Intermediates
Optimizing Sulcardine Sulfate Production: A Novel Synthetic Route for High-Purity API Intermediates
The pharmaceutical landscape for anti-arrhythmic agents continues to evolve, driven by the demand for safer, more efficient manufacturing processes for novel structural compounds. Patent CN112110879A introduces a groundbreaking preparation method for Sulcardine free base, a critical precursor to Sulcardine sulfate, a Class III anti-arrhythmic drug with significant clinical potential. This technology addresses long-standing inefficiencies in traditional synthesis by reordering reaction steps to maximize regioselectivity and minimize waste. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, understanding this mechanistic shift is crucial for securing a stable supply chain of high-quality active ingredients.
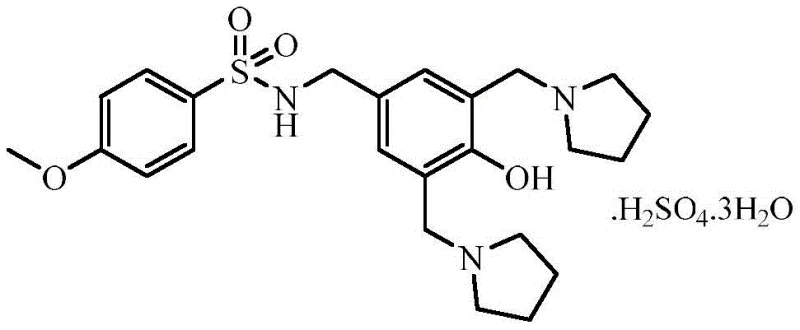
Sulcardine sulfate acts on Na+, K+, and Ca++ plasma channels, offering a relatively safe visceral medicine profile. However, its commercial viability has historically been hampered by complex purification steps and low yields associated with legacy synthetic routes. The disclosed invention pivots away from these bottlenecks, offering a pathway that is not only chemically superior but also industrially robust. By focusing on the strategic protection of the amino group and the timing of the Mannich reaction, the process achieves a total yield of 50-55%, a figure that signals a mature, production-ready methodology suitable for commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Sulcardine sulfate, as detailed in patents like CN1172908C and US6605635B1, relied on a sequence where sulfonylation occurred before the Mannich reaction. This traditional approach suffered from severe regiochemical challenges. When the Mannich reaction was attempted on the sulfonylated intermediate, the ortho-position of the methoxy group on the benzene ring was susceptible to substitution. This lack of selectivity led to the formation of difficult-to-remove N-pyrrolidine methyl substituted impurities, drastically compromising the purity of the final API. Furthermore, the workup procedures were notoriously cumbersome, requiring the reaction mixture to be poured into large volumes of water, followed by repeated pH adjustments using sodium hydroxide and hydrochloric acid. These steps necessitated multiple suction filtrations and generated substantial wastewater, creating both economic and environmental burdens for manufacturers aiming for cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
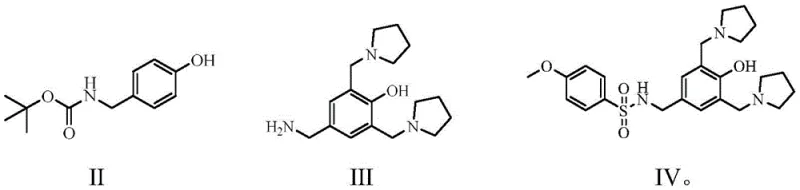
The innovative strategy presented in CN112110879A fundamentally alters the synthetic logic by prioritizing the Mannich reaction on a Boc-protected phenol derivative (Formula II). By conducting the Mannich reaction first, the process exploits the powerful ortho-directing effect of the hydroxyl group on a simplified benzene ring structure. This ensures that substitution occurs specifically at the ortho-positions relative to the hydroxyl group, effectively blocking side reactions at other sites. Following this highly selective functionalization, the amino protecting group is removed, and the resulting amine (Formula III) undergoes sulfonylation. This reversal of steps eliminates the problematic ortho-methoxy substitution entirely. The result is a cleaner reaction profile with significantly reduced impurity generation and a streamlined isolation process that avoids the excessive pH swinging and solvent usage of the past.
Mechanistic Insights into Regioselective Mannich Functionalization
The core chemical advantage of this process lies in the electronic activation of the aromatic ring during the initial Mannich step. In the starting material, tert-butyl 4-hydroxybenzylcarbamate (Formula II), the phenolic hydroxyl group is a potent activator for electrophilic aromatic substitution. When reacted with paraformaldehyde and pyrrolidine, the resulting iminium ion species attacks the positions ortho to the hydroxyl group with high fidelity. This is mechanistically superior to attacking a ring that already bears a bulky sulfonamide group, which can sterically hinder access and electronically deactivate the ring. The use of ethanol as a solvent under reflux conditions (82-86°C) facilitates this transformation efficiently, achieving yields of approximately 89% for the intermediate amine (Formula III) after deprotection. This high conversion rate is critical for maintaining throughput in a high-purity pharmaceutical intermediates production line.
Furthermore, the deprotection and subsequent sulfonylation steps are engineered to minimize degradation. The removal of the Boc group is achieved under mild acidic conditions (pH 2-4) followed by basification, avoiding harsh reagents that could damage the sensitive pyrrolidine rings. The final coupling with 4-methoxybenzenesulfonyl chloride is performed in DMF with triethylamine at controlled temperatures (0-30°C). This careful thermal management prevents exothermic runaways and ensures the formation of the sulfonamide bond without generating sulfonic acid byproducts. The cumulative effect of these mechanistic controls is a final free base yield of 72-75%, providing a robust foundation for the final salt formation step.
How to Synthesize Sulcardine Free Base Efficiently
The synthesis of Sulcardine free base via this patented route involves a logical progression of protection, functionalization, and coupling. The process begins with the condensation of a protected aminophenol with formaldehyde and pyrrolidine, followed by acid-mediated deprotection to reveal the reactive amine. This amine is then coupled with the sulfonyl chloride to form the core scaffold. The detailed operational parameters, including specific molar ratios and temperature controls, are essential for replicating the high yields reported in the patent. For process chemists looking to implement this technology, the following guide outlines the standardized workflow derived from the experimental examples.
- Perform a Mannich reaction on tert-butyl 4-hydroxybenzylcarbamate (Formula II) using paraformaldehyde and pyrrolidine in ethanol under reflux to generate the bis-pyrrolidinomethyl intermediate.
- Execute amino deprotection by treating the intermediate with hydrochloric acid followed by basification with ammonia water to isolate 4-hydroxy-3,5-bis(1-pyrrolidinomethyl)benzylamine (Formula III).
- React Formula III with 4-methoxybenzenesulfonyl chloride in DMF using triethylamine as a base to yield Sulcardine free base (Formula IV), followed by salt formation with sulfuric acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage is the drastic simplification of the downstream processing. By eliminating the need for multiple pH adjustments and the associated solid-liquid separations found in legacy methods, the new process reduces labor intensity and equipment occupancy time. This operational efficiency translates directly into lower manufacturing overheads. Additionally, the avoidance of chloroform and diethyl ether—solvents that are increasingly restricted due to environmental and safety regulations—removes the need for specialized containment and disposal protocols. This shift not only lowers compliance costs but also mitigates the risk of supply disruptions caused by solvent availability issues, ensuring a more resilient supply chain for reducing lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The improved regioselectivity of the Mannich reaction significantly reduces the formation of structural impurities that are costly to remove via chromatography or repeated recrystallization. By preventing these impurities at the source, the process minimizes material loss during purification, thereby enhancing the overall mass balance. The higher yields reported (50-55% total vs. lower historical benchmarks) mean that less raw material is required to produce the same amount of API, driving down the cost of goods sold (COGS). Furthermore, the use of common solvents like ethanol and DMF instead of specialized ethers allows for more efficient solvent recovery and recycling, contributing to substantial cost savings in large-scale production.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials, such as 4-methoxybenzenesulfonyl chloride and Boc-protected amines, ensures a steady input stream for manufacturing. The robustness of the reaction conditions, which tolerate slight variations in temperature and stoichiometry without catastrophic failure, makes the process highly reproducible across different batches and manufacturing sites. This consistency is vital for maintaining continuous supply to downstream drug product manufacturers. The simplified workup also reduces the turnaround time between batches, allowing facilities to increase their annual production capacity without significant capital investment in new infrastructure.
- Scalability and Environmental Compliance: The elimination of chloroform addresses a major environmental, health, and safety (EHS) concern, aligning the manufacturing process with modern green chemistry principles. This makes the facility more attractive to auditors and regulatory bodies, smoothing the path for regulatory filings. The reduced wastewater generation, stemming from the avoidance of excessive aqueous washes and pH adjustments, lowers the burden on effluent treatment plants. These factors combined make the process highly scalable, capable of moving seamlessly from kilogram-scale development to multi-ton commercial production while maintaining strict environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential partners and licensees. Understanding these details is key to evaluating the feasibility of adopting this technology for your specific production needs.
Q: How does the new synthesis route improve impurity profiles compared to conventional methods?
A: By performing the Mannich reaction prior to sulfonylation, the new route leverages the strong ortho-directing effect of the hydroxyl group on a single benzene ring. This prevents the unwanted ortho-substitution of the methoxy group that plagues traditional routes, significantly reducing N-pyrrolidine methyl substituted impurities.
Q: What are the yield improvements observed in the patented process?
A: The novel method achieves a total yield of 50-55% for Sulcardine sulfate. Specifically, the yield for the Sulcardine free base step reaches 72-75%, with the intermediate amine synthesis yielding approximately 89%, representing a substantial efficiency gain over previous laboratory-scale processes.
Q: Does this process eliminate hazardous solvents like chloroform?
A: Yes. Unlike prior art which utilized chloroform for extraction and diethyl ether for refinement, this optimized protocol relies on ethanol, dichloromethane, and DMF. This shift aligns better with modern environmental compliance standards and simplifies solvent recovery in bulk drug workshops.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulcardine Sulfate Supplier
The technological advancements detailed in CN112110879A represent a significant leap forward in the manufacture of anti-arrhythmic agents, yet realizing this potential requires a partner with deep technical expertise and industrial capacity. NINGBO INNO PHARMCHEM stands ready to bridge the gap between patent theory and commercial reality. As a premier CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific solvent systems and thermal requirements of this synthesis, ensuring that every batch meets stringent purity specifications. With our rigorous QC labs and commitment to quality assurance, we guarantee that the Sulcardine intermediates supplied meet the highest global standards for safety and efficacy.
We invite pharmaceutical companies and research institutions to collaborate with us to further optimize this promising route. Whether you require custom synthesis services or bulk supply of the free base or sulfate salt, our team is prepared to deliver. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are eager to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your drug development pipeline and accelerate your time to market.
