Advanced Manufacturing of 7-Quinolyl-3,5-Dihydroxyhept-6-enoate for Statin Production
Advanced Manufacturing of 7-Quinolyl-3,5-Dihydroxyhept-6-enoate for Statin Production
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for critical active pharmaceutical ingredient (API) intermediates, particularly those serving the cardiovascular therapeutic sector. Patent CN1241914C introduces a significant technological advancement in the preparation of 7-quinolinyl-3,5-dihydroxyhept-6-enoate, a pivotal building block for HMG-CoA reductase inhibitors commonly known as statins. This patent discloses a highly efficient method that addresses longstanding challenges in the selective reduction of beta-hydroxy ketones to 1,3-diols. By optimizing the post-reduction workup procedure, the invention enables the production of the target intermediate in high yield and exceptional purity, which is paramount for downstream drug synthesis. The methodology described offers a compelling alternative to prior art, specifically targeting the inefficiencies associated with boron byproduct removal. For global supply chain stakeholders, this represents a viable pathway to secure reliable supplies of high-quality statin precursors while mitigating environmental and operational risks inherent in older chemical processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,3-diol structures from beta-hydroxy ketones has relied heavily on sodium borohydride reductions mediated by boron compounds to ensure stereoselectivity and chemoselectivity. However, the legacy methods documented in earlier patents, such as Japanese Patent Laid-Open Publication No. Hei 1-279866 and European Patent No. 0304063B1, suffer from significant downstream processing drawbacks. A primary bottleneck in these conventional approaches is the tedious and often hazardous removal of boron complexes formed during the reaction. Traditional protocols typically necessitate the use of excessive amounts of hydrogen peroxide in homogeneous solutions or require energy-intensive azeotropic distillation with large volumes of methanol to decompose the boron species. These steps not only generate substantial chemical waste, posing environmental compliance challenges, but also complicate the isolation of the final product. In many cases, failure to completely remove these boron ligands results in the product remaining as an intractable oil, rendering it unsuitable for further purification or crystallization, thereby drastically reducing overall process efficiency and throughput.
The Novel Approach
The innovative process outlined in CN1241914C circumvents these historical limitations through a refined biphasic oxidative workup strategy. Instead of relying on massive excesses of oxidants or solvent-heavy distillation steps, this method employs a two-phase system comprising toluene and water. Following the initial reduction of the ketone precursor using sodium borohydride in the presence of a dialkylmethoxyborane, the reaction mixture is treated with hydrogen peroxide in the presence of an inorganic base such as sodium carbonate or potassium carbonate. This specific combination facilitates the efficient cleavage of the carbon-boron bonds under mild conditions. The use of a biphasic system allows for the effective partitioning of the organic product into the toluene layer while boron byproducts are sequestered in the aqueous phase. This strategic modification not only streamlines the purification workflow but also ensures that the target 7-quinolinyl-3,5-dihydroxyhept-6-enoate can be isolated as a high-purity crystalline solid. The ability to obtain a crystalline product directly from the workup is a transformative improvement, eliminating the need for complex chromatographic separations and ensuring consistent quality for industrial applications.
Mechanistic Insights into Boron-Mediated Selective Reduction
The core chemical transformation driving this synthesis is the highly selective reduction of a beta-hydroxy ketone to a syn-1,3-diol, a structural motif critical for the biological activity of HMG-CoA reductase inhibitors. The mechanism initiates with the coordination of the boron compound, specifically diethylmethoxyborane or similar dialkylboranes, to the hydroxyl group of the substrate. This coordination activates the adjacent carbonyl group and directs the delivery of the hydride from sodium borohydride to a specific face of the ketone, ensuring the desired stereochemical outcome. The formation of a cyclic borate ester intermediate is key to this selectivity, locking the conformation of the molecule during the reduction event. This precise control over stereochemistry is essential because the biological efficacy of the final statin drug is strictly dependent on the spatial arrangement of the hydroxyl groups on the heptenoate side chain. Any deviation in stereochemistry would result in inactive or potentially harmful impurities, making the fidelity of this reduction step non-negotiable for pharmaceutical grade manufacturing.
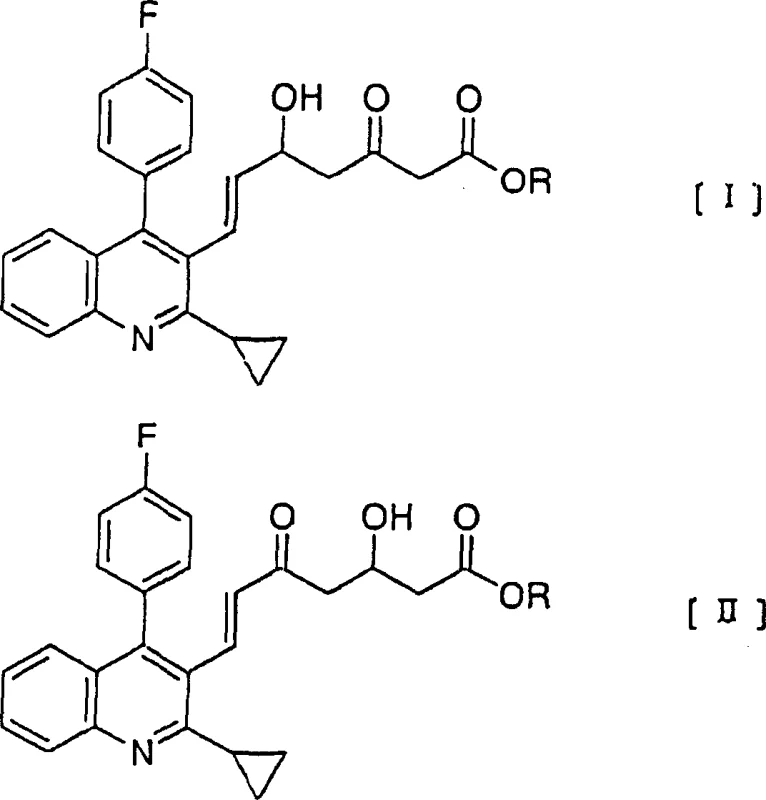
Following the reduction, the resulting organoboron intermediate must be converted back to the free diol. In the absence of the patented oxidative workup, the boron ligand remains tightly bound to the oxygen atoms, creating a stable complex that resists standard aqueous extraction. The innovation lies in the use of alkaline hydrogen peroxide within a toluene-water biphasic medium. The base activates the hydrogen peroxide, generating a nucleophilic perhydroxyl anion that attacks the electron-deficient boron center. This triggers a migration of the alkyl group from boron to oxygen, effectively hydrolyzing the borate ester and releasing the free hydroxyl groups of the diol product. The toluene solvent plays a dual role here: it solubilizes the organic diol product, protecting it from potential over-oxidation, and it forms a distinct phase that simplifies the separation of inorganic boron salts. This mechanistic elegance ensures that the final product, as depicted in the reaction scheme, is obtained with minimal impurities and maximum structural integrity, ready for subsequent coupling reactions in the statin synthesis pipeline.
How to Synthesize 7-Quinolyl-3,5-Dihydroxyhept-6-enoate Efficiently
The practical execution of this synthesis requires careful attention to temperature control and reagent stoichiometry to maximize yield and safety. The process begins by dissolving the beta-hydroxy ketone precursor, such as the ethyl ester derivative, in a mixture of tetrahydrofuran (THF) and methanol. This solution must be cooled to cryogenic temperatures, typically ranging from -90°C to -60°C, before the addition of the reducing agents. A separate suspension of sodium borohydride and the boron mediator in THF is prepared and added dropwise to maintain the low temperature, which is critical for controlling the exothermic nature of the reduction and preserving stereoselectivity. Once the reduction is complete, the reaction is quenched, and the solvents are exchanged for toluene to prepare for the oxidative workup. The detailed standardized synthesis steps, including specific molar ratios and agitation speeds for optimal phase transfer, are provided in the guide below.
- Dissolve the beta-hydroxy ketone precursor in a mixed solvent of THF and methanol, then cool the solution to cryogenic temperatures between -90°C and -60°C.
- Add a suspension of sodium borohydride and a boron compound such as diethylmethoxyborane in THF to the cooled substrate solution to effect selective reduction.
- Treat the reaction mixture in a toluene-water biphasic system with hydrogen peroxide and an inorganic base like sodium carbonate to remove boron ligands and isolate the crystalline diol product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN1241914C offers tangible benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the isolation process, which directly translates to reduced operational costs and shorter production cycles. By enabling the crystallization of the product directly from the reaction workup, the need for resource-intensive purification techniques such as column chromatography or multiple recrystallizations from diverse solvent systems is eliminated. This streamlining of the downstream process reduces the consumption of solvents and consumables, leading to significant cost reduction in pharmaceutical intermediate manufacturing. Furthermore, the ability to produce a crystalline solid enhances the stability and shelf-life of the intermediate, facilitating easier storage and transportation logistics without the degradation risks associated with oily residues.
- Cost Reduction in Manufacturing: The elimination of excessive methanol usage for azeotropic distillation and the reduction in hydrogen peroxide consumption significantly lower the raw material costs associated with the workup phase. Traditional methods often require vast quantities of solvents to drive the equilibrium of boron removal, whereas this biphasic method achieves complete conversion with stoichiometric efficiency. Additionally, the recovery and recycling of the toluene solvent are more straightforward compared to mixed alcohol systems, further enhancing the economic viability of the process. The reduction in waste disposal costs, particularly regarding boron-containing effluents, adds another layer of financial benefit, making this route highly attractive for large-scale production budgets.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures a more reliable supply of high-purity statin intermediates. Conventional methods that yield oily products are prone to batch-to-batch variability and purification failures, which can cause unpredictable delays in the supply chain. In contrast, the crystallization capability of this novel method provides a clear pass/fail criterion for quality control, ensuring that every batch meets stringent specifications before leaving the facility. This consistency reduces the risk of supply disruptions for downstream API manufacturers, allowing for more accurate demand forecasting and inventory management. The use of commercially available and stable reagents like sodium borohydride and diethylmethoxyborane further secures the supply chain against raw material shortages.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is markedly superior to prior art. The biphasic system minimizes the volume of hazardous waste generated, aligning with increasingly strict global environmental regulations. The avoidance of large-scale methanol distillation reduces the energy footprint of the manufacturing process, contributing to sustainability goals. Moreover, the controlled use of hydrogen peroxide in a two-phase system mitigates the risks of thermal runaway associated with bulk oxidant handling in single-phase reactors. This safety profile makes the process easier to scale from pilot plant to commercial tonnage, ensuring that supply can be ramped up to meet market demand without compromising safety standards or regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and technical disclosures within CN1241914C, providing clarity on the operational parameters and benefits of the technology. Understanding these nuances is crucial for technical teams evaluating the feasibility of integrating this intermediate into their existing production lines. The responses highlight the specific advantages of the biphasic workup and the critical nature of the boron removal step in achieving pharmaceutical-grade purity.
Q: Why is the biphasic hydrogen peroxide treatment critical in this synthesis?
A: The biphasic treatment with hydrogen peroxide and an inorganic base is essential for cleaving the boron-oxygen bonds formed during reduction. Without this step, the product remains complexed with boron ligands, preventing crystallization and resulting in an oily residue that is difficult to purify.
Q: What are the advantages of this method over conventional azeotropic removal techniques?
A: Conventional methods often require large volumes of methanol for azeotropic distillation to remove boron compounds, which is energy-intensive and inefficient. This patented method utilizes a direct oxidative workup in a toluene-water system, significantly simplifying the process and reducing solvent consumption.
Q: Can this process be scaled for industrial production of statin intermediates?
A: Yes, the process is designed for industrial applicability. It avoids the environmental hazards associated with excessive hydrogen peroxide usage in single-phase systems and allows for the isolation of the product as high-purity crystals, which is a key requirement for scalable pharmaceutical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Quinolyl-3,5-Dihydroxyhept-6-enoate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global pharmaceutical supply chain. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in CN1241914C can be translated into reliable industrial reality. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our facility is equipped to handle the cryogenic conditions and biphasic processing required for this synthesis, guaranteeing a consistent supply of this vital statin precursor to support your drug development and manufacturing needs.
We invite you to collaborate with us to optimize your supply chain for HMG-CoA reductase inhibitor production. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our implementation of this patented technology can reduce your overall cost of goods. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to ensure the uninterrupted availability of high-purity pharmaceutical intermediates, driving efficiency and success in your cardiovascular therapeutic programs.
