Advanced Synthesis of Asymmetric Linear Seven-Membered Fused Thiophene Isomers for High-Mobility Organic Electronics
Introduction to Next-Generation Organic Semiconductor Intermediates
The rapid evolution of organic field-effect transistors (OFETs) and organic light-emitting diodes (OLEDs) demands materials with superior charge carrier mobility and optimized solid-state packing. Patent CN111100142B introduces a groundbreaking class of asymmetric linear seven-membered fused thiophene isomers that address the limitations of traditional symmetric architectures. By strategically breaking molecular symmetry, this innovation expands the intermolecular conjugation system and enhances π-π stacking interactions, which are critical for facilitating efficient charge transport in the solid state. This technical breakthrough provides a robust pathway for developing high-performance organic semiconductors, offering a reliable electronic chemical supplier with access to next-generation molecular scaffolds that promise significantly improved device performance metrics.
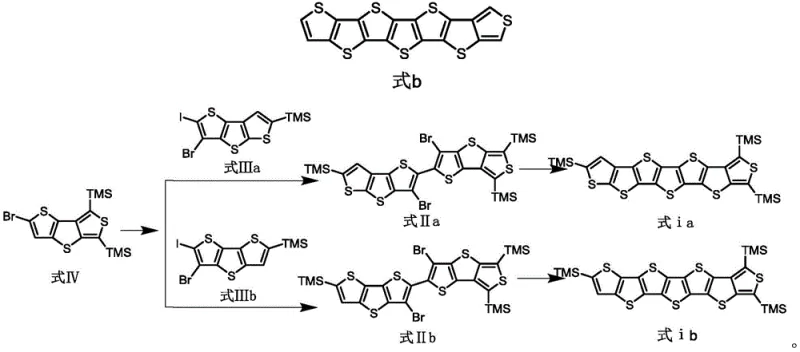
Historically, linear fused thiophenes have been synthesized with symmetrical structures, which often limit the tunability of their electronic properties and packing motifs. The disclosed technology overcomes these barriers by providing a versatile synthetic route to asymmetric isomers, specifically formulas Ia and Ib, where R can be hydrogen or a trimethylsilyl group. This structural diversity allows materials scientists to fine-tune energy levels and solubility profiles without compromising the extended conjugation length essential for high mobility. For R&D directors seeking to push the boundaries of organic electronics, this patent represents a pivotal shift towards more sophisticated molecular designs that can be reliably manufactured at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for linear fused thiophenes have predominantly focused on symmetric molecules, such as those depicted in formulas 1 through 4 of the background art. These symmetric structures, while easier to conceptualize, often suffer from restricted conformational flexibility and less optimal packing arrangements in the solid state, leading to suboptimal carrier mobility values typically ranging around 0.045 to 0.06 cm²V⁻¹s⁻¹ for shorter oligomers. Furthermore, conventional methods often struggle with regioselectivity when attempting to functionalize specific positions on the thiophene core, leading to complex mixtures of isomers that are difficult and costly to separate. This lack of precision not only reduces overall yield but also introduces impurities that can act as charge traps, severely degrading the performance of the final electronic device.
The Novel Approach
The novel approach detailed in this patent utilizes a strategic combination of bromine migration and Negishi coupling to construct the asymmetric carbon skeleton with high precision. By employing a bromine dance rearrangement followed by transmetallation to zinc, the synthesis ensures that the coupling occurs exclusively at the desired position, effectively bypassing the regioselectivity issues plaguing direct lithiation methods. This methodology allows for the modular assembly of the seven-membered fused ring system, enabling the incorporation of different end-groups or substituents to further tune the material properties. The result is a streamlined process that delivers high-purity asymmetric thiophenes with expanded conjugation systems, directly addressing the need for materials with enhanced π-π stacking and superior charge transport capabilities.
Mechanistic Insights into Bromine Migration and Negishi Coupling
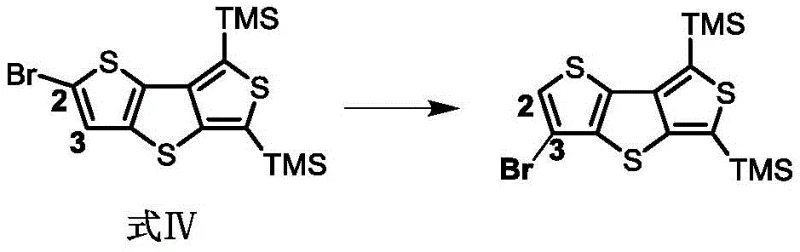
The cornerstone of this synthesis is the controlled bromine migration reaction, which serves as a powerful tool for regiocontrol in heterocyclic chemistry. In this process, the starting material, Formula IV, is treated with lithium diisopropylamide (LDA) at cryogenic temperatures, typically between -78°C and 0°C. This induces a halogen dance rearrangement where the bromine atom migrates from the 2-position to the 3-position, generating a carbanion at the 2-position. This specific intermediate is then quenched with zinc chloride to form a stable organozinc reagent, which is far less reactive and more tolerant of functional groups than the corresponding organolithium species. This mechanistic nuance is crucial for preventing side reactions and ensuring that the subsequent cross-coupling proceeds with high fidelity.
Following the formation of the organozinc species, the synthesis proceeds via a Palladium-catalyzed Negishi coupling with a brominated thiophene precursor (Formula IIIa or IIIb). The use of Pd(PPh3)4 as the catalyst facilitates the oxidative addition and reductive elimination cycles necessary to forge the carbon-carbon bond between the thiophene units. This step is critical for extending the conjugated backbone while maintaining the asymmetry of the molecule. Subsequent steps involve a bromine-lithium exchange followed by cyclization using diphenylsulfonyl sulfide, which closes the final ring to form the seven-membered fused system. The precise control over each mechanistic step ensures that the final product, whether Formula Ia or Ib, possesses the exact structural configuration required for optimal electronic performance.
How to Synthesize Asymmetric Linear Seven-Membered Fused Thiophene Efficiently
The synthesis of these advanced semiconductor intermediates requires strict adherence to anhydrous conditions and precise temperature control to maximize yield and purity. The process begins with the preparation of the organozinc reagent via bromine migration, followed by the palladium-catalyzed coupling to build the molecular backbone. The final stages involve ring closure and deprotection to reveal the active conjugated system. Detailed operational parameters, including specific molar ratios and reaction times, are critical for reproducibility and are essential for any laboratory aiming to replicate these high-value materials. The detailed standardized synthesis steps are outlined in the guide below.
- Perform bromine migration on the starting thiophene derivative using LDA at low temperatures followed by ZnCl2 quenching to generate an organozinc reagent.
- Execute a Palladium-catalyzed Negishi coupling reaction between the organozinc intermediate and a brominated thiophene precursor to extend the conjugated system.
- Conduct bromine-lithium exchange followed by cyclization with diphenylsulfonyl sulfide to close the ring, and finally remove TMS protecting groups.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthetic route offers substantial advantages over traditional methods used for producing organic semiconductor materials. The reliance on well-established reactions such as Negishi coupling and standard lithiation techniques means that the process can be easily scaled using existing infrastructure without the need for specialized or exotic equipment. This compatibility with standard chemical manufacturing protocols significantly reduces the barrier to entry for commercial production, ensuring a more stable and continuous supply of these critical electronic chemicals. Furthermore, the high regioselectivity of the bromine migration step minimizes the formation of difficult-to-remove isomeric impurities, thereby simplifying downstream purification processes and reducing overall production costs.
- Cost Reduction in Manufacturing: The synthetic strategy eliminates the need for complex, multi-step protection and deprotection sequences often required in symmetric syntheses, leading to a more atom-economical process. By utilizing commercially available reagents like LDA, ZnCl2, and Pd(PPh3)4, the method avoids the high costs associated with proprietary catalysts or rare earth metals. The high yield reported in the examples suggests that raw material consumption is optimized, which translates directly into lower cost of goods sold (COGS) for the final high-purity organic semiconductor.
- Enhanced Supply Chain Reliability: The starting materials and reagents utilized in this process are commodity chemicals with robust global supply chains, mitigating the risk of shortages that often plague specialty synthesis. The modular nature of the Negishi coupling allows for flexibility in sourcing different thiophene precursors, providing procurement managers with multiple options to secure raw materials. This flexibility ensures that production schedules can be maintained even if specific suppliers face disruptions, guaranteeing consistent delivery timelines for downstream electronics manufacturers.
- Scalability and Environmental Compliance: The reaction conditions, primarily involving standard organic solvents like THF and ether, are well-understood in terms of waste management and solvent recovery, facilitating compliance with environmental regulations. The process avoids the generation of heavy metal waste streams associated with some alternative coupling methods, as the palladium catalyst is used in relatively low loading and can be recovered. This environmental profile makes the technology highly attractive for large-scale commercial scale-up of complex electronic chemicals, aligning with modern green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these asymmetric thiophene isomers. Understanding these details is vital for stakeholders evaluating the feasibility of integrating these materials into their product pipelines. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation.
Q: What is the primary advantage of asymmetric over symmetric seven-membered fused thiophenes?
A: Asymmetric structures expand the intermolecular conjugation system and enhance π-π stacking interactions, which significantly improves carrier mobility compared to traditional symmetric analogs.
Q: How does the bromine migration step improve the synthesis efficiency?
A: The bromine migration allows for the precise positioning of the reactive site for subsequent organozinc formation, enabling regioselective coupling that is difficult to achieve with direct lithiation methods.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the method utilizes standard organic transformations like Negishi coupling and uses commercially available reagents, making it highly adaptable for commercial scale-up of complex electronic chemicals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Asymmetric Linear Seven-Membered Fused Thiophene Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis for advanced electronic materials, leveraging deep expertise in organometallic chemistry to deliver high-value intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of asymmetric thiophene meets the exacting standards required for high-performance organic electronics.
We invite potential partners to engage with our technical procurement team to discuss how this patented technology can be adapted to your specific material requirements. By requesting a Customized Cost-Saving Analysis, you can gain insights into how our optimized manufacturing processes can reduce your overall material costs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of technical excellence and reliability.
