Advanced Manufacturing of Erlotinib Hydrochloride: A Novel Safe and Scalable Synthetic Route
The pharmaceutical landscape for oncology treatments continues to evolve, with Erlotinib Hydrochloride standing as a critical epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor for treating non-small cell lung cancer. As detailed in patent CN101735157B, a groundbreaking preparation method has been established that fundamentally reengineers the synthetic pathway to enhance safety, purity, and industrial feasibility. This technical insight report analyzes the novel four-step synthesis starting from 6,7-dimethoxyquinazoline-4-one, which circumvents the hazardous nitration and hydrogenation steps prevalent in legacy manufacturing processes. For R&D Directors and Supply Chain Heads, understanding this shift is vital, as it represents a transition from high-risk batch processing to a controlled, solid-intermediate driven workflow. The patent explicitly outlines a route that avoids deep cooling and high-temperature extremes, ensuring that the production of this high-purity API intermediate remains both economically viable and environmentally compliant. By adopting this methodology, manufacturers can secure a more reliable Erlotinib Hydrochloride supplier chain that is resilient to the regulatory and safety pressures of modern chemical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Erlotinib Hydrochloride has relied heavily on routes involving nitration reactions followed by catalytic hydrogenation, as illustrated in prior art patents such as US5747498. These conventional methods present severe engineering bottlenecks, primarily due to the use of mixed acid nitration which is highly exothermic and difficult to control on a large scale. The risk of polynitration and the formation of dark-brown oily by-products significantly complicates purification, often requiring specialized hydrogenation workshops and noble metal catalysts that drive up capital expenditure. Furthermore, the intermediates in these traditional routes are frequently liquids, necessitating complex extraction and distillation steps that increase solvent consumption and waste generation. The reliance on unstable halogenated reagents for side-chain introduction further exacerbates impurity profiles, leading to lower overall yields and inconsistent quality. For procurement managers, these factors translate into volatile pricing and supply chain fragility, as the safety hazards associated with nitration can lead to unplanned production shutdowns and regulatory scrutiny.
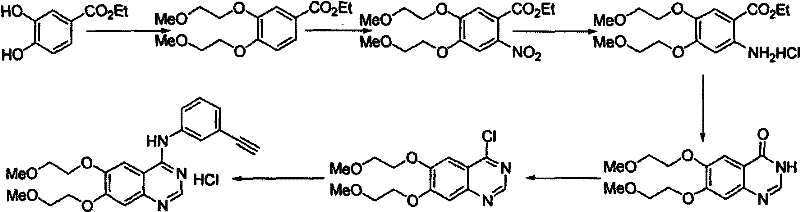
The Novel Approach
In stark contrast, the novel approach disclosed in CN101735157B introduces a paradigm shift by utilizing 6,7-dimethoxyquinazoline-4-one as the starting material, thereby completely bypassing the dangerous nitration step. This innovative pathway leverages a direct chlorination followed by amination, demethylation, and a highly specific side-chain introduction using tosic acid ethylene glycol monomethyl ether ester. The strategic advantage lies in the physical state of the intermediates; each step generates a solid product that can be isolated through simple filtration and crystallization, drastically simplifying post-processing. This solid-phase progression not only enhances purity by allowing for the physical removal of impurities at each stage but also eliminates the need for complex liquid-liquid separations. The use of stable tosylate reagents for the final alkylation ensures that side reactions on the quinazoline ring are minimized, resulting in a cleaner reaction profile. This method offers substantial cost savings by removing the need for expensive noble metal catalysts and high-pressure hydrogenation equipment, making it an ideal solution for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into the Four-Step Synthetic Pathway
The core of this technological breakthrough lies in the precise control of reaction mechanisms across four distinct stages, beginning with the chlorination of 6,7-dimethoxyquinazoline-4-one using reagents such as oxalyl chloride. This step is conducted under mild reflux conditions without the need for additional solvents, generating 4-chloro-6,7-dimethoxyquinazoline with high efficiency. The subsequent amination reaction with 3-aminophenylacetylene is performed in absolute ethanol, where the reflux temperature accelerates the reaction rate while maintaining a homogeneous system that favors the formation of the amine hydrochloride salt. A critical mechanistic advantage is observed in the demethylation step, where aluminum chloride is employed to selectively remove methyl groups at the 6 and 7 positions. This Lewis acid-mediated cleavage is highly controllable, preventing the degradation of the sensitive ethynyl group on the phenyl ring, which is a common failure point in less optimized routes. The final alkylation utilizes a tosylate ester under alkaline conditions, which reacts specifically with the phenolic hydroxyls generated in the previous step, ensuring that the methoxyethyl side chain is introduced with high regioselectivity and minimal by-product formation.
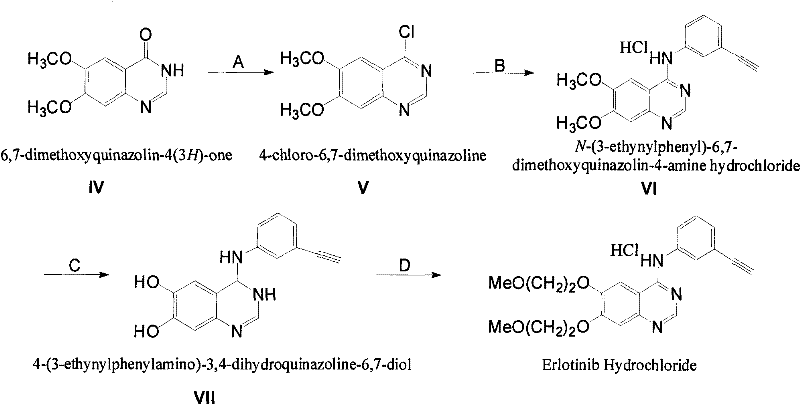
Impurity control is inherently built into this synthetic design through the physical properties of the intermediates and the specificity of the reagents used. Unlike liquid intermediates in traditional routes that trap impurities, the solid intermediates in this novel pathway allow for effective purification via washing and recrystallization, significantly reducing the burden on final chromatographic purification. The use of tosic acid ethylene glycol monomethyl ether ester instead of halogenated ethers eliminates the risk of dihalo-ethane impurities and reduces the pungency and instability associated with volatile alkylating agents. Furthermore, the reaction conditions are tuned to avoid extreme pH swings or temperatures that could degrade the quinazoline core, ensuring that the final API meets stringent purity specifications. For R&D teams, this means a more robust process window where minor deviations in temperature or stoichiometry do not lead to catastrophic batch failures. The mechanistic stability of the quinazoline ring throughout the sequence ensures that the final product possesses a consistent impurity profile, which is crucial for regulatory approval and commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Erlotinib Hydrochloride Efficiently
The implementation of this synthesis route requires a structured approach to maximize yield and safety, beginning with the preparation of the chloro-intermediate followed by sequential functionalization. The process is designed to be operationally simple, utilizing common solvents like ethanol and acetonitrile while avoiding specialized high-pressure equipment. Detailed standard operating procedures for each step, including specific molar ratios and temperature controls, are essential to replicate the high yields reported in the patent data. The following guide outlines the critical operational parameters required to achieve industrial-grade quality.
- Chlorinate 6,7-dimethoxyquinazoline-4-one using oxalyl chloride to form 4-chloro-6,7-dimethoxyquinazoline.
- React the chloro-intermediate with 3-aminophenylacetylene to generate the amine hydrochloride salt.
- Perform demethylation using aluminum chloride to remove methyl groups at positions 6 and 7.
- Introduce the methoxyethyl side chain using tosic acid ethylene glycol monomethyl ether ester under alkaline conditions.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this novel synthesis route offers transformative benefits for procurement and supply chain management by fundamentally altering the cost and risk structure of production. The elimination of nitration and hydrogenation steps removes significant safety liabilities, reducing insurance costs and the need for specialized hazardous material handling infrastructure. The reliance on solid intermediates that can be purified by filtration rather than distillation leads to a drastic reduction in solvent consumption and energy usage, directly contributing to lower manufacturing overheads. Additionally, the use of stable, commercially available reagents like tosic acid esters ensures a reliable supply of raw materials, mitigating the risk of shortages associated with custom-synthesized alkylating agents. These factors combine to create a supply chain that is not only more cost-effective but also more resilient to market fluctuations and regulatory changes.
- Cost Reduction in Manufacturing: The removal of noble metal catalysts and high-pressure hydrogenation equipment results in significant capital expenditure savings and lower operational costs. By avoiding the complex purification steps required for oily liquid intermediates, the process reduces solvent waste and energy consumption, leading to substantial cost savings in the overall production budget. The high yield and purity of the solid intermediates minimize material loss, ensuring that raw material costs are optimized throughout the synthesis.
- Enhanced Supply Chain Reliability: The use of stable reagents and mild reaction conditions reduces the risk of production delays caused by safety incidents or equipment failures. The availability of starting materials like 6,7-dimethoxyquinazoline-4-one from multiple suppliers ensures a continuous flow of resources, preventing bottlenecks in the manufacturing schedule. This reliability is crucial for maintaining consistent delivery timelines to downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on simple unit operations like filtration and crystallization, which are easily adapted from laboratory to plant scale. The reduction in hazardous waste and solvent usage aligns with strict environmental regulations, facilitating easier permitting and compliance. This environmental friendliness enhances the long-term sustainability of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Erlotinib synthesis route. These answers are derived directly from the patent specifications and are designed to clarify the operational advantages and safety improvements over traditional methods. Understanding these details is essential for stakeholders evaluating the feasibility of adopting this technology for commercial production.
Q: How does this new route improve safety compared to traditional nitration methods?
A: The novel route eliminates the use of mixed acid nitration and catalytic hydrogenation, which are prone to exothermic runaway reactions and require high-pressure equipment. Instead, it utilizes mild chlorination and solid-phase intermediate isolation, significantly reducing operational hazards.
Q: What are the purity advantages of using tosic acid ethylene glycol monomethyl ether ester?
A: Unlike unstable halogenated ethyl methyl ethers used in prior art, the tosic acid ester is chemically stable and generates fewer by-products. This specificity prevents side reactions on the quinazoline ring, leading to higher crude purity and simplified downstream purification.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process is designed for industrial scalability. All intermediates are solids that can be easily purified by filtration and crystallization, avoiding difficult liquid-liquid separations. The reaction conditions are mild, avoiding extreme temperatures or deep cooling, which facilitates cost reduction in pharmaceutical intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Erlotinib Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global pharmaceutical market. Our CDMO capabilities are specifically engineered to support the scale-up of complex pathways like the one described in CN101735157B, ensuring that technical innovations are successfully translated into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our rigorous QC labs to guarantee stringent purity specifications for every batch. Our team of expert chemists is dedicated to optimizing these processes further, ensuring that our clients receive a high-purity API intermediate that meets the highest international standards.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis to evaluate how this novel route can impact your specific production economics. We encourage you to reach out for specific COA data and route feasibility assessments to ensure that your project moves forward with the highest level of confidence and technical support.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
