Scalable Synthesis of Penta-N-Ts-Kanamycin B for High-Purity Antibiotic Production
Introduction to Advanced Aminoglycoside Intermediate Manufacturing
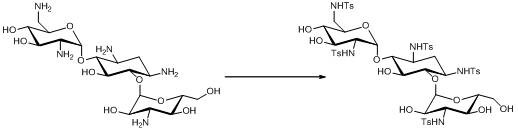
The pharmaceutical industry continuously seeks robust pathways for synthesizing complex antibiotic precursors, and patent CN111704636B presents a significant breakthrough in the preparation of 1,3,2’,6’,3’’-penta-N-p-toluenesulfonyl kanamycin B. This specific intermediate is critical for the production of Debekacin, a broad-spectrum semi-synthetic aminoglycoside antibiotic known for its synergistic effects and low cross-resistance. The disclosed technology addresses long-standing challenges in yield optimization and purity control by utilizing a sophisticated multi-solvent system that facilitates efficient N-tosylation. By shifting away from traditional binary solvent mixtures, this innovation enables reaction conditions that are not only milder but also inherently safer for large-scale operations. For R&D directors and procurement specialists, understanding the nuances of this protective group strategy is essential for securing a reliable supply chain of high-value antibiotic intermediates. The method demonstrates how precise solvent engineering can transform a standard protection step into a high-efficiency unit operation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of protected kanamycin derivatives has relied on binary solvent systems, typically involving water and dioxane, which impose severe operational constraints on manufacturing facilities. Conventional protocols often necessitate rigorous temperature control, frequently requiring ice-bath cooling to maintain the reaction mixture below 10°C to prevent side reactions and degradation. Furthermore, these older methods frequently suffer from suboptimal yields, often hovering around 72% to 83%, which translates to significant raw material waste and increased cost of goods sold. The post-processing in traditional routes is equally cumbersome, often involving energy-intensive concentration steps via rotary evaporation to remove bulk solvents before precipitation can occur. This concentration step not only increases utility costs but also poses thermal risks to the heat-sensitive aminoglycoside scaffold. Additionally, the resulting solids from conventional methods can be prone to agglomeration, complicating filtration and drying processes in large reactors.
The Novel Approach
The innovative methodology described in the patent data overcomes these bottlenecks by employing a ternary or quaternary solvent system comprising water, an alcohol, and a water-immiscible organic solvent such as dichloromethane or ethyl acetate. This multi-phase environment dramatically enhances the solubility compatibility between the highly polar kanamycin B and the organic p-toluenesulfonyl chloride reagent, facilitating a more homogeneous and efficient reaction kinetics. Crucially, the process operates effectively within a broad temperature window of 0°C to 30°C, eliminating the need for expensive and energy-draining cryogenic cooling infrastructure. The workup procedure is revolutionized by the direct addition of an inert anti-solvent like hexane or heptane, which induces immediate crystallization without prior concentration. This simplification results in superior solid properties, characterized by free-flowing crystals that do not cake or clump, thereby streamlining the isolation phase and ensuring consistent product quality suitable for downstream processing.
Mechanistic Insights into Multi-Solvent N-Tosylation
The core of this chemical transformation lies in the selective N-tosylation of the five amine functionalities present on the kanamycin B scaffold, a reaction that requires precise control over nucleophilicity and solvation effects. In the presence of a base such as sodium carbonate or potassium carbonate, the amine groups are deprotonated to form nucleophilic species that attack the sulfur atom of the p-toluenesulfonyl chloride. The unique contribution of the mixed solvent system is its ability to stabilize the transition states and manage the byproduct hydrochloric acid effectively. Water serves to dissolve the inorganic base and the sugar substrate, while the organic co-solvents ensure the sulfonyl chloride remains in solution and accessible for reaction. This balance prevents the formation of localized hot spots of acidity that could lead to glycosidic bond hydrolysis, a common degradation pathway in aminoglycoside chemistry. The result is a clean conversion to the penta-N-protected species with minimal formation of partially protected impurities or hydrolyzed byproducts.
Impurity control is further enhanced by the thermodynamic stability provided by the solvent matrix during the crystallization phase. When the inert solvent is introduced, the solubility product of the target penta-N-tosylated compound is exceeded, driving rapid nucleation. Because the reaction conversion is driven to completion (monitored by TLC or HPLC to ensure raw material depletion), the crystallizing solid is inherently enriched in the desired product. The specific choice of inert anti-solvents like methyl tert-butyl ether or heptane ensures that polar impurities and unreacted salts remain in the mother liquor rather than co-precipitating. This mechanism acts as an in-situ purification step, allowing the process to achieve purity levels exceeding 95% directly from the reactor filter cake. Such high purity at the intermediate stage reduces the burden on subsequent purification steps in the total synthesis of the final antibiotic API.
How to Synthesize 1,3,2’,6’,3’’-penta-N-p-toluenesulfonyl kanamycin B Efficiently
Implementing this synthesis route requires careful attention to the ratios of the multi-component solvent system and the sequential addition of reagents to maintain reaction homogeneity. The process begins with the dissolution of kanamycin B and the acid binding agent in the aqueous-alcoholic phase, followed by the introduction of the organic co-solvent to create the reaction medium. Once the temperature is stabilized within the 0-30°C range, the p-toluenesulfonyl chloride is added, and the mixture is agitated for a period ranging from 8 to 18 hours to ensure full conversion. The detailed standardized synthesis steps, including specific mass ratios and stirring speeds optimized for pilot and plant scales, are outlined in the structured guide below for technical teams to review.
- Dissolve Kanamycin B and an acid binding agent in a mixed solvent system comprising water, alcohol, and an organic solvent like dichloromethane.
- Add p-toluenesulfonyl chloride at a controlled temperature between 0°C and 30°C and stir for 8 to 18 hours to ensure complete reaction.
- Add an inert solvent such as hexane or heptane to induce crystallization, then filter and dry to obtain the high-purity intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented methodology offers substantial strategic benefits regarding cost structure and operational reliability. The shift to a milder temperature profile significantly reduces the energy load on facility HVAC and chilling systems, which is a major contributor to overhead costs in fine chemical manufacturing. Moreover, the elimination of the solvent concentration step prior to isolation drastically cuts down on utility consumption related to steam and vacuum generation, while simultaneously shortening the overall batch cycle time. These efficiencies translate into a more competitive cost position for the intermediate, allowing downstream API manufacturers to optimize their margins without compromising on quality standards. The robustness of the crystallization process also minimizes the risk of batch failures due to oiling out or poor solid formation, ensuring a predictable and steady output of material.
- Cost Reduction in Manufacturing: The process achieves significant cost savings by removing the need for energy-intensive distillation or rotary evaporation steps typically required to concentrate reaction mixtures. By utilizing a direct crash-out crystallization method with inexpensive inert solvents, the operational expenditure associated with solvent recovery and thermal management is drastically lowered. Furthermore, the improved yield profile means that less starting material is required per kilogram of output, effectively reducing the raw material cost basis. The use of commodity chemicals like sodium carbonate and common organic solvents ensures that input costs remain stable and predictable, shielding the supply chain from volatility associated with specialized reagents.
- Enhanced Supply Chain Reliability: Sourcing consistency is greatly improved as the method relies on widely available, bulk-grade solvents and reagents rather than niche or hazardous chemicals like dioxane. The simplified workup procedure reduces the complexity of the manufacturing schedule, allowing for faster turnaround times between batches and increasing overall facility throughput. This agility enables suppliers to respond more rapidly to fluctuations in demand from API manufacturers, reducing the risk of stockouts or delivery delays. The high purity of the crude product also reduces the likelihood of downstream processing bottlenecks, ensuring a smooth flow of materials through the value chain.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the avoidance of extreme cryogenic conditions and the reduction in solvent handling steps align well with modern green chemistry principles. The process generates less waste solvent volume per unit of product due to the elimination of concentration steps, simplifying effluent treatment and disposal protocols. The formation of free-flowing, non-agglomerated solids facilitates safer handling and packaging, reducing dust generation and exposure risks for operators. These factors collectively make the process highly scalable from pilot plants to multi-ton commercial production lines without requiring extensive re-engineering of safety systems.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational parameters and quality outcomes expected when adopting this novel protective group strategy. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into existing manufacturing portfolios. The answers reflect the specific advantages in yield, purity, and processing ease that distinguish this method from legacy technologies.
Q: What are the advantages of the multi-solvent system in this synthesis?
A: The use of three or more solvents, including water, alcohol, and organic phases, significantly improves the solubility of both the polar starting material and the organic reagent, leading to yields exceeding 90% and purity above 95% without complex purification steps.
Q: How does this method improve safety compared to conventional processes?
A: By operating at a mild temperature range of 0-30°C, the process eliminates the need for extreme cryogenic cooling (below 10°C) required in older methods, thereby reducing energy consumption and thermal runaway risks during exothermic tosylation.
Q: Is the post-processing suitable for large-scale manufacturing?
A: Yes, the method allows for direct crystallization by adding an inert solvent without the need for energy-intensive concentration or distillation steps, resulting in non-agglomerated solids that are easy to filter and dry on an industrial scale.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3,2’,6’,3’’-penta-N-p-toluenesulfonyl kanamycin B Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of life-saving antibiotics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering this key aminoglycoside intermediate with stringent purity specifications, leveraging our rigorous QC labs to verify every batch against the highest pharmacopeial standards. Our facility is equipped to handle the specific solvent systems and safety requirements of this process, guaranteeing a consistent supply of material that meets the exacting needs of pharmaceutical partners.
We invite you to collaborate with us to optimize your supply chain for aminoglycoside antibiotic production. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
