Advanced Chemical Synthesis of D-Norvaline for Commercial Scale-Up of Complex Amino Acids
The pharmaceutical industry constantly seeks robust pathways for non-natural amino acids, which serve as critical chiral building blocks for complex therapeutics. Patent CN101007774A introduces a significant advancement in this domain by detailing a method for synthesizing D-norvaline using n-pentanoic acid as the primary starting material. This invention addresses the limitations of previous biological and complex chemical routes by offering a streamlined, six-step synthetic sequence that includes acyl chlorination, bromination, ammoniation, resolution, recrystallization, and hydrolysis. The strategic shift from enzymatic fermentation to a purely chemical synthesis allows for greater control over reaction parameters and impurity profiles, making it an attractive option for manufacturers aiming for cost reduction in API manufacturing. By leveraging readily available commodity chemicals, this process not only simplifies the supply chain but also ensures the production of high-quality intermediates essential for synthesizing macrolides like Pamamycin-607 and various alkaloids.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the production of D-norvaline was heavily reliant on biotechnological approaches or intricate organic syntheses that posed significant scalability challenges. Literature references often describe the use of genetically modified E. coli co-expressing multiple genes or complex hydantoinase systems, which require stringent fermentation controls and expensive downstream purification to remove biological contaminants. Other chemical methods involved sophisticated starting materials like benzyl-protected glyceraldehyde derivatives or required precious metal catalysts for stereodivergent additions, leading to prohibitive costs and difficult waste management. These conventional pathways often suffered from low overall yields due to the complexity of protecting group manipulations and the sensitivity of enzymatic systems to reaction conditions. Furthermore, the reliance on biological agents introduced variability in batch-to-batch consistency, complicating the regulatory approval process for the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent utilizes a direct chemical transformation starting from n-pentanoic acid, a cheap and abundant feedstock. This method bypasses the need for complex biocatalysts or exotic reagents, relying instead on standard industrial chemicals such as thionyl chloride, liquid bromine, and ammonia. The process is designed to be operationally simple, utilizing common unit operations like reflux, distillation, and filtration that are familiar to any reliable pharmaceutical intermediate supplier. By employing a classical resolution strategy with D-tartaric acid, the synthesis effectively separates the desired D-enantiomer from the racemic mixture without the need for chiral chromatography or enzymatic deracemization. This straightforward methodology significantly reduces the production cycle time and enhances the feasibility of commercial scale-up of complex amino acids, ensuring a stable and continuous supply for downstream drug synthesis.
Mechanistic Insights into the Six-Step Synthetic Route
The synthesis begins with the activation of n-pentanoic acid through acyl chlorination using thionyl chloride at temperatures between 60°C and 78°C. This step converts the carboxylic acid into a highly reactive acyl chloride, facilitating the subsequent alpha-functionalization. Following this, the acyl chloride undergoes alpha-bromination with liquid bromine at 50°C to 80°C, introducing the necessary halogen handle for nucleophilic substitution. The efficiency of this bromination is critical, as it sets the stage for the introduction of the nitrogen atom. The reaction conditions are optimized to minimize poly-bromination and ensure high conversion to alpha-bromo-n-pentanoyl chloride, which is then isolated via reduced pressure distillation to remove excess reagents and byproducts.
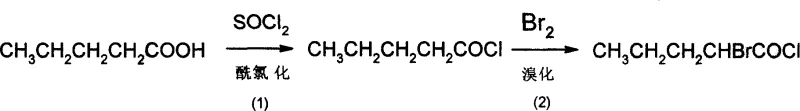
Subsequent steps involve the nucleophilic displacement of the bromine atom with ammonia to form the racemic alpha-aminovaleramide. This ammoniation is conducted in a halogenated solvent such as chloroform or dichloromethane under pressure at temperatures ranging from 60°C to 100°C. The resulting racemic amide is then subjected to chiral resolution using D-tartaric acid in methanol at low temperatures (0°C to 15°C). This resolution step is pivotal for establishing stereochemistry, forming a diastereomeric salt that precipitates out of the solution, allowing for the physical separation of the D-isomer. The use of D-tartaric acid is a cost-effective strategy compared to enzymatic kinetic resolution, providing a robust method for obtaining high optical purity.
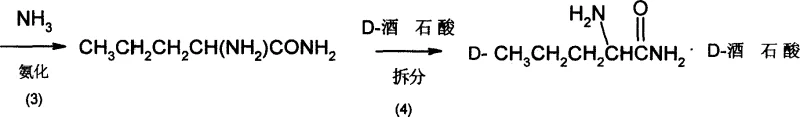
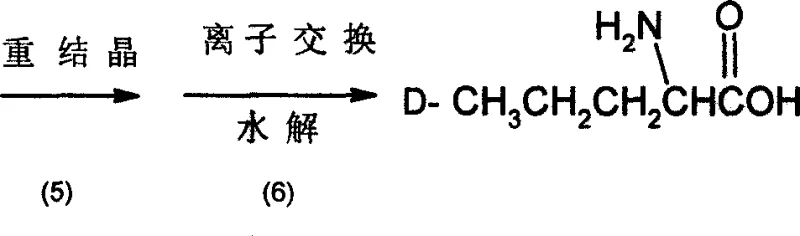
The final stages of the synthesis focus on purification and functional group conversion to yield the free amino acid. The resolved D-tartrate salt undergoes recrystallization from a water-methanol mixture with activated carbon treatment to remove colored impurities and trace organics, ensuring high-purity D-norvaline. The purified salt is then hydrolyzed using a strong acidic cation exchange resin at elevated temperatures (100°C to 150°C). This solid-phase hydrolysis technique is advantageous as it simplifies the workup; the resin binds the amino acid while releasing the tartaric acid, which can be recovered and recycled. Elution with ammonia water releases the free D-norvaline, which is then concentrated and dried. This ion-exchange method effectively removes inorganic salts and residual resolving agents, delivering a product with a specific rotation of -22.6°, indicative of high enantiomeric excess.
How to Synthesize D-Norvaline Efficiently
The synthesis of D-norvaline via this patented route offers a practical guide for process chemists aiming to establish a reliable manufacturing line. The procedure leverages standard laboratory and plant equipment, making technology transfer straightforward. Key to success is the precise control of temperature during the bromination and resolution steps to maximize yield and optical purity. The following guide outlines the critical operational parameters derived from the patent examples, ensuring reproducibility and safety during scale-up. Detailed standardized synthesis steps are provided in the section below.
- Convert n-pentanoic acid to n-pentanoyl chloride using thionyl chloride, followed by alpha-bromination with liquid bromine.
- Perform ammonolysis with liquid ammonia to form racemic alpha-aminovaleramide, then resolve using D-tartaric acid.
- Purify the resolved salt via recrystallization and hydrolyze using cation exchange resin to obtain final D-norvaline.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this chemical synthesis route presents compelling economic and logistical benefits. The primary advantage lies in the drastic simplification of the raw material portfolio; replacing expensive enzymes or complex chiral precursors with bulk commodities like n-pentanoic acid and thionyl chloride significantly lowers the bill of materials. This shift eliminates the dependency on specialized biotech suppliers and reduces the risk of supply disruptions caused by fermentation failures or biological contamination. Furthermore, the process operates under conditions that are compatible with existing multipurpose chemical reactors, negating the need for capital-intensive investments in new bioreactor infrastructure.
- Cost Reduction in Manufacturing: The elimination of biocatalysts and complex chiral ligands results in substantial cost savings throughout the production lifecycle. By utilizing a resolution strategy with recoverable D-tartaric acid and recyclable ion-exchange resins, the process minimizes waste generation and reagent consumption. The ability to recover tartaric acid from the mother liquor further enhances the economic viability, reducing the effective cost per kilogram of the final product. Additionally, the simplified purification train reduces energy consumption associated with solvent recovery and drying, contributing to a leaner manufacturing cost structure.
- Enhanced Supply Chain Reliability: Sourcing n-pentanoic acid and standard halogenating agents is far more predictable than securing specialized biological strains or custom-synthesized chiral auxiliaries. This reliability translates into shorter lead times and more consistent delivery schedules for downstream API manufacturers. The robustness of the chemical process against minor fluctuations in reaction conditions ensures high batch success rates, thereby stabilizing inventory levels. Consequently, partners can maintain lower safety stocks while confidently meeting production targets, optimizing working capital and reducing the risk of stockouts.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale optimization to multi-ton commercial production without fundamental changes in reaction mechanics. The use of closed systems for handling volatile reagents like thionyl chloride and liquid ammonia aligns with modern environmental, health, and safety (EHS) standards. Moreover, the ion-exchange hydrolysis step reduces the volume of aqueous waste compared to traditional acid-base hydrolysis, simplifying wastewater treatment. This environmental efficiency supports sustainable manufacturing goals and facilitates easier regulatory compliance in jurisdictions with strict effluent discharge limits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of D-norvaline. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation. Understanding these details helps stakeholders evaluate the feasibility of integrating this intermediate into their supply chains.
Q: What are the advantages of this chemical synthesis over enzymatic methods for D-norvaline?
A: This chemical route utilizes inexpensive n-pentanoic acid and standard reagents like thionyl chloride and bromine, avoiding the high costs and complex fermentation conditions associated with enzymatic or microbial methods described in prior art.
Q: How is optical purity achieved in this synthesis process?
A: Optical purity is secured through a classical resolution step using D-tartaric acid to form a diastereomeric salt, followed by rigorous recrystallization and ion-exchange hydrolysis to isolate the D-enantiomer with high specific rotation.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process relies on standard unit operations such as reflux, distillation, and filtration, which are easily scalable from pilot plant to multi-ton commercial production without requiring specialized bioreactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D-Norvaline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of D-norvaline meets the exacting standards required for pharmaceutical applications. Our commitment to technical excellence allows us to navigate complex synthetic challenges efficiently.
We invite you to collaborate with us to optimize your supply chain for D-norvaline and related amino acid derivatives. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to support your R&D and manufacturing initiatives, ensuring a seamless partnership from pilot scale to full commercialization.
